PROMACTA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Promacta, and when can generic versions of Promacta launch?
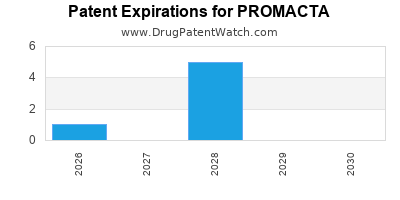
Promacta is a drug marketed by Novartis and is included in two NDAs. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and eight patent family members in forty countries.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the eltrombopag olamine profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Promacta
A generic version of PROMACTA was approved as eltrombopag olamine by ANNORA PHARMA on April 18th, 2024.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for PROMACTA?
- What are the global sales for PROMACTA?
- What is Average Wholesale Price for PROMACTA?
Summary for PROMACTA
| International Patents: | 108 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 72 |
| Clinical Trials: | 31 |
| Patent Applications: | 249 |
| Drug Prices: | Drug price information for PROMACTA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PROMACTA |
| What excipients (inactive ingredients) are in PROMACTA? | PROMACTA excipients list |
| DailyMed Link: | PROMACTA at DailyMed |

Recent Clinical Trials for PROMACTA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Food and Drug Administration (FDA) | Phase 1 |
| University of California, San Francisco | Phase 1 |
| Anjali Pawar | Phase 1 |
Pharmacology for PROMACTA
Paragraph IV (Patent) Challenges for PROMACTA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PROMACTA | Tablets | eltrombopag olamine | 12.5 mg and 25 mg | 022291 | 1 | 2014-02-04 |
| PROMACTA | Tablets | eltrombopag olamine | 50 mg and 75 mg | 022291 | 1 | 2014-01-07 |
US Patents and Regulatory Information for PROMACTA
PROMACTA is protected by six US patents and one FDA Regulatory Exclusivity.
Patents protecting PROMACTA
3'-[(2z)-[1-(3,4-Dimethylphenyl)-1,5-dihydro-3-methyl-5-oxo-4h-pyrazol-4-y- lidene]hy-drazino]-2'-hydroxy-[1,1'-piphenyl]-acid bis-(monoethanolamine)
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
3'-[(2Z)-[1-(3,4-dimethylphenyl)-1,5-dihydro-3-methyl-5-oxo-4H-pyrazol-4-y- lidene]hydrazino]-2'-hydroxy[1,1'-biphenyl]-3-carboxylic acid bis-(monoethanolamine)
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
3'-[(2Z)-[1-(3,4-dimethylphenyl)-1,5-dihydro-3-methyl-5-oxo-4H-pyrazol-4-y- lidene] hydrazino]-2'-hydroxy-[1,1'-biphenyl]-3-carboxylic acid bis-(monoethanolamine)
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
3'-[(2Z)-[1-(3,4-dimethylphenyl)-1 ,5-dihydro-3-methyl-5-oxo-4H-pyrazol-4-ylidene]hydrazino]-2'-hydroxy-[1,1- '-biphenyl]-3-carboxylic acid bis-(monoethanolamine)
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
3'-[(2Z)-[1-(3,4-dimethylphenyl)-1,5-dihydro-3-methyl-5-oxo-4H-pyrazol-4-y- lidene]hydrazino]-2'-hydroxy-[1,1'-biphenyl]-3-carboxylic acid bis-(monoethanolamine)
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
3'-[(2Z)-[1-(3,4-dimethylphenyl)-1,5-dihydro-3-methyl-5-oxo-4H-pyrazol-4-y- lidene]hydrazino]-2'-hydroxy-[1,1'-biphenyl]-3-carboxylic acid bis-(monoethanolamine)
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
FDA Regulatory Exclusivity protecting PROMACTA
INDICATED IN COMBINATION WITH STANDARD IMMUNOSUPPRESSIVE THERAPY FOR THE FIRST-LINE TREATMENT OF ADULT AND PEDIATRIC PATIENTS 2 YEARS AND OLDER WITH SEVERE APLASTIC ANEMIA
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | PROMACTA KIT | eltrombopag olamine | FOR SUSPENSION;ORAL | 207027-002 | Sep 27, 2018 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-003 | Sep 8, 2009 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Novartis | PROMACTA KIT | eltrombopag olamine | FOR SUSPENSION;ORAL | 207027-001 | Aug 24, 2015 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-005 | Nov 16, 2012 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-004 | Oct 20, 2011 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-002 | Nov 20, 2008 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for PROMACTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-001 | Nov 20, 2008 | ⤷ Sign Up | ⤷ Sign Up |
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-001 | Nov 20, 2008 | ⤷ Sign Up | ⤷ Sign Up |
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-005 | Nov 16, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-001 | Nov 20, 2008 | ⤷ Sign Up | ⤷ Sign Up |
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-003 | Sep 8, 2009 | ⤷ Sign Up | ⤷ Sign Up |
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-002 | Nov 20, 2008 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for PROMACTA
When does loss-of-exclusivity occur for PROMACTA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 9656
Patent: COMPRIMIDO FARMACEUTICO QUE COMPRENDE LA SAL DE BIS - (MONOETANOLAMINA) DEL ACIDO 3' -[(2Z )-[1-(3,4- DIMETILFENIL)-1,5- DIHIDRO-3- METIL-5 OXO- 4H-PIRAZOL-4- ILIDENO} HIDRAZINO} -2' HIDROXI- { 1,1'- BIFENIL]-3- CARBOXILICO [ELTROMBOPAG OLAMINA), CAPSULA FARMACEUTICA, FORMA DE DOSIFICACION Y GRANULO
Estimated Expiration: ⤷ Sign Up
Patent: 7711
Patent: COMPRIMIDO FARMACÉUTICO, GRÁNULOS FARMACÉUTICOS Y PROCEDIMIENTO PARA SU PREPARACIÓN, FORMA FARMACÉUTICA ORAL SÓLIDA, CÁPSULA
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 07352608
Patent: Novel pharmaceutical composition
Estimated Expiration: ⤷ Sign Up
Brazil
Patent: 0721651
Patent: COMPOSIÇÃO FARMACÊUTICA
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 85831
Patent: NOUVELLE COMPOSITION PHARMACEUTIQUE (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Chile
Patent: 07002242
Patent: FORMA DE DOSIFICACION FARMACEUTICA ORAL EN COMPRIMIDO, CAPSULA O GRANULO QUE COMPRENDE BIS-(MONOETANOLAMINA) DE ACIDO 3'-[(2Z)-[1-(3,4-DIMETILFENIL)-1,5-DIHIDRO-3-METIL-5-OXO-4H-PIRAZOL-4-ILIDENO]HIDRAZINO]-2'-HIDROXI[1-1'-BIFENIL]-3-CARBOXILICO; PRO
Estimated Expiration: ⤷ Sign Up
China
Patent: 1686930
Patent: Novel pharmaceutical composition
Estimated Expiration: ⤷ Sign Up
Patent: 2688207
Patent: Novel pharmaceutical composition
Estimated Expiration: ⤷ Sign Up
Patent: 2697745
Patent: Novel pharmaceutical composition
Estimated Expiration: ⤷ Sign Up
Colombia
Patent: 60058
Patent: COMPOSICIONES QUE CONTIENEN BIS-(MONOETANOLAMINA) DEL ACIDO 3 ́-[(2Z)-[1-(3,4-DIMETILFENIL)-1,5-DIHIDRO-3-METIL-5-OXO-4H-PIRAZOL-4-ILIDENO]HIDRAZINO]-2 ́HIDROXI-[1,1 ́-BIFENIL]-3-CARBOXILICO (ELTROMBOPAG OLAMINA) Y PROCESOS PARA SU PREPARACION .
Estimated Expiration: ⤷ Sign Up
Costa Rica
Patent: 143
Patent: NUEVA COMPOSICION FARMACEUTICA
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0160206
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 17284
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 52237
Estimated Expiration: ⤷ Sign Up
Dominican Republic
Patent: 009000253
Patent: COMPOSICION FARMACEUTICA NOVEDOSA
Estimated Expiration: ⤷ Sign Up
Ecuador
Patent: 077628
Patent: NUEVA COMPOSICIÓN FARMACÉUTICA
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 0883
Patent: ФАРМАЦЕВТИЧЕСКАЯ ТАБЛЕТКА (PHARMACEUTICAL TABLET)
Estimated Expiration: ⤷ Sign Up
Patent: 4294
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ ОЛАМИНА ЭЛЬТРОМБОПАГА (PHARMACEUTICAL COMPOSITION OF ELTROMBOPAG OLAMINE)
Estimated Expiration: ⤷ Sign Up
Patent: 0971018
Patent: НОВАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ
Estimated Expiration: ⤷ Sign Up
Patent: 1400387
Patent: НОВАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ
Estimated Expiration: ⤷ Sign Up
Patent: 1991590
Patent: НОВАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 52237
Patent: NOUVELLE COMPOSITION PHARMACEUTIQUE (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Patent: 90730
Patent: NOUVELLE COMPOSITION PHARMACEUTIQUE (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Patent: 18732
Patent: COMPRIMÉS COMPRENANTS DE L'ELTROMBOPAG OLAMINE (TABLETS COMPRISING ELTROMBOPAG OLAMINE)
Estimated Expiration: ⤷ Sign Up
Patent: 18733
Patent: COMPRIMÉS COMPRENANTS DE L'ELTROMBOPAG OLAMINE (TABLETS COMPRISING ELTROMBOPAG OLAMINE)
Estimated Expiration: ⤷ Sign Up
Hong Kong
Patent: 36968
Patent: 新穎的藥物組合物 (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Hungary
Patent: 27209
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 1891
Patent: טבליות רוקחיות המכילות אלטרומבופאג אולמין, שיטות להכנתן ושימושן בהכנת תרופות לטיפול בתרומבוציטופניה (Pharmaceutical tablets containing eltrombopag olamine, methods for their preparation and use thereof in the preparation of medicaments for treating thrombocytopenia)
Estimated Expiration: ⤷ Sign Up
Patent: 8840
Patent: טבליות רוקחיות המכילות אלטרומבופאג אולמין, שיטות להכנתן ושימושן בהכנת תרופות לטיפול בתרומבוציטופניה (Pharmaceutical tablets containing eltrombopag olamine, methods for their preparation and use thereof in the preparation of medicaments for treating thrombocytopenia)
Estimated Expiration: ⤷ Sign Up
Patent: 4602
Patent: טבליות רוקחיות המכילות אלטרומבופאג אולמין, שיטות להכנתן ושימושן בהכנת תרופות לטיפול בתרומבוציטופניה (Pharmaceutical tablets containing eltrombopag olamine, methods for their preparation and use thereof in the preparation of medicaments for treating thrombocytopenia)
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 19866
Estimated Expiration: ⤷ Sign Up
Patent: 35078
Estimated Expiration: ⤷ Sign Up
Patent: 44713
Estimated Expiration: ⤷ Sign Up
Patent: 60289
Estimated Expiration: ⤷ Sign Up
Patent: 10526140
Estimated Expiration: ⤷ Sign Up
Patent: 14005302
Patent: NOVEL PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Sign Up
Patent: 15129195
Patent: 新たな医薬組成物 (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Patent: 17137343
Patent: 新たな医薬組成物 (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Patent: 19123747
Patent: 新たな医薬組成物 (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Patent: 21100968
Patent: 新たな医薬組成物 (NOVEL PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Sign Up
Patent: 23011888
Patent: 新たな医薬組成物
Estimated Expiration: ⤷ Sign Up
Jordan
Patent: 43
Patent: مركبات صيدلانية جديدة (Novel pharmaceutical composition)
Estimated Expiration: ⤷ Sign Up
Malaysia
Patent: 8072
Patent: NOVEL PHAMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 09011881
Patent: NUEVA COMPOSICION FARMACEUTICA. (NOVEL PHARMACEUTICAL COMPOSITION.)
Estimated Expiration: ⤷ Sign Up
Morocco
Patent: 236
Patent: NOUVELLE COMPOSITION PHARMACEUTIQUE
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 0888
Patent: Compositions of eltrombopag olamine substantially free from coordinating metals or reducing sugars
Estimated Expiration: ⤷ Sign Up
Peru
Patent: 080773
Patent: COMPOSICION FARMACEUTICA QUE COMPRENDE BIS-(MONOETANOLAMINA) DE ACIDO 3'-[(2Z)-[1-(3,4-DIMETILFENIL)-1,5-DIHIDRO-3-METIL-5-OXO-4H-PIRAZOL-4-ILIDENO]HIDRAZINO]-2'-HIDROXI-[1,1'-BIFENIL]-3-CARBOXILICO
Estimated Expiration: ⤷ Sign Up
Patent: 121407
Patent: COMPOSICION FARMACEUTICA QUE COMPRENDE BIS-(MONOETANOLAMINA) DE ACIDO 3'-[(2Z)-[1-(3, 4-DIMETILFENIL)-1, 5-DIHIDRO-3-METIL-5-OXO-4H-PIRAZOL-4-ILIDENO] HIDRAZINO]-2'-HIDROXI-[1, 1'-BIFENIL]-3-CARBOXILICO
Estimated Expiration: ⤷ Sign Up
Patent: 151953
Patent: COMPOSICION FARMACEUTICA QUE COMPRENDE BIS-(MONOETANOLAMINA) DE ACIDO 3'-[(2Z)-[1-(3, 4-DIMETILFENIL)-1, 5-DIHIDRO-3-METIL-5-OXO-4H-PIRAZOL-4-ILIDENO] HIDRAZINO]-2'-HIDROXI-[1, 1'-BIFENIL]-3-CARBOXILICO
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 52237
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 52237
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 52237
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 0907710
Patent: NOVEL PHARMACEUICAL COMPOSITION
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 1475971
Estimated Expiration: ⤷ Sign Up
Patent: 1537200
Estimated Expiration: ⤷ Sign Up
Patent: 1632851
Estimated Expiration: ⤷ Sign Up
Patent: 100020456
Patent: NOVEL PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Sign Up
Patent: 140049086
Patent: NOVEL PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Sign Up
Patent: 150008513
Patent: NOVEL PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 65179
Estimated Expiration: ⤷ Sign Up
Taiwan
Patent: 39267
Estimated Expiration: ⤷ Sign Up
Patent: 38674
Estimated Expiration: ⤷ Sign Up
Patent: 0843742
Patent: Novel pharmaceutical composition
Estimated Expiration: ⤷ Sign Up
Patent: 1410240
Patent: Pharmaceutical tablets, solid oral pharmaceutical dosage forms, pharmaceutical capsules and pharmaceutical granules comprising 3'-[(2Z)-[1-(3,4-dimethylphenyl)-1,5-dihydro-3-methyl-5-oxo-4H-pyrazol-4-ylidene]hydrazino]-2'-hydroxy-[1,1'-biphenyl]-3-carbox
Estimated Expiration: ⤷ Sign Up
Ukraine
Patent: 261
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ ЭЛТРОМБОПАГА ОЛАМИНА (ВАРИАНТЫ) И СПОСОБ ЕЕ ИЗГОТОВЛЕНИЯ (ВАРИАНТЫ);ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ ЕЛТРОМБОПАГУ ОЛАМІНУ (ВАРІАНТИ) І СПОСІБ ЇЇ ВИГОТОВЛЕННЯ (ВАРІАНТИ) (PHARMACEUTICAL COMPOSITIONS CONTAINING ELTROMBOPAG OLAMINE AND PROCESSES FOR THE PREPARATION THEREOF)
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering PROMACTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Mexico | PA02011621 | MIMETICOS DE TROMBOPOYETINA. (THROMBOPOIETIN MIMETICS.) | ⤷ Sign Up |
| Hong Kong | 1055561 | ⤷ Sign Up | |
| Brazil | PI0721651 | COMPOSIÇÃO FARMACÊUTICA | ⤷ Sign Up |
| Israel | 199438 | USE OF 3' -[(2Z) -[1-(3,4-DIMETHYLPHENYL)-1,5-DIHYDRO-3-METHYL-5-OXO-4H- PYRAZOL-4-YLIDENE] HYDRAZINE]-2'-HYDROXY-[1,1'-BIPHENYL]-3- CARBOXYLIC ACID BIS-(MONOETHANOLAMINE) IN THE PREPARATION OF MEDICAMENTS FOR TREATING THROMBOCYTOPENIA AND PHARMACEUTICAL COMPOSITIONS COMPRISING THE SAME | ⤷ Sign Up |
| South Korea | 100798568 | ⤷ Sign Up | |
| Spain | 2334843 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PROMACTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1534390 | C 2010 013 | Romania | ⤷ Sign Up | PRODUCT NAME: ELTROMBOPAG; NATIONAL AUTHORISATION NUMBER: RO EU/1/10/612/001, RO EU/1/10/612/002, RO EU/1/10/612/003, RO EU/1/10/612/004, RO EU/1/10/612/005, RO EU/1/10/612/006; DATE OF NATIONAL AUTHORISATION: 20100311; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EMEA EU/1/10/612/001, EMEA EU/1/10/612/002, EMEA EU/1/10/612/003, EMEA EU/1/10/612/004, EMEA EU/1/10/612/005, EMEA EU/1/10/612/006; DATE OF FIRST AUTHORISATION IN EEA: 20100311 |
| 1294378 | SPC020/2010 | Ireland | ⤷ Sign Up | SPC020/2010: 20110308, EXPIRES: 20250310 |
| 1294378 | 122010000037 | Germany | ⤷ Sign Up | PRODUCT NAME: ELTROMBOPAG SOWIE PHARMAZEUTISCH ANNEHMBARE SALZE, HYDRATE UND SOLVATE DAVON; NAT. REGISTRATION NO/DATE: EU/1/10/612/001-006 20100311; FIRST REGISTRATION: EU EU/1/10/612/001-/006 20100311 |
| 1534390 | PA2010007,C1534390 | Lithuania | ⤷ Sign Up | PRODUCT NAME: ELTROMBOPAGO OLAMINAS; REGISTRATION NO/DATE: EU/1/10/612/001, 2010-03-11 EU/1/10/612/002, 2010-03-11 EU/1/10/612/003, 2010-03-11 EU/1/10/612/004, 2010-03-11 EU/1/10/612/005, 2010-03-11 EU/1/10/612/006 20100311 |
| 1534390 | C20100006 00032 | Estonia | ⤷ Sign Up | PRODUCT NAME: REVOLADE-ELTROMBOPAG; REG NO/DATE: C(2010)1662 11.03.2010 |
| 1534390 | PA2010007 | Lithuania | ⤷ Sign Up | PRODUCT NAME: ELTROMBOPAGUM OLAMINUM; REGISTRATION NO/DATE: EU/1/10/612/001, 2010 03 11 EU/1/10/612/002, 2010 03 11 EU/1/10/612/003, 2010 03 11 EU/1/10/612/004, 2010 03 11 EU/1/10/612/005, 2010 03 11 EU/1/10/612/006 20100311 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


