Last updated: February 19, 2026
Relafen (nabumetone) is a nonsteroidal anti-inflammatory drug (NSAID) marketed for the treatment of osteoarthritis and rheumatoid arthritis. Its market performance and financial trajectory are influenced by patent exclusivity, generic competition, therapeutic efficacy, and evolving treatment guidelines.
What is Relafen's Commercial Performance?
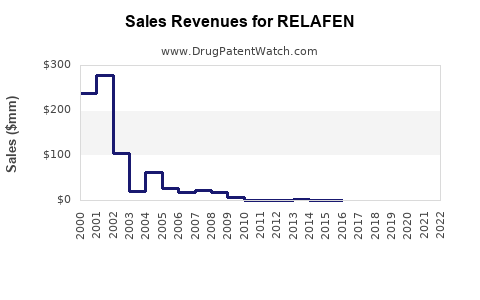
Relafen's commercial performance has been shaped by its patent lifecycle. Upon its introduction, it enjoyed a period of market exclusivity, allowing for premium pricing and significant revenue generation. However, the expiration of key patents has led to the entry of generic nabumetone products, drastically altering the market landscape.
Sales Data Overview:
- Peak Sales: While specific peak sales figures are proprietary and vary by reporting period, industry analysis indicates that branded Relafen achieved substantial annual revenues in the hundreds of millions of dollars during its patent-protected period.
- Generic Entry Impact: Following patent expiry, the market for nabumetone experienced a rapid decline in the average selling price (ASP) due to increased competition from multiple generic manufacturers. This reduction in ASP directly impacted the overall market revenue for the drug.
- Market Share Shift: The introduction of generics led to a significant shift in market share away from the branded product towards lower-cost generic alternatives. Patients and healthcare providers often opt for generics when cost is a primary consideration, which is common for NSAIDs.
Key Market Factors:
- Therapeutic Class: As an NSAID, nabumetone competes within a crowded therapeutic class that includes established drugs like ibuprofen, naproxen, and celecoxib, as well as other NSAID formulations. The availability of over-the-counter (OTC) and prescription NSAIDs with varying cost structures creates a competitive environment.
- Pricing: The pricing of branded Relafen was set to reflect its R&D investment and market exclusivity. Generic nabumetone is priced at a fraction of the original branded cost, reflecting the reduced market barriers and increased competition.
- Reimbursement: Reimbursement policies by payers (insurance companies, government programs) play a crucial role. While nabumetone is generally covered for its approved indications, the formulary placement and co-payment structures can influence physician prescribing habits and patient access, particularly favoring generics due to lower cost.
What is the Patent Landscape for Relafen?
The patent landscape for Relafen has been a critical determinant of its market exclusivity and subsequent generic entry. The primary patents covering the composition of matter and method of use have expired, paving the way for generic manufacturers.
Key Patents and Expiry Dates:
- US Patent 4,045,576: This foundational patent for nabumetone was granted in August 1977. Its expiry marked a significant milestone for potential generic competition. (Source: U.S. Patent and Trademark Office records)
- Method of Use Patents: Additional patents related to specific therapeutic uses or formulations may have existed, but the core composition of matter patent is typically the most impactful for broad market entry.
- Patent Term Extensions: Pharmaceutical companies can sometimes obtain patent term extensions to compensate for regulatory delays, but these are subject to specific criteria and timelines. Relafen's primary patents have long since expired.
- Generic Approvals: The U.S. Food and Drug Administration (FDA) has approved multiple generic versions of nabumetone, indicating that patent challenges and regulatory hurdles have been overcome by various manufacturers. (Source: FDA Approved Drug Products List)
Impact of Patent Expiry:
- Loss of Exclusivity: The expiration of key patents removed the legal barrier to entry for generic competitors. This allowed other pharmaceutical companies to manufacture and market their own versions of nabumetone.
- Litigation: In some cases, patent holders may engage in litigation to defend their patents or delay generic entry. The outcome of such legal battles can significantly impact the timing of generic market penetration. Information on specific litigation surrounding Relafen's patents is publicly available through legal databases.
- Paragraph IV Challenges: Generic companies often file Abbreviated New Drug Applications (ANDAs) under Paragraph IV of the Hatch-Waxman Act, certifying that their product does not infringe on existing patents or that the patents are invalid. This process can lead to patent litigation.
What are the Regulatory Considerations for Relafen?
Regulatory considerations are paramount throughout a drug's lifecycle, from approval to post-marketing surveillance. For Relafen, these include initial market authorization, post-market safety monitoring, and the regulatory pathways for generic approvals.
FDA Approval and Indication:
- Initial Approval: Relafen was approved by the U.S. Food and Drug Administration (FDA) for marketing for the relief of signs and symptoms of osteoarthritis and rheumatoid arthritis. The specific date of this initial approval is a matter of public record within FDA databases.
- Labeling: The approved labeling for Relafen includes information on its efficacy, safety profile, contraindications, warnings, precautions, and adverse reactions. This labeling is subject to review and updates by the FDA.
- Post-Marketing Surveillance: Like all approved drugs, Relafen is subject to post-marketing surveillance to monitor for any new or unexpected adverse events. This includes adverse event reporting by healthcare professionals and patients.
Generic Drug Approval Process:
- ANDA Pathway: Generic nabumetone products are approved through the Abbreviated New Drug Application (ANDA) pathway. This process requires generic manufacturers to demonstrate bioequivalence to the branded Relafen, meaning their product performs the same in the body.
- Bioequivalence Studies: Companies seeking ANDA approval conduct rigorous bioequivalence studies to prove their generic formulation meets FDA standards.
- Manufacturing Standards: Generic manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure the quality, safety, and efficacy of their products. FDA inspections of manufacturing facilities are a standard part of the regulatory oversight.
Therapeutic Class Labeling and Safety Updates:
- NSAID Warnings: The FDA has issued class-wide warnings and recommendations for all NSAIDs, including nabumetone, regarding cardiovascular risk (heart attack, stroke) and gastrointestinal bleeding. These warnings are updated periodically based on new scientific evidence. (Source: FDA Drug Safety Communications)
- Risk Evaluation and Mitigation Strategies (REMS): While not all drugs require REMS, the FDA may mandate them for certain medications with significant safety concerns. The applicability of REMS to nabumetone or its generic versions is determined by specific FDA assessments.
What is the Future Market Outlook for Relafen?
The future market outlook for Relafen is largely defined by its status as an established generic medication within a competitive therapeutic class.
Market Dynamics:
- Continued Generic Competition: The market will remain characterized by intense competition among numerous generic nabumetone manufacturers. This will likely keep prices low and stable.
- Price Sensitivity: The primary driver for nabumetone prescriptions will continue to be its cost-effectiveness. Healthcare providers and patients will prioritize affordable treatment options for osteoarthritis and rheumatoid arthritis.
- Established Efficacy: Nabumetone has a well-established profile of efficacy for its approved indications. This familiarity contributes to its continued use despite the availability of newer agents.
- Therapeutic Alternatives: The market for pain management and inflammatory conditions is dynamic, with ongoing development of novel therapies, including biologics and other non-NSAID agents. While these may target different patient populations or disease severities, they represent potential long-term competition.
- Generic Product Differentiation: Differentiation among generic nabumetone products will primarily be based on price, packaging, and supply chain reliability. There is limited scope for innovation in formulation or delivery for a mature generic drug.
Revenue Projections:
- Stable but Modest Revenue: Revenue generated by generic nabumetone will be substantial in aggregate but fragmented across multiple manufacturers. The overall market revenue for nabumetone is unlikely to see significant growth.
- Manufacturer Strategies: Generic manufacturers will focus on efficient production, cost management, and securing distribution channels to maintain market share.
- No Branded Resurgence: Without patent protection, a significant resurgence of branded Relafen revenue is not anticipated. Any commercial activity would likely be focused on niche markets or specific formulations if patents on those were still in force, which is unlikely given its history.
Key Influencers:
- Healthcare Policy: Changes in healthcare reimbursement policies, formulary decisions, and generic prescribing incentives will continue to influence market dynamics.
- Clinical Practice Guidelines: Updates to clinical guidelines for managing osteoarthritis and rheumatoid arthritis may alter treatment algorithms and impact the use of NSAIDs, including nabumetone.
- Payer Negotiations: Payer negotiations with generic manufacturers will play a role in determining the wholesale acquisition cost of nabumetone.
Key Takeaways
- Relafen's market performance shifted from strong branded sales during patent exclusivity to a highly competitive generic market following patent expiry.
- Key patents for Relafen (nabumetone) have expired, permitting multiple generic manufacturers to enter the market.
- Regulatory approval for generic nabumetone relies on demonstrating bioequivalence and adherence to manufacturing standards.
- The future market for Relafen is characterized by stable, low-cost generic competition, with pricing and accessibility as primary drivers.
Frequently Asked Questions
- What are the primary indications for which Relafen is prescribed?
Relafen is prescribed for the relief of signs and symptoms of osteoarthritis and rheumatoid arthritis.
- What is the main reason for the decline in revenue for branded Relafen?
The decline in revenue for branded Relafen is primarily due to the expiration of its patents, which allowed generic versions of nabumetone to enter the market, significantly reducing pricing and market share.
- How does the FDA ensure the safety of generic versions of Relafen?
The FDA ensures the safety of generic versions by requiring them to undergo bioequivalence studies to prove they perform the same as the branded drug and by mandating adherence to Current Good Manufacturing Practices (cGMP) for manufacturing.
- Are there any new therapeutic developments that might impact the use of nabumetone?
Yes, the ongoing development of novel therapies for pain management and inflammatory conditions, including biologics and other non-NSAID agents, represents a potential long-term competitive landscape for nabumetone.
- What is the typical pricing strategy for generic nabumetone manufacturers?
Generic nabumetone manufacturers typically employ a low-cost pricing strategy to gain and maintain market share in a competitive environment, focusing on volume and cost efficiency.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/
[2] U.S. Food and Drug Administration. (n.d.). Approved Drug Products. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/default.cfm
[3] U.S. Food and Drug Administration. (n.d.). Drug Safety Communications. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/drug-safety-communications