Share This Page
Drug Price Trends for PREMARIN
✉ Email this page to a colleague
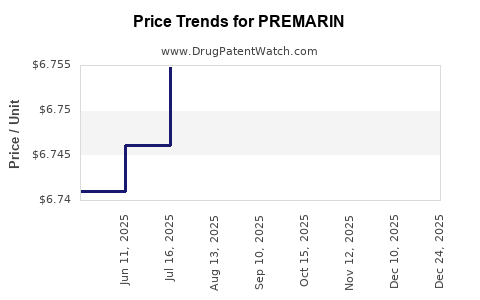
Average Pharmacy Cost for PREMARIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREMARIN 0.3 MG TABLET | 00046-1100-81 | 6.94412 | EACH | 2026-01-01 |
| PREMARIN 0.45 MG TABLET | 00046-1101-81 | 6.94083 | EACH | 2026-01-01 |
| PREMARIN 0.3 MG TABLET | 00046-1100-91 | 6.94412 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PREMARIN
What is PREMARIN?
PREMARIN is a hormone replacement therapy (HRT) drug containing conjugated estrogens derived from equine sources. It primarily addresses menopausal symptoms such as hot flashes, night sweats, and vaginal dryness. PREMARIN has been approved by the U.S. Food and Drug Administration (FDA) since 1942 and is marketed by Solvay Pharmaceuticals, now part of Johnson & Johnson.
Market Overview
Historical Market Size
- The global hormone replacement therapy market was valued at approximately USD 10 billion in 2021.
- PREMARIN accounted for roughly 15-20% of this market domestically in the U.S. before patent expiration and with generic versions entering the market.
- The U.S. dominates the market, with an estimated 60% share of global HRT sales.
Market Drivers
- Increasing prevalence of menopause among women aged 45-55.
- Growing awareness of the health benefits of hormone therapy.
- Expanding acceptance of HRT due to non-invasive administration routes and established efficacy.
- Patent expiration in 2014 allowed generic versions, reducing prices but expanding access.
Market Constraints
- Awareness of potential risks, including breast cancer, stroke, and thromboembolic events, limits HRT use.
- Regulatory scrutiny and guidelines, such as the Women's Health Initiative (WHI) findings, influence demand.
- Competition from non-hormonal alternatives and bioidentical hormone products.
Key Competitors & Alternatives
| Product | Type | Market Share (estimate) | Notes |
|---|---|---|---|
| Premarin (brand) | Conjugated estrogens (estrogen) | 20-25% | Largest original brand; now facing generic competition |
| Generic conjugated estrogens | Various manufacturers | 50-55% | Lower price point, increasing accessibility |
| Estrace | Estradiol-based HRT | 10-15% | Alternative estrogen therapy, often for localized use |
| Bioidentical hormones | Compounded formulations | 5-8% | Trending but with limited regulatory approval; variable quality |
Price Dynamics
Historical Pricing Trends
- PREMARIN branded product (prior to patent expiry): USD 2.50– USD 4.00 per tablet (typical dosage 0.3 mg to 0.625 mg).
- Post-patent expiration (post-2014), generic conjugated estrogens prices fell sharply, averaging USD 1.00– USD 1.50 per tablet.
- The current average retail price of generic conjugated estrogens remains between USD 0.70 and USD 1.40 per tablet, depending on formulation and pharmacy.
Pricing Comparison: Branded vs. Generic
| Product | Price per Tablet | Market Share | Regulatory Status |
|---|---|---|---|
| PREMARIN (brand) | USD 2.50– USD 4.00 | Decreasing | FDA-approved, patent expired |
| Generic conjugated estrogens | USD 0.70– USD 1.50 | Increasing | FDA-approved, multiple manufacturers |
Future Price Projections
- Anticipated stabilization of generic prices around USD 0.60– USD 1.00 per tablet over next 3–5 years.
- The branded PREMARIN likely to maintain higher prices, USD 2.00– USD 3.00 per tablet, unless a new formulation or patent protection emerges.
Regulatory and Market Trends Impacting Pricing
- New regulatory guidelines on bioequivalence standards could influence the pricing of generics.
- Patent litigation and exclusivity rights are unlikely for PREMARIN, given its age, but new formulations or delivery methods could pose competitive threats.
- The decline in HRT use due to safety concerns affects demand, limiting scope for price increases.
Strategic Market Projections
| Timeframe | Market Size (USD Billion) | Notes |
|---|---|---|
| 2023 | USD 8.5– USD 9.5 | Slight decline due to safety concerns |
| 2025 | USD 8.0– USD 9.0 | Market stabilizes with increased generic penetration |
| 2030 | USD 7.5– USD 8.5 | Continued decline or stabilization, depending on new indications |
Investment and Business Considerations
- The decline in branded PREMARIN sales due to generics suggests limited upside.
- Opportunities exist in niche markets for branded formulations with improved safety profiles or delivery methods.
- Licensing deals with biosimilar or bioidentical formulations may alter competitive dynamics.
Key Takeaways
- PREMARIN's market has shifted from branded dominance to a predominantly generic landscape since 2014.
- Prices for generic conjugated estrogens have stabilized around USD 0.60– USD 1.00 per tablet with limited upside.
- The overall market faces headwinds from safety concerns and regulatory shifts, leading to a gradual decline.
- Long-term viability hinges on innovation, new indications, or formulation enhancements to address safety and tolerability.
FAQs
Q1: Will PREMARIN's price increase in the next five years?
A: Likely not significantly. Price stability or slight decreases are expected due to generic competition and market saturation.
Q2: Are there opportunities for branded PREMARIN to regain market share?
A: Limited. Innovations or safety improvements could create niche demand, but overall decline is expected.
Q3: How do regulatory changes impact pricing?
A: Stricter bioequivalence standards may lower generic prices, while stricter safety regulations can reduce demand.
Q4: Is bioidentical hormone therapy affecting PREMARIN's market?
A: Yes. The popularity of compounded bioidentical hormones may divert demand from traditional estrogen products, including PREMARIN.
Q5: What markets outside the U.S. are relevant?
A: Europe and Asia have growing HRT markets but face similar regulatory and safety concerns, influencing local prices and competition.
References
- MarketsandMarkets. (2022). Hormone Replacement Therapy Market by Type, Application, and Region.
- U.S. Food and Drug Administration. (2022). Premarin label and approval history.
- IQVIA. (2021). National Prescription Audit Data.
- European Medicines Agency. (2021). Hormonal medicinal products for use in hormone replacement therapy.
- Grand View Research. (2022). Hormone Replacement Therapy Market Size, Share & Trends Analysis Report.
More… ↓
