Last updated: February 19, 2026
Perphenazine, an established antipsychotic medication, exhibits a stable but mature market. Its primary therapeutic indications, schizophrenia and severe nausea and vomiting, remain consistent. The drug's financial trajectory is characterized by consistent, albeit low, revenue streams driven by generic manufacturing and its established place in treatment guidelines. Patent expirations have led to widespread generic competition, suppressing price increases and limiting significant market expansion.
What is the Current Market Size and Segmentation for Perphenazine?
The global market for perphenazine is difficult to quantify precisely due to its generic status and inclusion within broader antipsychotic and antiemetic market reports. However, estimates place the annual global sales in the tens of millions of U.S. dollars. The market is segmented by:
- Therapeutic Indication:
- Schizophrenia and other psychotic disorders.
- Severe nausea and vomiting, particularly post-operative.
- Formulation:
- Oral tablets.
- Injectable solutions.
- Geographic Region: North America, Europe, and Asia-Pacific represent the largest markets, driven by healthcare infrastructure and prescription volumes. Emerging markets show potential for gradual growth as access to mental health and supportive care increases.
- End-User: Hospitals, psychiatric institutions, and retail pharmacies constitute the primary distribution channels.
What is the Historical Patent Landscape for Perphenazine?
Perphenazine's core patents have long expired, enabling generic manufacturing. The original patent for perphenazine was filed by Schering Corporation and granted in the 1950s. Subsequent patents likely covered specific formulations, manufacturing processes, or combination therapies, but these have also aged out of protection.
- Original Patent Filing Era: Mid-1950s.
- Last Major Expirations: Prior to the year 2000.
This extensive patent expiry is the primary driver behind the drug's current market structure. The absence of active market exclusivity for any specific originator product allows for unhindered generic competition.
Who are the Key Manufacturers and Competitors in the Perphenazine Market?
The perphenazine market is dominated by generic pharmaceutical manufacturers. There are no significant originator companies actively marketing a branded version with market exclusivity. Key players include companies specializing in the production of established generic drugs.
- Major Generic Manufacturers (Examples, not exhaustive):
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Generic pharmaceutical divisions of larger pharmaceutical conglomerates.
Competition is primarily price-based, with manufacturers vying for market share through cost-efficient production and distribution. The market is characterized by a fragmented supplier base rather than a few dominant players.
What are the Regulatory Pathways and Requirements for Perphenazine?
As an established drug with a long history of use, perphenazine's regulatory pathways are well-defined. New market entrants or manufacturers of generic versions must demonstrate bioequivalence to the reference listed drug (RLD).
- Key Regulatory Bodies:
- U.S. Food and Drug Administration (FDA).
- European Medicines Agency (EMA).
- Other national regulatory authorities.
- Generic Approval Pathway: Abbreviated New Drug Application (ANDA) in the U.S., requiring demonstration of equivalence.
- Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) is mandatory.
- Post-Market Surveillance: Ongoing pharmacovigilance and reporting of adverse events are required.
The regulatory hurdles are standard for generic drugs, meaning that approvals are generally achievable for manufacturers meeting quality and bioequivalence standards.
What are the Current Pricing Trends and Reimbursement Policies for Perphenazine?
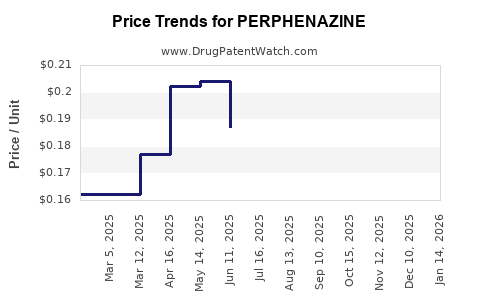
Perphenazine pricing is characteristic of a mature generic drug market. Prices are low and subject to downward pressure from competition. Reimbursement policies are generally favorable due to its inclusion in standard treatment guidelines and formulary listings.
- Pricing:
- Low per-unit cost, typically in the range of cents to a few dollars per tablet or vial depending on dosage and packaging.
- Prices are sensitive to bulk purchasing and contract negotiations.
- Reimbursement:
- Covered by most public and private health insurance plans in developed countries.
- Often included in hospital formularies and national health service drug lists.
- Reimbursement rates are typically based on established generic drug pricing benchmarks.
The financial trajectory of perphenazine is therefore tied to volume rather than price appreciation.
What are the Key Market Drivers and Restraints for Perphenazine?
The market for perphenazine is driven by its established efficacy, low cost, and continued use in specific patient populations. Restraints are primarily linked to the availability of newer therapeutic agents with improved side-effect profiles and market penetration.
- Market Drivers:
- Established Efficacy: Proven effectiveness in treating schizophrenia and severe emesis.
- Cost-Effectiveness: Significantly lower cost compared to newer antipsychotics.
- Treatment Guidelines: Continued inclusion in standard care protocols for specific indications.
- Accessibility: Wide availability of generic formulations globally.
- Market Restraints:
- Side Effect Profile: Potential for extrapyramidal symptoms (EPS) and other adverse effects, leading to preference for newer agents.
- Availability of Newer Antipsychotics: Second-generation antipsychotics (SGAs) often have better tolerability and broader efficacy in certain patient subgroups.
- Limited Innovation: No significant new drug development or expanded indications for perphenazine.
- Market Saturation: The generic market is mature with established supply chains.
What is the Future Outlook and Growth Potential for Perphenazine?
The future outlook for perphenazine is one of continued stability rather than significant growth. Its role will likely remain as a cost-effective option for specific patient populations and in resource-limited settings.
- Projected Growth Rate: Low single-digit percentage growth, driven primarily by population growth and increasing healthcare access in emerging markets.
- Key Future Trends:
- Sustained Demand in Niche Indications: Continued use in severe nausea where other agents may be contraindicated or less effective.
- Role in Cost-Containment Strategies: Adoption in healthcare systems seeking to manage rising pharmaceutical costs.
- Potential for Combination Therapies: While unlikely to be a focus of new development, existing off-label or established combination uses may persist.
- Geographic Expansion: Gradual uptake in emerging markets as healthcare infrastructure improves.
Significant market expansion is not anticipated. The drug's financial trajectory will continue to be dictated by generic sales volume.
Key Takeaways
Perphenazine operates in a mature, price-competitive generic drug market. Its financial trajectory is characterized by stable, low-margin revenues driven by consistent demand in schizophrenia and severe emesis, particularly where cost is a significant factor. The absence of patent protection and the availability of newer, more tolerable agents limit significant growth potential. Manufacturers' focus is on cost-efficient production and distribution to maintain market share.
Frequently Asked Questions
1. What is the primary mechanism of action of perphenazine?
Perphenazine is a dopamine antagonist. It blocks dopamine D2 receptors in the brain, which is believed to be the primary mechanism for its antipsychotic effects and its ability to reduce nausea and vomiting.
2. Are there any new clinical trials or research involving perphenazine?
As a long-established drug, there is minimal ongoing clinical trial activity for perphenazine related to new indications or novel formulations. Research typically focuses on its use within existing therapeutic areas or comparative studies against newer agents.
3. What are the main side effects associated with perphenazine?
Common side effects include drowsiness, dry mouth, blurred vision, constipation, and dizziness. More serious side effects can include extrapyramidal symptoms (movement disorders like tremors and stiffness), tardive dyskinesia, and neuroleptic malignant syndrome.
4. How does perphenazine compare to newer antipsychotics in terms of efficacy and safety?
Perphenazine is generally considered less effective and to have a more challenging side effect profile, particularly concerning extrapyramidal symptoms, compared to second-generation antipsychotics (SGAs). SGAs often offer improved tolerability and a reduced risk of motor side effects.
5. What is the typical dosage range for perphenazine in treating schizophrenia?
Dosage for schizophrenia is highly individualized and depends on patient response and tolerance. Typical oral dosages can range from 8 mg to 24 mg per day, divided into multiple doses, though higher doses may be used under strict medical supervision.
6. Can perphenazine be used during pregnancy?
The use of perphenazine during pregnancy is generally not recommended unless the potential benefits outweigh the potential risks to the fetus. It should only be used if clearly needed and prescribed by a healthcare professional.
7. What are the key differences between perphenazine and other phenothiazines?
Perphenazine is a member of the phenothiazine class of antipsychotics. While sharing a similar mechanism of action (dopamine blockade), other phenothiazines like chlorpromazine and thioridazine differ in their specific receptor binding affinities, potency, and side effect profiles. Perphenazine is considered a higher-potency, lower-sedating phenothiazine compared to some others in the class.
8. How is perphenazine administered for severe nausea and vomiting?
For severe nausea and vomiting, perphenazine is often administered intramuscularly as an injectable solution. Oral formulations are also used.
9. What is the shelf life of perphenazine tablets and injectable solutions?
The shelf life of perphenazine products typically ranges from 2 to 5 years, depending on the specific manufacturer, formulation, and storage conditions. This information is detailed on the product packaging.
10. In which countries is perphenazine most widely prescribed?
Perphenazine is prescribed globally. Its widespread use is noted in countries with established healthcare systems and in regions where cost-effective treatment options are prioritized, including North America, Europe, and parts of Asia.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/information-drug-products/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[2] European Medicines Agency. (n.d.). European Public Assessment Reports (EPARs). Retrieved from https://www.ema.europa.eu/en/medicines/human/EPARs
[3] IMS Health (now IQVIA). (Various Years). Market share data and sales reports for antipsychotic and antiemetic drug classes. (Proprietary data, specific report titles unavailable for public citation).
[4] Schering Corporation. (1950s). Original patent filings for Perphenazine. (Public patent databases such as USPTO, Espacenet).
[5] National Institute of Mental Health. (n.d.). Schizophrenia. Retrieved from https://www.nimh.nih.gov/health/topics/schizophrenia
[6] World Health Organization. (n.d.). International Classification of Diseases (ICD). Retrieved from https://www.who.int/standards/classifications/international-classification-of-diseases