Last updated: February 19, 2026
Oxaprozin, a nonsteroidal anti-inflammatory drug (NSAID), operates within a mature segment of the pharmaceutical market characterized by established therapeutic roles and significant generic competition. Its market trajectory is primarily shaped by its efficacy in treating inflammatory and pain conditions, balanced against the pricing pressures inherent in a post-patent landscape.
What is Oxaprozin's Therapeutic Indication and Mechanism of Action?
Oxaprozin is indicated for the relief of signs and symptoms of rheumatoid arthritis and osteoarthritis. It functions as a non-selective inhibitor of cyclooxygenase (COX) enzymes, COX-1 and COX-2. By inhibiting these enzymes, oxaprozin reduces the synthesis of prostaglandins, which are mediators of inflammation, pain, and fever. This mechanism aligns with other NSAIDs, positioning it as an option for symptomatic relief in chronic inflammatory conditions.
What is the Current Market Status of Oxaprozin?
Oxaprozin is primarily marketed as a generic drug. The original patent protection for oxaprozin expired decades ago, leading to widespread generic availability. This has resulted in a highly competitive market with multiple manufacturers offering the drug at significantly lower price points than innovator brands. The market for oxaprozin is therefore driven by volume and cost-effectiveness rather than innovation or premium pricing.
Key Market Characteristics:
- Generic Domination: The market is dominated by generic manufacturers.
- Price Sensitivity: Pricing is a critical factor in market share due to generic competition.
- Established Efficacy: The drug's therapeutic profile is well-understood and accepted.
- Mature Market: Oxaprozin is not a novel drug; its growth potential is limited by its established place in therapy.
Who are the Key Manufacturers and Suppliers?
The manufacturing and supply chain for oxaprozin involves numerous pharmaceutical companies specializing in generic drug production. These companies source active pharmaceutical ingredients (APIs) and formulate finished dosage forms. Specific market share data for individual generic manufacturers is not publicly disclosed in the same manner as for patented drugs. However, companies with robust generic portfolios and established distribution networks are typically significant players.
Examples of companies that have historically produced or currently supply oxaprozin include:
- Teva Pharmaceutical Industries
- Mylan N.V. (now Viatris)
- Apotex Inc.
- Sandoz (a division of Novartis)
- Various regional generic drug manufacturers.
The competitive landscape is dynamic, with companies adjusting production based on API costs, regulatory approvals, and market demand.
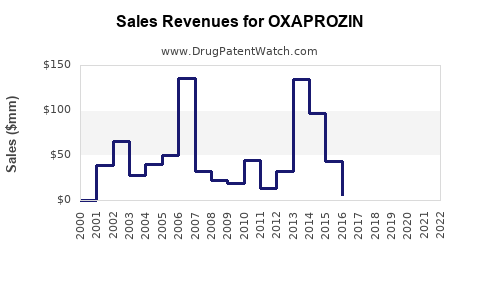
What is the Global Sales Performance of Oxaprozin?
As a generic drug, aggregate global sales figures for oxaprozin are not tracked by major market research firms in the same detailed fashion as branded pharmaceuticals. Sales performance is largely consolidated within the broader NSAID generic market. Revenue generated by individual generic products is typically lower and more fragmented compared to patented drugs.
Estimates suggest that the global market for NSAIDs, encompassing both branded and generic products, is substantial, with generics forming the largest segment by volume. However, the specific contribution of oxaprozin to this total is difficult to isolate due to the lack of dedicated market research reports for individual generic NSAIDs. Pricing is often measured in cents per unit, with revenue driven by high-volume sales to wholesalers, pharmacies, and healthcare systems.
Factors Influencing Sales:
- Prescription Volume: Dependent on physician prescribing habits for osteoarthritis and rheumatoid arthritis.
- Reimbursement Policies: Payer formularies and co-payment structures influence patient access and physician choice.
- Competition from Other NSAIDs: Oxaprozin competes with other generics like ibuprofen, naproxen, diclofenac, and celecoxib (a COX-2 selective inhibitor, though often available generically).
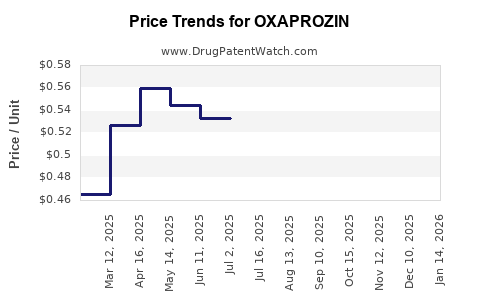
What is the Pricing Structure and Profitability for Generic Oxaprozin?
The pricing of generic oxaprozin is dictated by competitive market forces. Following patent expiry, multiple manufacturers enter the market, driving prices down. The profit margins for generic drugs are generally lower than for patented drugs, relying on high production volumes and efficient supply chains.
The typical pricing structure involves:
- API Cost: The cost of sourcing bulk oxaprozin API.
- Manufacturing and Formulation: Expenses associated with producing tablets or capsules.
- Packaging and Distribution: Costs for labeling, bottling, and shipping.
- Regulatory Compliance: Costs associated with maintaining good manufacturing practices (GMP) and regulatory filings.
- Wholesaler and Pharmacy Markup: Markups applied at each stage of the distribution channel.
Profitability is achieved through economies of scale. Companies that can produce oxaprozin at the lowest cost per unit, while maintaining quality standards, are best positioned. This often involves large-scale manufacturing facilities and optimized supply chain management. Prices can vary based on the strength of the tablet (e.g., 600 mg) and the number of units per bottle.
Indicative Pricing (Wholesale Acquisition Cost - WAC):
| Drug Strength |
Typical Unit Count |
Indicative WAC Range (USD) |
| 600 mg |
30 tablets |
$5 - $15 |
| 600 mg |
100 tablets |
$10 - $30 |
Note: These figures represent approximate wholesale acquisition costs and can fluctuate significantly based on the supplier, contract terms, and market conditions. Actual selling prices to consumers will be higher due to pharmacy markups and insurance.
What are the Intellectual Property and Patent Expiries?
Oxaprozin was originally developed by Eli Lilly and Company. The primary composition of matter patent for oxaprozin has long since expired. This means that no company holds exclusive rights to the molecule itself.
- Original Composition of Matter Patent: Expired.
- Method of Use Patents: Any patents related to specific new therapeutic uses or formulations would also have expired or are subject to challenges.
The absence of patent protection is the defining characteristic of oxaprozin's market status. This allows any manufacturer meeting regulatory requirements to produce and sell the drug. There are no significant ongoing patent disputes or litigation that would impede generic competition.
What are the Regulatory Landscapes Affecting Oxaprozin?
Oxaprozin is subject to the regulatory oversight of health authorities worldwide, primarily the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Generic drugs require Abbreviated New Drug Applications (ANDAs) in the U.S. or similar marketing authorization applications in other regions. These applications demonstrate bioequivalence to the reference listed drug (RLD).
Key Regulatory Considerations:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure drug quality, safety, and efficacy. Regular inspections by regulatory bodies are standard.
- ANDA Approval Process: Generic companies must submit extensive data proving their product is therapeutically equivalent to the RLD.
- Pharmacovigilance: Post-market surveillance for adverse events is a continuous requirement.
- Labeling Requirements: Generic labels must be equivalent to the RLD label in terms of indications, contraindications, warnings, and dosage, with specific exceptions for intellectual property.
- API Sourcing: Regulatory bodies scrutinize the origin and quality of active pharmaceutical ingredients.
The regulatory environment for generics is well-established, creating a stable but competitive framework. Changes in regulatory standards or enforcement can impact manufacturing costs and market access.
What are the Competitive Alternatives to Oxaprozin?
Oxaprozin competes within the broader NSAID class and with other pain management modalities.
Direct NSAID Competitors (Generic and Branded):
- Ibuprofen: Widely available, low cost, multiple strengths.
- Naproxen: Long-acting, common generic.
- Diclofenac: Available in various formulations, including topical.
- Indomethacin: Potent NSAID, often used for gout flares.
- Celecoxib (Celebrex): COX-2 selective inhibitor, initially branded, now available generically. Offers a potentially reduced risk of gastrointestinal side effects compared to non-selective NSAIDs.
- Meloxicam: Preferentially selective for COX-2.
Other Pain Management Options:
- Acetaminophen (Paracetamol): Analgesic and antipyretic, generally safer GI profile than NSAIDs.
- Opioid Analgesics: For severe pain, but carry significant risks of addiction and side effects.
- Disease-Modifying Antirheumatic Drugs (DMARDs): For rheumatoid arthritis, these drugs target the underlying disease process, not just symptoms. Examples include methotrexate, sulfasalazine, and biologics.
- Physical Therapy and Rehabilitation: Non-pharmacological approaches.
The choice of treatment depends on the specific condition, patient comorbidities, severity of symptoms, and risk tolerance for side effects. Oxaprozin's position is as one among many options for symptomatic relief of inflammation and pain.
What is the Financial Trajectory and Future Outlook for Oxaprozin?
The financial trajectory for oxaprozin is characterized by stability within the generic drug market, with limited prospects for significant growth. Revenue streams are primarily derived from sustained, high-volume sales.
Current Financial State:
- Revenue: Consistent but modest, driven by generic sales volume.
- Profitability: Dependent on efficient manufacturing and cost control by generic companies.
- Market Share: Stable within the generic NSAID segment, unlikely to expand significantly.
Future Outlook:
- Continued Generic Competition: The market will remain highly competitive.
- Pricing Pressure: Downward price pressure is expected to persist.
- Therapeutic Stagnation: No significant new indications or formulations are anticipated.
- Competition from Newer Therapies: While oxaprozin addresses inflammatory conditions, advancements in biologics and targeted therapies for diseases like rheumatoid arthritis may shift treatment paradigms, potentially reducing reliance on traditional NSAIDs for certain patient populations. However, for osteoarthritis and general inflammatory pain, NSAIDs remain a first-line or adjunctive therapy.
- Cost-Containment Measures: Healthcare systems worldwide continue to seek cost efficiencies, favoring generic medications. This supports the sustained use of oxaprozin.
The financial future of oxaprozin is intrinsically linked to the broader generic pharmaceutical market. It is unlikely to experience a resurgence or significant market expansion. Its value proposition lies in its established efficacy and affordability as a generic option for inflammatory and pain management.
Key Takeaways
- Oxaprozin is a generic NSAID used for rheumatoid arthritis and osteoarthritis, facing intense price competition.
- Its market is dominated by numerous generic manufacturers, with no significant patent protection remaining.
- Global sales are consolidated within the broader generic NSAID market, with revenue driven by high-volume, low-margin sales.
- The drug competes with a wide array of other NSAIDs and alternative pain management therapies.
- The future financial trajectory is stable but limited, characterized by sustained generic sales and persistent pricing pressure.
Frequently Asked Questions
-
What is the primary risk associated with long-term oxaprozin use?
The primary risks associated with long-term NSAID use, including oxaprozin, involve gastrointestinal issues such as ulcers and bleeding, as well as potential cardiovascular and renal adverse effects.
-
Can oxaprozin be used for acute pain management?
While primarily indicated for chronic inflammatory conditions like rheumatoid arthritis and osteoarthritis, oxaprozin can also be used for the symptomatic relief of acute pain associated with inflammation.
-
How does oxaprozin compare in terms of potency to other generic NSAIDs like ibuprofen or naproxen?
Potency varies between NSAIDs, but oxaprozin is generally considered to have a long half-life, allowing for once-daily dosing and providing sustained anti-inflammatory and analgesic effects comparable to other widely used NSAIDs.
-
Are there any specific patient populations for whom oxaprozin is contraindicated?
Oxaprozin is contraindicated in patients with a known hypersensitivity to oxaprozin or other NSAIDs, and in those who have experienced asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. It is also generally avoided in patients with active peptic ulceration or a history of gastrointestinal bleeding.
-
What are the typical dosage forms and strengths available for generic oxaprozin?
Oxaprozin is typically available as oral tablets, most commonly in a 600 mg strength, formulated for once-daily administration.
Citations
[1] Eli Lilly and Company. (n.d.). Oxaprozin (Daypro) prescribing information. U.S. Food & Drug Administration.
[2] National Institutes of Health. (n.d.). Oxaprozin. MedlinePlus.
[3] U.S. Food & Drug Administration. (n.d.). Generic Drugs.
[4] European Medicines Agency. (n.d.). Medicines.