RAYALDEE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Rayaldee, and what generic alternatives are available?
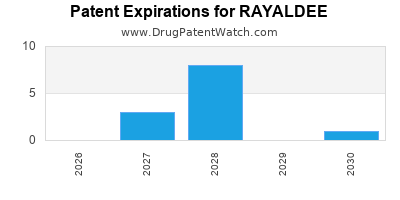
Rayaldee is a drug marketed by Eirgen and is included in one NDA. There are sixteen patents protecting this drug.
This drug has one hundred and ninety-two patent family members in thirty-eight countries.
The generic ingredient in RAYALDEE is calcifediol. There are two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the calcifediol profile page.
DrugPatentWatch® Generic Entry Outlook for Rayaldee
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be March 14, 2034. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for RAYALDEE?
- What are the global sales for RAYALDEE?
- What is Average Wholesale Price for RAYALDEE?
Summary for RAYALDEE
| International Patents: | 192 |
| US Patents: | 16 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 75 |
| Clinical Trials: | 1 |
| Patent Applications: | 3,406 |
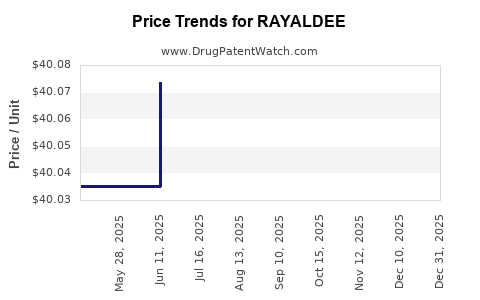
| Drug Prices: | Drug price information for RAYALDEE |
| What excipients (inactive ingredients) are in RAYALDEE? | RAYALDEE excipients list |
| DailyMed Link: | RAYALDEE at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for RAYALDEE
Generic Entry Date for RAYALDEE*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE, EXTENDED RELEASE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for RAYALDEE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| OPKO Health, Inc. | Phase 2 |
Pharmacology for RAYALDEE
| Drug Class | Vitamin D3 Analog |
US Patents and Regulatory Information for RAYALDEE
RAYALDEE is protected by twenty-two US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of RAYALDEE is ⤷ Start Trial.
This potential generic entry date is based on patent 11,253,528.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | RX | Yes | Yes | 8,778,373 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | RX | Yes | Yes | 10,300,078 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | RX | Yes | Yes | 9,925,147 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | RX | Yes | Yes | 10,213,442 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | RX | Yes | Yes | 8,426,391 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | RX | Yes | Yes | 10,357,502 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for RAYALDEE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Eirgen | RAYALDEE | calcifediol | CAPSULE, EXTENDED RELEASE;ORAL | 208010-001 | Jun 17, 2016 | 6,582,727 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for RAYALDEE
When does loss-of-exclusivity occur for RAYALDEE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 5576
Patent: FORMULACIÓN DE VITAMINA D DE LIBERACIÓN MODIFICADA ESTABILIZADA Y MÉTODO PARA ADMINISTRARLA
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 14228069
Patent: Stabilized modified release vitamin D formulation and method of administring same
Estimated Expiration: ⤷ Start Trial
Patent: 19200268
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2015023658
Patent: formulação de vitamina d de liberação modificada, estabilizada e método de administração desta
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 05409
Patent: FORMULATION DE VITAMINE D A LIBERATION MODIFIEE STABILISEE ET SON PROCEDE D'ADMINISTRATION (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 15002659
Patent: Formulación de vitamina d de liberación modificada estabilizada y método de administración de la misma
Estimated Expiration: ⤷ Start Trial
China
Patent: 5246464
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME
Estimated Expiration: ⤷ Start Trial
Patent: 1346071
Patent: 稳定化修饰释放维生素D制剂和施用其的方法 (Stabilized modified release vitamin D formulation and method of administring same)
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 190178
Patent: FORMULACIÓN DE VITAMINA D DE LIBERACIÓN MODIFICADA Y ESTABILIZADA Y MÉTODO DE ADMINISTRACIÓN DE LA MISMA
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0201284
Estimated Expiration: ⤷ Start Trial
Patent: 0201869
Estimated Expiration: ⤷ Start Trial
Patent: 0211265
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 23167
Estimated Expiration: ⤷ Start Trial
Patent: 23568
Estimated Expiration: ⤷ Start Trial
Patent: 24393
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 68172
Estimated Expiration: ⤷ Start Trial
Patent: 32773
Estimated Expiration: ⤷ Start Trial
Patent: 50016
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 23024864
Patent: FORMULACIÓN DE VITAMINA D DE LIBERACIÓN MODIFICADA Y ESTABILIZADA Y MÉTODO DE ADMINISTRACIÓN DE LA MISMA
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8867
Patent: СТАБИЛИЗИРОВАННЫЙ СОСТАВ ВИТАМИНА D С МОДИФИЦИРОВАННЫМ ВЫСВОБОЖДЕНИЕМ (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION)
Estimated Expiration: ⤷ Start Trial
Patent: 1591809
Patent: СТАБИЛИЗИРОВАННЫЙ СОСТАВ ВИТАМИНА D С МОДИФИЦИРОВАННЫМ ВЫСВОБОЖДЕНИЕМ И СПОСОБ ЕГО ВВЕДЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 1991774
Patent: СТАБИЛИЗИРОВАННЫЙ СОСТАВ ВИТАМИНА D С МОДИФИЦИРОВАННЫМ ВЫСВОБОЖДЕНИЕМ И СПОСОБ ЕГО ВВЕДЕНИЯ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 68172
Patent: FORMULATION DE VITAMINE D À LIBÉRATION MODIFIÉE STABILISÉE ET SON PROCÉDÉ D'ADMINISTRATION (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 32773
Patent: FORMULATION DE VITAMINE D À LIBÉRATION MODIFIÉE STABILISÉE ET SON PROCÉDÉ D'ADMINISTRATION (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 50016
Patent: FORMULATION DE VITAMINE D À LIBÉRATION MODIFIÉE STABILISÉE ET SON PROCÉDÉ D'ADMINISTRATION (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 88638
Patent: FORMULATION DE VITAMINE D À LIBÉRATION MODIFIÉE STABILISÉE ET SON PROCÉDÉ D'ADMINISTRATION (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Germany
Patent: 2014011525
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 20128
Patent: 穩定化修飾釋放維生素 製劑和施用其的方法 (STABILIZED MODIFIED RELEASE VITAMIN FORMULATION AND METHOD OF ADMINISTRING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 20362
Patent: 穩定化修飾釋放維生素 製劑和施用其的方法 (STABILIZED MODIFIED RELEASE VITAMIN FORMULATION AND METHOD OF ADMINISTRING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 56895
Patent: 穩定化修飾的釋放維生素D製劑及其給藥方法 (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 51923
Estimated Expiration: ⤷ Start Trial
Patent: 52014
Estimated Expiration: ⤷ Start Trial
Patent: 55591
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 1456
Patent: פורמולציה מיוצבת ובעלת שחרור מבוקר של ויטמין, d , ושיטות לנתינתה (Stabilized modified release vitamin d formulation and method of administring same)
Estimated Expiration: ⤷ Start Trial
Patent: 4841
Patent: פורמולציה מיוצבת ובעלת שחרור מבוקר של ויטמין d , ושיטות לנתינתה (Stabilized modified release vitamin d formulation and method of administering same)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 92051
Estimated Expiration: ⤷ Start Trial
Patent: 33268
Estimated Expiration: ⤷ Start Trial
Patent: 82832
Estimated Expiration: ⤷ Start Trial
Patent: 16517429
Patent: 安定化調整放出ビタミンD製剤及びその投与方法
Estimated Expiration: ⤷ Start Trial
Patent: 18012737
Patent: 安定化調整放出ビタミンD製剤及びその投与方法 (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING THE SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 19135264
Patent: 安定化調整放出ビタミンD製剤及びその投与方法 (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING THE SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 21155460
Patent: 安定化調整放出ビタミンD製剤及びその投与方法 (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING THE SAME)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 68172
Estimated Expiration: ⤷ Start Trial
Patent: 32773
Estimated Expiration: ⤷ Start Trial
Patent: 50016
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 4092
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Patent: 0794
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 15012625
Patent: FORMULACION DE VITAMINA D DE LIBERACION MODIFICADA ESTABILIZADA Y METODO PARA ADMINISTRATIVA Y METODO PARA ADMINISTRAR LA MISMA. (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME.)
Estimated Expiration: ⤷ Start Trial
Patent: 20011736
Patent: FORMULACION DE VITAMINA D DE LIBERACION MODIFICADA ESTABILIZADA Y METODO PARA ADMINISTRAR LA MISMA. (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 1924
Patent: Stabilized modified release vitamin d formulation and method of administering same
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 21007
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 151761
Patent: FORMULACION DE VITAMINA D DE LIBERACION MODIFICADA Y ESTABILIZADA Y METODO DE ADMINISTRACION DE LA MISMA
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 015502162
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Patent: 021551127
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 68172
Estimated Expiration: ⤷ Start Trial
Patent: 32773
Estimated Expiration: ⤷ Start Trial
Patent: 50016
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 68172
Estimated Expiration: ⤷ Start Trial
Patent: 32773
Estimated Expiration: ⤷ Start Trial
Patent: 50016
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 02000424
Estimated Expiration: ⤷ Start Trial
Patent: 02000651
Estimated Expiration: ⤷ Start Trial
Patent: 02100439
Estimated Expiration: ⤷ Start Trial
Saudi Arabia
Patent: 5361134
Patent: صيغة مستقرة ذات إطلاق متحكم فيه لمركب فيتامين د وطريقة إعطائها (Stabilized controlled release formulation of compound vitamin d and method of administering same)
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 846
Patent: STABILIZOVANA FORMULACIJA VITAMINA D SA MODIFIKOVANIM OSLOBAĐANJEM I POSTUPAK ZA DAVANJE ISTE (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 132
Patent: STABILIZOVANA FORMULACIJA VITAMINA D SA MODIFIKOVANIM OSLOBAĐANJEM I POSTUPAK ZA NJENU PRIMENU (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 176
Patent: STABILIZOVANA FORMULACIJA VITAMINA D SA MODIFIKOVANIM OSLOBAĐANJEM I POSTUPAK ZA NJENU PRIMENU (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201703517V
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Patent: 201507323P
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 68172
Estimated Expiration: ⤷ Start Trial
Patent: 32773
Estimated Expiration: ⤷ Start Trial
Patent: 50016
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1847947
Estimated Expiration: ⤷ Start Trial
Patent: 2203003
Estimated Expiration: ⤷ Start Trial
Patent: 140113374
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Patent: 140140004
Patent: STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME
Estimated Expiration: ⤷ Start Trial
Patent: 190095216
Patent: 안정화되고 변형된 비타민 D 방출 제형 및 이의 투여 방법 (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 210078463
Patent: 안정화되고 변형된 비타민 D 방출 제형 및 이의 투여 방법 (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTERING SAME)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 09477
Estimated Expiration: ⤷ Start Trial
Patent: 34900
Estimated Expiration: ⤷ Start Trial
Patent: 82567
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1707689
Patent: Stabilized modified release vitamin D formulation and method of administering same
Estimated Expiration: ⤷ Start Trial
Patent: 59753
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3386
Patent: СТАБІЛІЗОВАНИЙ СКЛАД ВІТАМІНУ D ІЗ МОДИФІКОВАНИМ ВИВІЛЬНЕННЯМ І СПОСІБ ЙОГО ВВЕДЕННЯ (STABILIZED MODIFIED RELEASE VITAMIN D FORMULATION AND METHOD OF ADMINISTRING SAME)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering RAYALDEE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Chile | 2015002659 | ⤷ Start Trial | |
| Finland | 3335712 | ⤷ Start Trial | |
| Eurasian Patent Organization | 201991774 | ⤷ Start Trial | |
| Hungary | E055591 | ⤷ Start Trial | |
| South Korea | 20170085141 | ⤷ Start Trial | |
| Japan | 5931042 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for RAYALDEE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2481400 | CR 2020 00059 | Denmark | ⤷ Start Trial | PRODUCT NAME: CALCIFEDIOL; NAT. REG. NO/DATE: 62564 20200910; FIRST REG. NO/DATE: DE 2202115.00.00 20200818 |
| 2481400 | 122020000079 | Germany | ⤷ Start Trial | FORMER OWNERS: OPKO IRELAND GLOBAL HOLDINGS, LTD., DUBLIN 24, IE; OPKO RENAL, LLC, MIAMI, FL 33137, US |
| 2481400 | 301085 | Netherlands | ⤷ Start Trial | PRODUCT NAME: CALCIFEDIOL IN IEDERE VORM ZOALS BESCHERMD DOOR HET BASISOCTROOI; NATIONAL REGISTRATION NO/DATE: 124799 20200922; FIRST REGISTRATION: DE 2202115.00.00 20200819 |
| 2968172 | 132021000000074 | Italy | ⤷ Start Trial | PRODUCT NAME: CALCIFEDIOLO(RAYALDEE); AUTHORISATION NUMBER(S) AND DATE(S): 047870011, 20201201;PL 50784/0005, 20200721 |
| 2968172 | 301095 | Netherlands | ⤷ Start Trial | PRODUCT NAME: CALCIFEDIOL IN IEDERE VORM ZOALS BESCHERMD DOOR HET BASISOCTROOI; NATIONAL REGISTRATION NO/DATE: 124799 20200922; FIRST REGISTRATION: DE 2202115.00.00 20200819 |
| 2968172 | CR 2021 00005 | Denmark | ⤷ Start Trial | PRODUCT NAME: CALCIFEDIOL; NAT. REG. NO/DATE: 62564 20200910; FIRST REG. NO/DATE: DE 2202115.00.00 20200818 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for RAYALDEE
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.