QUINAPRIL Drug Patent Profile
✉ Email this page to a colleague
When do Quinapril patents expire, and when can generic versions of Quinapril launch?
Quinapril is a drug marketed by Actavis Elizabeth, Actavis Labs Fl Inc, Ani Pharms, Apotex Inc, Aurobindo Pharma Ltd, Chartwell Rx, Invagen Pharms, Lupin, Mylan, Pharmobedient, Prinston Inc, Sun Pharm Inds Ltd, Apotex, and Aurobindo Pharma. and is included in nineteen NDAs.
The generic ingredient in QUINAPRIL is hydrochlorothiazide; quinapril hydrochloride. There are thirty-two drug master file entries for this compound. Four suppliers are listed for this compound. Additional details are available on the hydrochlorothiazide; quinapril hydrochloride profile page.
AI Deep Research
Questions you can ask:
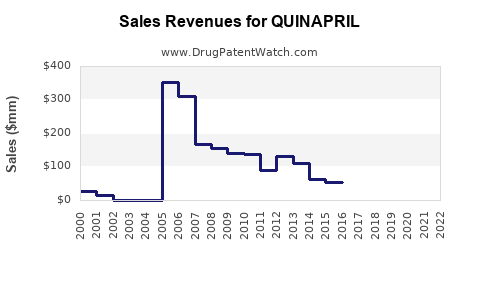
- What is the 5 year forecast for QUINAPRIL?
- What are the global sales for QUINAPRIL?
- What is Average Wholesale Price for QUINAPRIL?
Summary for QUINAPRIL
| US Patents: | 0 |
| Applicants: | 14 |
| NDAs: | 19 |
| Drug Prices: | Drug price information for QUINAPRIL |
| DailyMed Link: | QUINAPRIL at DailyMed |