Last updated: February 19, 2026
PROPECIA (finasteride 1mg) is a prescription medication approved for the treatment of male pattern baldness (androgenetic alopecia). Its market trajectory is shaped by its patent expiry, generic competition, and evolving therapeutic landscape. This analysis details PROPECIA's patent history, market performance, and future outlook.
What is the Patent Status of PROPECIA?
The primary patent protecting PROPECIA, U.S. Patent No. 4,377,584, was filed on September 15, 1980, and granted on March 21, 1984. This patent covered the compound finasteride and its use in treating conditions responsive to 5-alpha-reductase inhibition, including benign prostatic hyperplasia (BPH) and androgenetic alopecia.
A key method of use patent, U.S. Patent No. 5,118,684, was filed on February 20, 1991, and granted on June 2, 1992. This patent specifically claimed the use of finasteride in treating male pattern baldness by administering a daily dose of 1 mg. This patent was crucial for PROPECIA's specific indication.
- Original Composition of Matter Patent: U.S. Patent No. 4,377,584 (Granted March 21, 1984)
- Method of Use Patent for Androgenetic Alopecia: U.S. Patent No. 5,118,684 (Granted June 2, 1992)
These patents provided market exclusivity for Merck & Co., the developer of PROPECIA. The '684 patent, in particular, was the subject of significant litigation concerning its expiration and the subsequent entry of generic competitors.
When did PROPECIA's Core Patents Expire?
The expiration of the '684 method of use patent was a pivotal event for PROPECIA's market. While the exact expiration date can be complex due to extensions and patent challenges, the core protection for the 1 mg dose for hair loss effectively ended in the early to mid-2000s, paving the way for generic entry.
- U.S. Patent No. 5,118,684 Expiration: The patent expired on June 2, 2007. However, due to various factors, including Hatch-Waxman Act provisions and potential litigation outcomes, generic versions began appearing prior to this date. For instance,ANDA (Abbreviated New Drug Application) filings and challenges to patent validity often precede formal expiry.
The expiration of these patents triggered the influx of generic finasteride 1 mg products, significantly altering the competitive landscape and pricing structure for the treatment of male pattern baldness.
What is PROPECIA's Market Performance?
PROPECIA, as a branded pharmaceutical, experienced strong market performance during its period of patent exclusivity. Its introduction offered a novel, non-surgical treatment option for a large and motivated patient population.
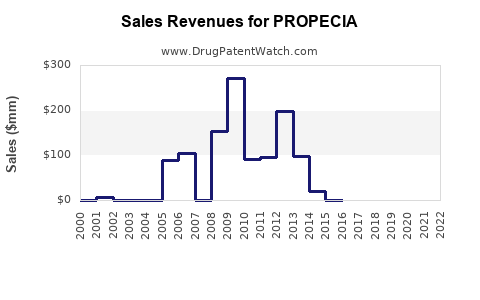
Merck & Co. reported significant sales for PROPECIA during its peak years. For example, in 2001, PROPECIA sales reached approximately $566 million globally [1]. By 2005, global sales remained robust, exceeding $470 million [1].
The market dynamics post-patent expiry are characterized by:
- Price Erosion: The introduction of generic finasteride 1 mg led to a substantial decrease in the average selling price per prescription.
- Market Share Shift: Branded PROPECIA lost significant market share to lower-cost generic alternatives.
- Volume Stability: Despite price erosion, the overall volume of finasteride 1 mg prescriptions remained relatively stable, indicating sustained demand for the treatment.
How has Generic Competition Impacted PROPECIA's Sales?
The entry of generic finasteride products directly impacted PROPECIA's revenue. Following the expiration of key patents, multiple pharmaceutical companies launched their own versions of finasteride 1 mg. This increased competition led to:
- Declining Branded Sales: Merck's sales of PROPECIA have seen a consistent decline since the peak years. By 2010, PROPECIA sales had fallen to approximately $150 million [1]. The decline continued in subsequent years, with global sales in 2019 reported at around $53 million [1].
- Increased Accessibility: Generic availability has made finasteride 1 mg treatment more affordable and accessible to a wider patient base.
- Market Fragmentation: The market for finasteride 1 mg is now fragmented among numerous generic manufacturers, leading to intense price competition among them.
Table 1: PROPECIA Global Sales Trend (USD Millions)
| Year |
Sales |
| 2001 |
566 |
| 2005 |
470 |
| 2010 |
150 |
| 2019 |
53 |
Source: Merck & Co. Annual Reports [1]
What is the Therapeutic Landscape for Hair Loss Treatments?
PROPECIA's therapeutic landscape has evolved since its initial approval in 1997. While finasteride 1 mg remains a cornerstone treatment for male pattern baldness, new research and alternative therapies have emerged.
What are the Primary Alternative Treatments for Androgenetic Alopecia?
Current therapeutic options for androgenetic alopecia include:
- Topical Minoxidil: Available over-the-counter, minoxidil is another widely used treatment that stimulates hair growth. It is often used in combination with finasteride.
- Low-Level Laser Therapy (LLLT): Devices such as laser combs and helmets are used to stimulate hair follicles.
- Platelet-Rich Plasma (PRP) Therapy: This involves injecting a patient's own concentrated platelets into the scalp to promote hair growth.
- Hair Transplantation: Surgical procedures to move hair follicles from denser areas of the scalp to balding areas.
- Emerging Therapies: Research continues into novel drug targets, including stem cell therapies, androgens pathway modulators, and compounds that affect the hair growth cycle.
How does Finasteride 1 mg Compare to Other Treatments?
Finasteride 1 mg's efficacy in treating male pattern baldness is well-established. It works by inhibiting the enzyme 5-alpha-reductase type II, which converts testosterone to dihydrotestosterone (DHT). DHT is the primary hormone responsible for shrinking hair follicles in genetically predisposed individuals.
- Efficacy: Studies demonstrate that finasteride 1 mg can halt hair loss and promote regrowth in a significant percentage of men [2].
- Mechanism: Unlike minoxidil, which primarily acts as a vasodilator, finasteride addresses the hormonal cause of hair loss.
- Safety Profile: PROPECIA has a generally favorable safety profile, although potential side effects, including sexual dysfunction, have been reported and are a subject of ongoing discussion and research.
- Cost: Branded PROPECIA was significantly more expensive than generic finasteride. Generic finasteride is cost-competitive with topical minoxidil.
The market presence of minoxidil, particularly its over-the-counter availability, provides a significant competitive alternative that does not require a prescription, broadening patient access without direct pharmaceutical competition.
What is the Future Outlook for Finasteride 1mg?
The future market for finasteride 1 mg is characterized by the continued dominance of generic products and the ongoing demand for effective hair loss treatments.
What are the Key Drivers for the Finasteride 1mg Market?
- Prevalence of Androgenetic Alopecia: Male pattern baldness is a widespread condition, affecting a large proportion of the male population worldwide. This sustained prevalence drives consistent demand.
- Established Efficacy and Safety: Finasteride 1mg has a long track record of efficacy and a generally understood safety profile, making it a trusted treatment option for both patients and healthcare providers.
- Cost-Effectiveness of Generics: The availability of affordable generic finasteride 1 mg ensures continued accessibility for a broad patient demographic.
- Combination Therapies: Finasteride is often used in combination with minoxidil, further solidifying its role in treatment regimens.
What are the Potential Challenges?
- Side Effect Concerns: Persistent concerns and ongoing research regarding potential sexual side effects, even after drug discontinuation (post-finasteride syndrome), may influence prescribing patterns and patient acceptance.
- Competition from Novel Therapies: As research progresses, new treatments with different mechanisms of action or improved safety profiles could emerge, potentially challenging finasteride's market position.
- Regulatory Scrutiny: As with any medication, finasteride is subject to ongoing regulatory review and potential label updates based on new safety data.
- Patient Education and Compliance: Effective management requires patient understanding of the treatment's mechanism, expected outcomes, and potential risks.
The market for finasteride 1 mg is mature and largely driven by generic competition. While branded PROPECIA sales are negligible, the active pharmaceutical ingredient (API) finasteride continues to be manufactured and marketed by numerous generic companies globally. The market's trajectory will be influenced by the balance between sustained demand, the competitive pricing of generics, and the evolving landscape of hair loss treatments.
Key Takeaways
- PROPECIA's patent protection, primarily U.S. Patent No. 5,118,684 for the 1 mg method of use, expired on June 2, 2007, allowing for generic market entry.
- Branded PROPECIA sales peaked in the early to mid-2000s, exceeding $500 million annually, but declined sharply post-patent expiry to approximately $53 million by 2019.
- Generic finasteride 1 mg has captured the majority of the market share due to price competition, making the treatment more accessible.
- Finasteride 1 mg remains a primary treatment for male pattern baldness, competing with topical minoxidil, LLLT, PRP, and hair transplantation.
- The future market for finasteride 1 mg is expected to be dominated by generics, driven by the high prevalence of androgenetic alopecia and the established efficacy of the drug, balanced against potential side effect concerns and emerging novel therapies.
Frequently Asked Questions
-
What is the difference between PROPECIA and Proscar?
PROPECIA (finasteride 1mg) is prescribed for male pattern baldness. Proscar (finasteride 5mg) is prescribed for benign prostatic hyperplasia (BPH). Both contain the same active ingredient, finasteride, but at different dosages for distinct medical conditions.
-
Can women use finasteride for hair loss?
Finasteride 1mg is not indicated for use in women and is contraindicated in women who are or may become pregnant due to the risk of birth defects in a male fetus.
-
Are there any long-term side effects associated with finasteride 1mg?
Potential side effects include decreased libido, erectile dysfunction, and ejaculatory disorders. While these are generally reversible upon discontinuation, there are reports and ongoing research into potential persistent side effects, sometimes referred to as post-finasteride syndrome.
-
How long does it take to see results from finasteride 1mg?
It typically takes at least three to six months of consistent daily use to observe noticeable results, such as a reduction in hair loss or evidence of regrowth. Continued use is generally required to maintain benefits.
-
Is finasteride 1mg available over-the-counter?
No, finasteride 1mg is a prescription-only medication and is not available over-the-counter.
Citations
[1] Merck & Co. (Various Years). Annual Reports. U.S. Securities and Exchange Commission Filings.
[2] Kaufman, K. D., Ellis, P. D., & Roenigk, R. P. (1998). Finasteride in the treatment of men with androgenetic alopecia. Journal of the American Academy of Dermatology, 39(3), S52-S58. https://doi.org/10.1016/s0190-9622(98)70007-0