Last updated: February 19, 2026
Pyrimethamine, an antiparasitic medication, exhibits a nuanced market position driven by its established efficacy against specific protozoal infections and evolving competitive landscapes. Its primary applications remain in treating toxoplasmosis and malaria, often in combination therapies. The market's financial trajectory is influenced by generic competition, regulatory actions, and the development of alternative treatments.
What is the current market size and projected growth for pyrimethamine?
The global pyrimethamine market is a mature segment within the antiparasitic drug class. Precise current market size figures are not publicly disclosed by individual manufacturers due to the drug's status as a generic. However, market analysis indicates a steady, albeit modest, growth trajectory. This growth is primarily sustained by its critical role in treating neglected tropical diseases (NTDs) and its continued use in specific patient populations.
Projected annual growth rates for the pyrimethamine market are estimated to be between 2% and 4% over the next five to seven years. This projection is based on the persistent prevalence of diseases like toxoplasmosis, particularly in immunocompromised individuals (e.g., those with HIV/AIDS or undergoing chemotherapy), and its inclusion in essential medicine lists by organizations such as the World Health Organization (WHO) [1]. The ongoing efforts to control malaria in endemic regions also contribute to sustained demand.
Who are the key manufacturers and suppliers of pyrimethamine?
The pyrimethamine market is characterized by the presence of several generic manufacturers, reflecting its patent expiry. Key global suppliers include:
- Dar Pharmaceutical Limited: A significant player in the production of generic pharmaceuticals, including pyrimethamine.
- Emtricitabine: While Emtricitabine is more known for HIV treatment, companies producing a broader range of antiviral and antiparasitic agents may include pyrimethamine in their portfolios.
- Sanofi S.A.: Historically, Sanofi has been involved in the production and distribution of pyrimethamine-based products.
- Teva Pharmaceutical Industries Ltd.: As a leading generic drug manufacturer, Teva is a potential supplier.
- Viatris Inc.: Formed by the merger of Mylan and Upjohn (a Pfizer division), Viatris is a major global supplier of generic medicines.
These manufacturers primarily operate in markets where pyrimethamine remains a cost-effective and essential treatment. Production is often concentrated in regions with established pharmaceutical manufacturing capabilities and regulatory frameworks that support generic drug production.
What are the primary therapeutic indications for pyrimethamine?
Pyrimethamine is primarily indicated for the treatment and prophylaxis of parasitic infections caused by protozoa. Its main therapeutic uses include:
- Toxoplasmosis: Pyrimethamine is a cornerstone treatment for toxoplasmosis, an infection caused by Toxoplasma gondii. It is frequently used in combination with sulfadiazine or clindamycin. This is particularly crucial for individuals with weakened immune systems, such as those with HIV/AIDS, organ transplant recipients, and patients undergoing chemotherapy. It is also used to treat congenital toxoplasmosis in newborns.
- Malaria: Historically, pyrimethamine was a significant antimalarial drug. While its monotherapy use for malaria has declined due to widespread resistance, it remains relevant in combination therapies, such as pyrimethamine/sulfadoxine (Fansidar), for certain forms of malaria or in specific geographical regions where resistance patterns permit its use.
- Other Protozoal Infections: In some instances, pyrimethamine may be used off-label or as part of combination regimens for other protozoal infections, though these indications are less common and often require specialist consultation.
What is the patent landscape for pyrimethamine?
Pyrimethamine was first patented in the mid-20th century. The original patents have long expired, making the drug a generic compound. Consequently, there are no active composition of matter patents that would prevent the generic manufacturing and sale of pyrimethamine.
The patent landscape is now primarily characterized by potential secondary patents related to:
- Formulation Improvements: Novel drug delivery systems, sustained-release formulations, or specific combinations with other active pharmaceutical ingredients (APIs) might be subject to patent protection.
- Manufacturing Processes: Improvements or novel methods for synthesizing pyrimethamine or its intermediates could be patented.
- New Indications: While less common for an established drug, new therapeutic uses discovered and validated for pyrimethamine could, in theory, be patentable, though such developments are rare for drugs with such a long history.
The absence of primary patents allows any pharmaceutical manufacturer with the capability to meet regulatory standards to produce and market generic pyrimethamine, contributing to market competition and price sensitivity.
What are the key regulatory considerations and challenges for pyrimethamine?
Regulatory bodies worldwide classify pyrimethamine as an essential medicine, facilitating its availability. However, several regulatory considerations and challenges exist:
- Generic Drug Approval Pathways: Manufacturers seeking to market generic pyrimethamine must demonstrate bioequivalence to the reference listed drug (RLD) through abbreviated new drug applications (ANDAs) or equivalent regulatory pathways in different jurisdictions.
- Quality Control and Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) is paramount. Regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) conduct rigorous inspections to ensure product quality, purity, and consistency.
- Labeling and Prescribing Information: Regulatory agencies mandate specific labeling requirements, including indications, contraindications, warnings, precautions, and adverse reactions, to ensure safe and effective use.
- Supply Chain Integrity: Ensuring the integrity of the global supply chain for pyrimethamine is a continuous regulatory focus, particularly in preventing counterfeiting and ensuring consistent availability, especially for public health programs.
- Antimicrobial Resistance Monitoring: For its use in malaria, regulatory agencies and public health bodies monitor resistance patterns. Changes in susceptibility can impact product recommendations and, indirectly, regulatory approval for specific indications or combinations.
- Pediatric Use Considerations: Regulatory bodies often require specific studies or data regarding the safety and efficacy of drugs in pediatric populations, which can be a factor for pyrimethamine given its use in congenital toxoplasmosis.
How does competition and the development of alternative therapies impact pyrimethamine's market share?
Competition in the pyrimethamine market is dominated by generic manufacturers, leading to price erosion and a focus on cost-efficiency in production. The main competitive dynamics include:
- Price Competition: With multiple generic suppliers, pricing is a significant factor influencing market share. Manufacturers with lower production costs and efficient distribution networks gain a competitive edge.
- Availability and Distribution: Consistent availability, especially in resource-limited settings, is crucial. Manufacturers with robust supply chains and established distribution channels are favored.
- Combination Therapies: Pyrimethamine's efficacy is often enhanced when used in combination with other drugs. The availability and market penetration of these fixed-dose combinations (FDCs) or co-packaged products influence pyrimethamine demand. For instance, pyrimethamine/sulfadoxine is a common antimalarial combination.
The development of alternative therapies presents a more significant long-term challenge:
- Newer Antimalarials: The emergence of artemisinin-based combination therapies (ACTs) has largely replaced older antimalarial drugs, including pyrimethamine monotherapy, in many regions due to improved efficacy and reduced resistance development [2]. However, pyrimethamine-based combinations might retain niche roles.
- Alternative Treatments for Toxoplasmosis: While pyrimethamine remains a standard of care for toxoplasmosis, research into novel antiparasitic agents and improved treatment regimens is ongoing. New drugs with better safety profiles or efficacy against resistant strains could eventually impact pyrimethamine's dominance.
- Prophylactic Strategies: Advances in diagnostics and preventative measures for toxoplasmosis, particularly in pregnant women and immunocompromised individuals, could indirectly influence the demand for treatment.
What is the pricing strategy and revenue generation for pyrimethamine products?
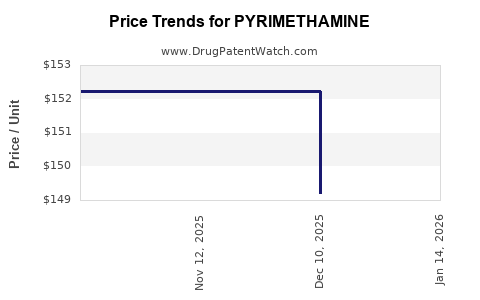
As a mature generic drug, pyrimethamine's pricing strategy is primarily driven by cost-plus models and competitive market forces.
- Generic Pricing: The price of generic pyrimethamine is significantly lower than its original branded counterpart. Manufacturers aim to achieve profitability through high sales volumes rather than premium pricing. Pricing is often determined by manufacturing costs, regulatory compliance expenses, and the prevailing market rates set by competitors.
- Public Health Programs: A substantial portion of pyrimethamine demand originates from public health initiatives, national formularies, and tenders from government health ministries and international organizations like the WHO and UNICEF. These programs often procure drugs at highly competitive prices, reflecting the need to maximize public health impact with limited budgets.
- Wholesale and Retail Markups: Revenue is generated through wholesale distribution to pharmacies, hospitals, and clinics. Retail markups then apply at the point of dispensing to the end consumer.
- Geographic Variations: Pricing can vary significantly by region due to differences in manufacturing costs, local market competition, import duties, and currency exchange rates.
- Combination Products: The revenue generated from pyrimethamine in combination products will be influenced by the pricing of the co-administered API and the overall market acceptance of the combination.
Specific revenue figures for individual pyrimethamine products are not typically disclosed by generic manufacturers. However, the overall revenue for pyrimethamine as a drug class is linked to its steady demand in essential medicine applications and its cost-effectiveness.
What are the future outlook and potential innovations for pyrimethamine?
The future outlook for pyrimethamine is characterized by continued reliance on its established therapeutic roles, particularly in specific populations and for neglected diseases, coupled with a limited scope for significant innovation.
- Sustained Demand in NTDs: Pyrimethamine is likely to maintain its position as a critical component in treating toxoplasmosis and potentially certain forms of malaria, especially in regions where alternative treatments are less accessible or affordable. Its inclusion on essential medicines lists reinforces this outlook.
- Focus on Accessibility and Affordability: Future efforts will likely concentrate on ensuring continued access to affordable pyrimethamine, particularly for low- and middle-income countries. This may involve optimizing manufacturing processes to further reduce costs and strengthening supply chain resilience.
- Limited Pharmaceutical Innovation: Given its generic status and long history, the likelihood of significant pharmaceutical innovation directly related to pyrimethamine itself (e.g., novel delivery systems or new molecular entities based on its structure) is low. Most research and development in antiparasitic therapy are focused on novel drug targets and mechanisms to combat resistance.
- Potential for Repurposing: While not a primary focus, like any established drug, there is a theoretical possibility of repurposing pyrimethamine for entirely new indications, though this would require extensive and costly research.
- Impact of Evolving Disease Epidemiology: Changes in the prevalence of toxoplasmosis and malaria, influenced by factors like climate change, global travel, and public health interventions, will indirectly shape the future demand for pyrimethamine.
Table 1: Pyrimethamine - Key Market Characteristics
| Characteristic |
Description |
| Drug Class |
Antiparasitic (Dihydrofolate Reductase Inhibitor) |
| Primary Indications |
Toxoplasmosis (treatment and prophylaxis), Malaria (historically, now in combinations) |
| Market Maturity |
Mature, primarily generic |
| Key Drivers |
Essential medicine status, prevalence of toxoplasmosis in immunocompromised, WHO essential medicine lists, cost-effectiveness. |
| Key Restraints |
Generic competition, widespread antimalarial resistance to monotherapy, development of alternative treatments. |
| Key Manufacturers |
Dar Pharmaceutical Limited, Teva Pharmaceutical Industries Ltd., Viatris Inc., Sanofi S.A. (historically), various other generic producers. |
| Regulatory Status |
Widely approved generic drug; subject to GMP, bioequivalence requirements. |
| Patent Status |
Original composition of matter patents expired; potential for secondary patents in formulation or manufacturing. |
| Pricing Strategy |
Cost-plus, competitive generic pricing, bulk procurement for public health programs. |
| Projected Growth |
Modest (2-4% annually), driven by continued essential use. |
| Future Outlook |
Continued niche but essential role, focus on affordability and accessibility, limited direct innovation. |
Key Takeaways
- Pyrimethamine is a mature generic antiparasitic with a steady, modest market growth projected at 2-4% annually, driven by its essential role in treating toxoplasmosis and, in combination, malaria.
- The market is characterized by intense competition among generic manufacturers, leading to price sensitivity and a focus on cost-effective production and distribution.
- Original patents have expired, leaving the landscape open to generic producers. Secondary patents may exist for formulations or manufacturing processes.
- Regulatory oversight focuses on GMP, bioequivalence for generics, and ensuring supply chain integrity, particularly for its use in public health programs.
- The development of newer antimalarial regimens (ACTs) and alternative therapies for toxoplasmosis poses a long-term competitive challenge, though pyrimethamine remains critical for specific patient populations and neglected diseases.
- Pricing is largely determined by generic market dynamics and bulk procurement for public health initiatives.
- Future market trajectory hinges on maintaining accessibility and affordability rather than significant product innovation.
FAQs
-
Will new patents emerge for pyrimethamine that could restrict generic availability?
While the original composition of matter patents have expired, secondary patents related to novel formulations, drug delivery systems, or manufacturing processes could theoretically be filed. However, significant innovation in this area for a drug of this age is unlikely to fundamentally alter the generic market.
-
What is the primary factor driving the demand for pyrimethamine in the current market?
The primary driver is its essential status for treating toxoplasmosis, particularly in immunocompromised individuals, and its continued, albeit reduced, role in combination antimalarial therapies, supported by its cost-effectiveness and inclusion on global essential medicine lists.
-
How is the price of pyrimethamine determined in global markets?
Prices are largely set by generic competition, manufacturing costs, regulatory compliance expenses, and the volume of procurement for public health programs. Pricing varies significantly by region and the purchasing power of the buyer.
-
Are there any major ongoing clinical trials or R&D efforts focused on pyrimethamine?
Given its generic status and long history, there are limited large-scale R&D efforts focused on developing new clinical applications or significantly improving pyrimethamine itself. The focus is more on ensuring its continued availability and affordability.
-
What impact does antimicrobial resistance have on the market for pyrimethamine?
Antimicrobial resistance, particularly to malaria parasites, has significantly reduced the use of pyrimethamine as a monotherapy for malaria. However, it can still be used in specific combination therapies where resistance patterns permit, and its role in treating toxoplasmosis remains largely unaffected by antimalarial resistance.
Citations
[1] World Health Organization. (2023). WHO Model List of Essential Medicines. Retrieved from https://www.who.int/ (Specific URL may vary with updates)
[2] World Health Organization. (2023). Malaria. Retrieved from https://www.who.int/news-room/fact-sheets/detail/malaria