Last updated: February 19, 2026
PREMARIN, a conjugated equine estrogen (CEE) therapy, has maintained a significant market presence since its introduction by Wyeth (now part of Pfizer) in 1942. Its primary indication is the treatment of moderate to severe vasomotor symptoms due to menopause and vulvar and vaginal atrophy (VVA). The drug's market trajectory is shaped by patent expirations, evolving therapeutic landscapes, and a persistent demand for established hormone replacement therapies.
What is PREMARIN's Current Market Share and Revenue?
PREMARIN's market share is subject to ongoing competition from newer hormone therapies and non-hormonal alternatives. While precise, real-time market share figures are proprietary, publicly available financial reports and industry analyses provide insights into its revenue contribution.
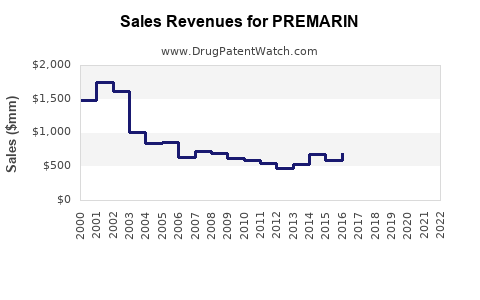
- 2023 Revenue: Pfizer reported global net revenue of $707 million for its Women's Health portfolio in 2023. This portfolio includes PREMARIN, along with other products like LOEPRIN and VIAGRA. The exact revenue attributable to PREMARIN is not separately itemized, but it remains a substantial component of this segment. In 2022, the Women's Health segment generated $717 million in revenue. [1]
- Historical Performance: PREMARIN's revenue has seen fluctuations. For example, in the first quarter of 2022, PREMARIN contributed $110 million in global sales, demonstrating its continued commercial viability. [2]
- Competitive Landscape: The market for menopausal symptom relief is crowded. Competitors include other estrogen-based therapies (both oral and transdermal), progestins, and a growing class of non-hormonal treatments such as SSRIs/SNRIs (e.g., paroxetine, escitalopram) and newer selective estrogen receptor modulators (SERMs) like fezolinetant (VEZNEO). [3]
What is the Patent Status of PREMARIN?
PREMARIN's original composition of matter patents have long expired. However, the drug's longevity is partly due to subsequent patents covering specific formulations, methods of use, and manufacturing processes.
- Original Patents: The foundational patents for PREMARIN expired decades ago, allowing for generic competition for the basic CEE formulation.
- Evergreening Efforts: Wyeth, and subsequently Pfizer, have engaged in strategies to protect market exclusivity beyond original patent expiry. This can include:
- Method of Use Patents: Patents claiming the use of PREMARIN for specific medical conditions or patient populations.
- Formulation Patents: Patents protecting novel delivery systems or specific combinations of active ingredients within a dosage form. For instance, patents related to vaginal creams or tablets.
- Process Patents: Patents covering improved or novel manufacturing methods.
- Generic Competition: Despite these efforts, generic versions of oral CEE are available, exerting downward pressure on pricing and market share for the branded product. The market penetration of generics varies by region and specific formulation.
- Orphan Drug Exclusivity: While not directly applicable to PREMARIN's primary indications for menopausal symptoms, some niche uses or specific formulations could potentially qualify for market exclusivity periods if they met orphan drug criteria, though this is unlikely for its established uses.
What are the Key Growth Drivers and Restraints for PREMARIN?
PREMARIN's market is influenced by several factors that both support and hinder its growth.
Growth Drivers
- Aging Global Population: The increasing number of postmenopausal women worldwide represents a persistent and expanding target demographic. As women live longer, the prevalence of menopausal symptoms requiring treatment is expected to rise.
- Established Efficacy and Familiarity: PREMARIN has been used for decades, resulting in a well-established understanding of its efficacy and side effect profile among healthcare providers and patients. This familiarity can lead to continued prescribing, particularly for women who have used it successfully in the past or prefer traditional therapies.
- Cost-Effectiveness (for some formulations): Compared to some newer, branded therapies, established generics of PREMARIN can offer a more cost-effective treatment option, especially in healthcare systems with budget constraints.
- Specific Indications: While the overall trend leans towards non-hormonal options for mild symptoms, PREMARIN remains a primary choice for managing severe vasomotor symptoms and for certain cases of vulvar and vaginal atrophy (VVA) where localized estrogen therapy is indicated.
Restraints
- Safety Concerns and Regulatory Scrutiny: The Women's Health Initiative (WHI) study, published in 2002, significantly impacted the use of menopausal hormone therapy (MHT), including PREMARIN. The study linked combined estrogen-progestin therapy to increased risks of breast cancer, stroke, and cardiovascular disease. While subsequent analyses have refined the understanding of risks and benefits based on age, duration of use, and individual patient factors, the initial safety concerns created a lasting perception of risk and led to a significant decline in MHT prescribing. [4]
- Emergence of Non-Hormonal Therapies: The development and FDA approval of non-hormonal treatments for hot flashes and other menopausal symptoms, such as neurokinin-3 receptor antagonists (e.g., fezolinetant), provide viable alternatives for women who are unwilling or unable to use MHT due to contraindications or personal preference. [5]
- Preference for Transdermal and Localized Delivery: Many healthcare providers and patients now favor transdermal estrogen patches or gels, and localized vaginal estrogen therapies, over oral CEE. These methods are perceived to have a lower systemic absorption and potentially a more favorable risk profile, especially for women with a history of thrombotic events or other contraindications to oral estrogen.
- Generic Competition: The availability of multiple generic versions of oral CEE fragments the market and drives down prices, reducing the revenue potential for the branded product.
- Stigma Associated with Hormone Therapy: Despite ongoing research and evolving clinical guidelines, a lingering stigma surrounds hormone replacement therapy, leading some women to avoid it altogether.
What is the Competitive Landscape for PREMARIN?
The competitive landscape for PREMARIN is dynamic, characterized by both direct and indirect competition.
Direct Competitors (Other CEE or Similar Estrogen Therapies)
- Generic Oral CEE: Multiple manufacturers produce generic versions of oral conjugated equine estrogens. These are often available at significantly lower price points than branded PREMARIN, directly impacting branded sales.
- Other Oral Estrogens: While not CEE, other oral estrogen formulations (e.g., synthetic estrogens like ethinyl estradiol, though less commonly used for menopause now) and micronized estradiol products compete.
- Transdermal Estrogen: Estradiol patches (e.g., Estraderm, Vivelle-Dot), gels (e.g., Divigel, EstroGel), and sprays offer systemic estrogen delivery with potentially different risk profiles than oral CEE.
- Vaginal Estrogen: For VVA, localized treatments such as vaginal creams (e.g., Estrace vaginal cream), tablets (e.g., Vagifem), and inserts offer targeted relief with minimal systemic absorption. PREMARIN also has a vaginal cream formulation, but it competes with other established products in this space.
Indirect Competitors (Non-Hormonal Therapies)
- SSRIs/SNRIs: Paroxetine (Paxil CR), escitalopram (Lunesta, though approved for insomnia, some use off-label for hot flashes), and venlafaxine are FDA-approved for treating moderate to severe hot flashes and night sweats.
- Neurokinin-3 (NK3) Receptor Antagonists: Fezolinetant (VEZNEO) is a novel, non-hormonal oral medication specifically targeting the thermoregulatory pathway in the brain to reduce hot flashes. Its approval marks a significant development in the non-hormonal MHT market. [5]
- Other Non-Hormonal Agents: Gabapentin, clonidine, and certain dietary supplements are also used, though often with less robust evidence of efficacy for severe symptoms compared to prescription medications.
Table 1: Comparative Overview of Menopausal Symptom Treatments
| Treatment Class |
Mechanism of Action |
Key Advantages |
Key Disadvantages |
Example Products |
| Conjugated Equine Estrogens (CEE) |
Replaces declining estrogen levels, targeting thermoregulatory centers in the hypothalamus. |
Long history of use, established efficacy for severe symptoms, cost-effective. |
Potential systemic risks (CV, VTE, Breast Ca. if combined with progestin), oral route can have higher hepatic effects. |
PREMARIN (oral, vaginal cream), Generic Oral CEE. |
| Other Oral Estrogens |
Systemic estrogen replacement. |
Alternative formulations for individuals intolerant to CEE. |
Similar systemic risk profile to oral CEE, liver first-pass metabolism. |
Estradiol (e.g., Estrace oral tablets), Ethinyl estradiol (rare). |
| Transdermal Estrogen |
Delivers estrogen through the skin, bypassing first-pass liver metabolism. |
Reduced risk of VTE and stroke compared to oral routes, potentially better for certain risk profiles. |
Skin irritation, can be more expensive than oral generics. |
Estradiol patches (e.g., Estraderm, Vivelle-Dot), gels, sprays. |
| Vaginal Estrogen |
Localized estrogen delivery to vaginal tissues. |
Minimal systemic absorption, effective for VVA symptoms, generally low risk. |
Primarily for local symptoms, not systemic vasomotor symptoms. |
Estradiol vaginal tablets/rings, Estrace vaginal cream. |
| SSRIs/SNRIs |
Modulate neurotransmitters involved in thermoregulation (serotonin, norepinephrine). |
Non-hormonal, no estrogen-related risks, also treats comorbid depression/anxiety. |
Can have side effects (nausea, dizziness, sexual dysfunction), variable efficacy. |
Paroxetine (Paxil CR), Venlafaxine, Escitalopram. |
| NK3 Receptor Antagonists |
Blocks NK3 receptor, interrupting the kisspeptin/neurokinin B pathway involved in hot flashes. |
Non-hormonal, directly targets the pathway for hot flashes, good efficacy. |
New class, potential for unknown long-term side effects, requires monitoring for liver function. |
Fezolinetant (VEZNEO). |
What is the Financial Trajectory and Outlook for PREMARIN?
PREMARIN's financial trajectory is characterized by a mature product lifecycle, marked by a gradual decline in volume offset by pricing power and market inertia.
- Mature Product: As a drug with a long history, PREMARIN operates in a mature market. Significant growth is unlikely. The primary focus for manufacturers of such products is often on defending market share and optimizing profitability through cost management and strategic pricing.
- Sustained Demand: Despite the advent of newer therapies, a segment of the patient population and healthcare providers continues to rely on PREMARIN due to its established track record and accessibility. This creates a baseline level of demand that supports ongoing revenue.
- Impact of Generic Erosion: The presence of generic competitors will continue to exert downward pressure on pricing and revenue growth. Pfizer's strategy likely involves managing the branded product's margin while leveraging its broader Women's Health portfolio.
- Potential for Decline: The increasing availability and preference for non-hormonal alternatives and newer hormonal therapies with perceived better safety profiles suggest a long-term trend of gradual decline in PREMARIN's market share and revenue. However, the pace of this decline will depend on the success of new entrants and evolving clinical guidelines.
- Geographic Variations: Revenue performance can vary significantly by region, influenced by local regulatory environments, healthcare reimbursement policies, and physician prescribing habits.
- Pfizer's Portfolio Management: PREMARIN's financial contribution is viewed within the context of Pfizer's larger Women's Health segment. While it may not be a growth engine, its consistent revenue generation is valuable for the company's diversified portfolio.
Key Takeaways
- PREMARIN's revenue is a significant component of Pfizer's Women's Health portfolio, with global sales in the hundreds of millions annually.
- Original patents have expired, leading to generic competition, but patent strategies related to formulations and methods of use have contributed to extended market protection.
- Growth drivers include an aging global population and PREMARIN's established efficacy, while restraints are primarily driven by safety concerns from past studies, the rise of non-hormonal alternatives, and preference for alternative delivery methods.
- The competitive landscape includes generic CEE, other oral and transdermal estrogens, and a growing array of non-hormonal therapies like SSRIs/SNRIs and novel NK3 receptor antagonists.
- PREMARIN's financial trajectory is that of a mature product, with its outlook characterized by sustained but likely declining revenue due to generic erosion and evolving treatment paradigms, offset by its persistent demand in certain patient segments.
FAQs
-
Will PREMARIN be discontinued due to newer therapies?
While newer therapies are gaining traction, PREMARIN is unlikely to be discontinued in the near term. Its long history of use, established efficacy for severe symptoms, and the existence of generic versions ensure a sustained, albeit potentially diminishing, market presence. Pfizer manages a portfolio of mature products, and PREMARIN continues to contribute revenue.
-
What are the primary risks associated with PREMARIN compared to newer menopausal hormone therapies?
The primary risks associated with oral CEE, including PREMARIN, are linked to historical findings from the Women's Health Initiative study, which indicated increased risks of cardiovascular events, stroke, and breast cancer when combined with a progestin. Newer therapies, particularly non-hormonal options and transdermal estrogen preparations, are often perceived to have a more favorable risk-benefit profile for certain patient populations by minimizing systemic exposure or avoiding hormonal mechanisms entirely.
-
How significant is the impact of generic PREMARIN on the branded product's market share and revenue?
The impact of generic PREMARIN is substantial. Generic versions, available at significantly lower price points, have eroded branded market share and revenue for decades. Pfizer's strategy likely involves focusing on specific patient segments or geographical markets where the branded product maintains a competitive advantage, alongside managing its overall Women's Health portfolio.
-
Is PREMARIN still considered a first-line treatment for menopausal symptoms?
PREMARIN, particularly oral CEE, is generally not considered the first-line treatment for mild to moderate menopausal symptoms in current clinical practice. Current guidelines often recommend lifestyle modifications and non-hormonal pharmacotherapies (SSRIs/SNRIs, NK3 antagonists) as first-line options, especially for women with contraindications to MHT or those seeking to avoid hormonal therapy. PREMARIN may still be considered for severe vasomotor symptoms or specific cases of vulvar and vaginal atrophy, particularly when other options are unsuitable or have failed.
-
What is the role of PREMARIN in treating vulvar and vaginal atrophy (VVA) versus systemic menopausal symptoms?
PREMARIN is available in both oral and vaginal cream formulations. The oral form addresses systemic symptoms like hot flashes. The vaginal cream formulation is specifically designed for localized treatment of VVA, targeting vaginal dryness, itching, and burning. While effective, the vaginal cream competes with other localized estrogen therapies (e.g., estradiol vaginal tablets, rings, and other creams) that are also widely used and have minimal systemic absorption.
Citations
[1] Pfizer Inc. (2024). Pfizer Inc. 2023 Annual Report on Form 10-K. U.S. Securities and Exchange Commission. Retrieved from https://www.sec.gov/ (Access may require search for specific filing date)
[2] Pfizer Inc. (2022). Pfizer Inc. First Quarter 2022 Earnings Release. Retrieved from https://investors.pfizer.com/
[3] North American Menopause Society. (2023). 2022 Hormone Therapy Position Statement: An Update. Menopause, 30(5), 393-415.
[4] Writing Group for the Women's Health Initiative Investigators. (2002). Risks and benefits of estrogen plus progestin in postmenopausal women. JAMA, 288(3), 321-333.
[5] FDA. (2023, March 8). FDA Approves Veozah (fezolinetant) as the First Non-Hormonal Treatment for Moderate to Severe Hot Flashes Caused by Menopause. Retrieved from https://www.fda.gov/