PRADAXA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Pradaxa, and when can generic versions of Pradaxa launch?
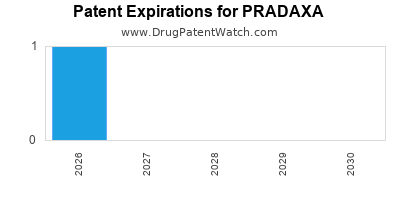
Pradaxa is a drug marketed by Boehringer Ingelheim and is included in two NDAs. There are three patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and forty-two patent family members in forty-four countries.
The generic ingredient in PRADAXA is dabigatran etexilate mesylate. There are twenty-six drug master file entries for this compound. Nine suppliers are listed for this compound. Additional details are available on the dabigatran etexilate mesylate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Pradaxa
A generic version of PRADAXA was approved as dabigatran etexilate mesylate by ALKEM LABS LTD on March 11th, 2020.
Summary for PRADAXA
| International Patents: | 142 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 4 |
| Raw Ingredient (Bulk) Api Vendors: | 12 |
| Clinical Trials: | 55 |
| Patent Applications: | 96 |
| Formulation / Manufacturing: | see details |
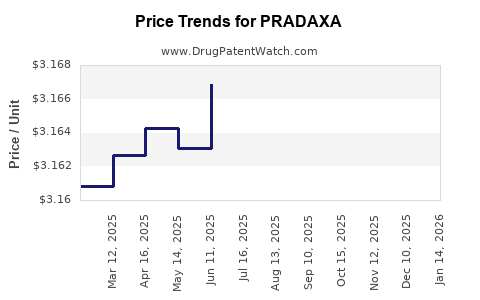
| Drug Prices: | Drug price information for PRADAXA |
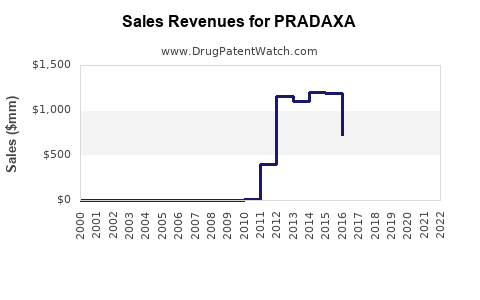
| Drug Sales Revenues: | Drug sales revenues for PRADAXA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PRADAXA |
| What excipients (inactive ingredients) are in PRADAXA? | PRADAXA excipients list |
| DailyMed Link: | PRADAXA at DailyMed |
Recent Clinical Trials for PRADAXA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Pfizer | Phase 1 |
| Laval University | Phase 2 |
| Bayer | Phase 1 |
Pharmacology for PRADAXA
| Drug Class | Direct Thrombin Inhibitor |
| Mechanism of Action | Thrombin Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for PRADAXA
Paragraph IV (Patent) Challenges for PRADAXA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PRADAXA | Capsules | dabigatran etexilate mesylate | eq. to 110 mg base | 022512 | 2 | 2015-12-15 |
| PRADAXA | Capsules | dabigatran etexilate mesylate | eq. to 75 mg base and 150 mg base | 022512 | 17 | 2014-10-20 |
US Patents and Regulatory Information for PRADAXA
PRADAXA is protected by three US patents and three FDA Regulatory Exclusivities.
Patents protecting PRADAXA
Film container
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
3-[(2-{[4-(hexyloxycarbonylaminoiminomethyl) phenylamino]methyl}-1-methyl-1H-benzimidazol-5-carbonyl)pyridin-2-ylamino- ]propionic acid ethylester methansulfonate and its use as a medicament
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Methods of using antibodies during anticoagulant therapy of dabigatran and/or related compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
FDA Regulatory Exclusivity protecting PRADAXA
TREATMENT OF VENOUS THROMBOEMBOLIC EVENTS (VTE) IN PEDIATRIC PATIENTS 8 TO LESS THAN 18 YEARS OF AGE WHO HAVE BEEN TREATED WITH A PARENTERAL ANTICOAGULANT FOR AT LEAST 5 DAYS AND TO REDUCE THE RISK OF RECURRENCE OF VTE IN PEDIATRIC PATIENTS 8 TO LESS THAN 18 YEARS OF AGE WHO HAVE BEEN PREVIOUSLY TREATED
Exclusivity Expiration: ⤷ Sign Up
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Sign Up
NEW PRODUCT
Exclusivity Expiration: ⤷ Sign Up
International Patents for PRADAXA
See the table below for patents covering PRADAXA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Luxembourg | 92117 | ⤷ Sign Up | |
| Portugal | 1660482 | ⤷ Sign Up | |
| Colombia | 5660265 | METANOSULFONATO DEL ESTER ETILICO DEL ACIDO 3-[(2-{[4-(HEXILOXI-CARBONILAMINO-IMINO-METIL)-FENILAMINO]-METIL}-1-METIL-1H-BENCI-MIDAZOL-5-CARBONIL)-PIRIDIN-2-IL-AMINO]-PROPIONICO Y SU UTILIZACION COMO MEDICAMENTO | ⤷ Sign Up |
| Taiwan | 200306188 | ⤷ Sign Up | |
| Cyprus | 1112796 | ⤷ Sign Up | |
| Japan | 2007056018 | MEDICINAL COMPOSITION FOR ORAL ADMINISTRATION CONTAINING 3-[(2-{[4-(HEXYLOXYCARBONYLAMINO-IMINO-METHYL)-PHENYLAMINO]-METHYL}-1-METHYL-1H-BENZIMIDAZOL-5-CARBONYL)-PYRIDIN-2-YL-AMINO]-PROPIONIC ACID ETHYL ESTER OR SALT THEREOF | ⤷ Sign Up |
| Estonia | 04716 | Diasendatud bitsüklilised heterotsüklid, nende valmistamine ja kasutamine ravimitena | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PRADAXA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1485094 | 54/2012 | Austria | ⤷ Sign Up | PRODUCT NAME: DABIGATRANETEXILAT UND SEINE SALZE, INSBESONDERE DABIGATRANETEXILATMESILAT; REGISTRATION NO/DATE: EU/1/08/442/001 - EU1/08/442/008 20080318 |
| 1870100 | 300538 | Netherlands | ⤷ Sign Up | PRODUCT NAME: DABIGATRAN ETEXILAAT MESILAAT; REGISTRATION NO/DATE: EU/1/08/442/001-008 20080318 |
| 0966454 | 08C0025 | France | ⤷ Sign Up | PRODUCT NAME: DABIGATRAN - ETEXILATE ET SES SELS EN PARTICULIER DABIGATRAN ETEXILATE MESILATE; REGISTRATION NO/DATE IN FRANCE: EU/1/08/442/001 DU 20080318; REGISTRATION NO/DATE AT EEC: EU/1/08/442/001 DU 20080318 |
| 1485094 | 132012902112338 | Italy | ⤷ Sign Up | PRODUCT NAME: DABIGATRAN ETEXILATO MESILATO(PRADAXA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/08/442/009-011-012-013-014, 20110801 |
| 2525812 | 17C1026 | France | ⤷ Sign Up | PRODUCT NAME: IDARUCIZUMAB; REGISTRATION NO/DATE: EU/1/15/1056 20151124 |
| 1870100 | 1290025-4 | Sweden | ⤷ Sign Up | PRODUCT NAME: DABIGATRANETEXILATMESILAT; REG. NO/DATE: EU/1/08/442/001-008 20080318 |
| 0966454 | CA 2008 00032 | Denmark | ⤷ Sign Up | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


