Last updated: February 19, 2026
Introduction
Otezla (apremilast), a small molecule inhibitor of phosphodiesterase 4 (PDE4), is approved for the treatment of moderate to severe plaque psoriasis, psoriatic arthritis, and Behçet's disease oral ulcers. Developed by Celgene, now part of Bristol Myers Squibb (BMS), Otezla has established a significant market presence by offering an oral, non-biologic treatment option. This analysis details its market dynamics, key growth drivers, competitive landscape, and projected financial trajectory based on available data.
Otezla's Approved Indications and Mechanism of Action
Otezla targets PDE4, an enzyme that degrades cyclic adenosine monophosphate (cAMP). By inhibiting PDE4, Otezla increases intracellular cAMP levels, leading to a reduction in the production of pro-inflammatory mediators and an increase in anti-inflammatory mediators. This dual action is central to its efficacy across its approved indications:
- Plaque Psoriasis: Otezla is indicated for adult patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy. Its efficacy is demonstrated in improving skin clearance and reducing disease severity.
- Psoriatic Arthritis: For adult patients with active psoriatic arthritis, Otezla is indicated to decrease articular inflammation. It has shown effectiveness in reducing joint pain, swelling, and physical function limitations.
- Behçet's Disease Oral Ulcers: Otezla is the first and only approved therapy for oral ulcers associated with Behçet's disease in adult patients. This indication addresses a significant unmet need for a targeted treatment.
Market Dynamics and Growth Drivers
Otezla's market position is influenced by several key dynamics:
- Oral Convenience: As an oral therapy, Otezla offers a distinct advantage over injectable biologics, appealing to patients seeking convenience and avoiding the challenges associated with self-injection. This has been a primary driver of its adoption since launch.
- Non-Biologic Profile: Its non-biologic nature addresses concerns related to immunogenicity and the potential for injection site reactions or systemic immunosuppression associated with biologic therapies. This also positions it as a potential option for patients who have failed or are contraindicated for biologics.
- Broad Patient Population: The prevalence of plaque psoriasis and psoriatic arthritis affects millions globally, providing a large addressable patient pool. The approval for Behçet's disease oral ulcers, while a smaller indication, represents a significant therapeutic advancement.
- Expansion into New Markets: Bristol Myers Squibb has pursued geographical expansion for Otezla, securing approvals and launching in numerous countries worldwide, thereby broadening its revenue base.
- Label Expansions and Real-World Evidence: Ongoing research and the accumulation of real-world evidence further support Otezla's efficacy and safety profile, potentially leading to expanded physician confidence and patient access.
Competitive Landscape
Otezla operates within a competitive market for inflammatory diseases, particularly for psoriasis and psoriatic arthritis. Its primary competitors include:
- Biologic Therapies:
- Tumor Necrosis Factor (TNF) inhibitors: Examples include adalimumab (Humira), etanercept (Enbrel), and infliximab (Remicade). These are often considered first-line systemic treatments for moderate to severe psoriasis and psoriatic arthritis due to their robust efficacy.
- Interleukin (IL) inhibitors: This class includes IL-12/23 inhibitors (e.g., ustekinumab [Stelara]), IL-17 inhibitors (e.g., secukinumab [Cosentyx], ixekizumab [Taltz]), and IL-23 inhibitors (e.g., guselkumab [Tremfya], risankizumab [Skyrizi]). These biologics offer high levels of efficacy and improved safety profiles in certain patient populations compared to older TNF inhibitors.
- Other Small Molecules:
- JAK inhibitors: Tofacitinib (Xeljanz) is approved for psoriatic arthritis and has also been explored for psoriasis. However, JAK inhibitors carry a boxed warning regarding serious infections, thrombosis, malignancy, and cardiovascular events, which can influence prescribing patterns.
- Newer Oral Therapies: The development of novel oral agents continues, posing a potential future threat to Otezla's market share.
Otezla's competitive advantage lies in its oral administration and its non-biologic status, positioning it as a valuable option for patients who prefer oral treatment or have contraindications to biologics. However, the high efficacy of many biologic agents, particularly IL inhibitors, often leads to their prioritization in treatment algorithms for severe disease.
Financial Trajectory and Key Performance Indicators
Bristol Myers Squibb has reported Otezla's financial performance consistently. Analysis of its revenue trends provides insights into its market penetration and growth potential.
Otezla Net Revenue (USD Billions)
| Year |
Net Revenue |
Year-over-Year Growth (%) |
| 2019 |
1.73 |
20.3 |
| 2020 |
2.18 |
25.4 |
| 2021 |
2.22 |
1.8 |
| 2022 |
2.42 |
9.0 |
| 2023 |
2.56 |
5.8 |
Source: Bristol Myers Squibb SEC Filings & Investor Reports (2019-2023)
Analysis of Financial Trends:
- Strong Initial Growth (2019-2020): The period following the acquisition by Bristol Myers Squibb in late 2019 saw robust growth, driven by market expansion and the integration into BMS's portfolio.
- Slowing Growth (2021-2022): Revenue growth decelerated in 2021, reflecting increased competition from newer biologics and the maturation of the market in established regions. 2022 showed a rebound in growth, indicating sustained demand.
- Continued Revenue Generation (2023): Otezla continues to be a significant revenue-generating asset for BMS, demonstrating its resilience in a competitive market. The growth in 2023, while modest, signifies ongoing prescription volume and market presence.
- Geographic Contribution: The majority of Otezla's revenue is generated in the United States, its largest and most developed market. International markets contribute a smaller but growing portion, reflecting ongoing global expansion efforts.
Factors Influencing Future Financial Performance:
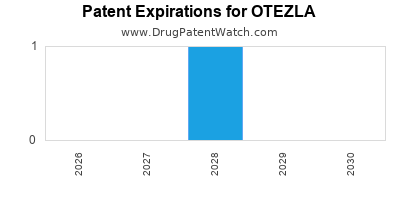
- Patent Expirations and Generic Competition: The patent landscape for Otezla is critical. As patents approach expiration, the threat of generic competition looms, which could significantly impact revenue. BMS has actively worked to extend market exclusivity through various intellectual property strategies and lifecycle management. For example, patents related to the formulation and method of use are key considerations.
- Lifecycle Management and New Formulations: BMS may explore strategies such as new formulations or combination therapies to maintain Otezla's market position and extend its commercial lifecycle.
- Market Access and Reimbursement: Continued favorable reimbursement policies and market access in key geographies are essential for maintaining prescription volumes. Payer restrictions on biologic therapies can indirectly benefit Otezla by making it a more accessible option.
- Emerging Therapies: The continuous development of novel treatment modalities, including next-generation biologics and advanced small molecules, will present ongoing competitive pressures.
- Pediatric Indications: Securing approvals for pediatric populations in its approved indications could expand the addressable market and drive future growth.
Otezla's Intellectual Property Landscape
A critical aspect of Otezla's financial trajectory is its patent protection. Bristol Myers Squibb has strategically filed patents covering various aspects of Otezla, including:
- Composition of Matter Patents: These are typically the strongest patents, covering the molecule itself.
- Method of Use Patents: These protect specific therapeutic applications, such as treatment for psoriasis or psoriatic arthritis.
- Formulation Patents: These cover specific dosage forms, delivery systems, or combinations.
- Process Patents: These relate to the methods of manufacturing the drug.
The expiration of key patents will open the door for generic manufacturers. Understanding the expiration dates of composition of matter and method of use patents is paramount for forecasting market exclusivity. For instance, while many early patents may have expired or are nearing expiration, BMS has been active in securing and defending secondary patents related to formulations and therapeutic uses. The precise dates and scope of these patents, along with any ongoing litigation, are subject to continuous monitoring.
Key Takeaways
Otezla has established itself as a leading oral, non-biologic treatment for plaque psoriasis, psoriatic arthritis, and Behçet's disease oral ulcers. Its convenience and safety profile continue to drive consistent revenue generation for Bristol Myers Squibb. While the market is competitive with highly effective biologic agents, Otezla's distinct profile ensures its continued relevance. The primary financial risk factor is the impending expiration of key patents and the subsequent entry of generic competitors. Bristol Myers Squibb's strategic patent management and potential lifecycle extensions will be critical in mitigating this risk and sustaining Otezla's commercial viability.
Frequently Asked Questions
-
What is the primary advantage of Otezla compared to biologic treatments for psoriasis and psoriatic arthritis?
Otezla's primary advantage is its oral administration, which offers greater convenience and avoids the need for injections. It is also a non-biologic therapy, which may appeal to patients concerned about immunogenicity or the potential side effects associated with biologics.
-
What is the projected impact of patent expirations on Otezla's revenue?
Patent expirations will enable the entry of generic versions of Otezla, which is expected to lead to a significant decline in net revenue for Bristol Myers Squibb due to price erosion and increased competition, similar to trends observed with other branded pharmaceuticals after patent expiry.
-
In which geographic regions does Otezla generate the most revenue?
The United States is Otezla's largest market, contributing the substantial majority of its global revenue. International markets represent a smaller but growing portion of its sales.
-
What is the mechanism of action by which Otezla treats inflammatory conditions?
Otezla inhibits phosphodiesterase 4 (PDE4), an enzyme involved in the inflammatory pathway. By inhibiting PDE4, Otezla increases intracellular cyclic adenosine monophosphate (cAMP) levels, which in turn modulates the production of pro-inflammatory and anti-inflammatory mediators, ultimately reducing inflammation.
-
Are there any other approved indications for Otezla beyond psoriasis, psoriatic arthritis, and Behçet's disease oral ulcers?
As of the latest available information, Otezla is approved for moderate to severe plaque psoriasis, active psoriatic arthritis, and oral ulcers associated with Behçet's disease in adult patients. No other major indications have been widely approved or are nearing widespread approval.
Citations
[1] Bristol Myers Squibb. (2019-2023). Form 10-K and 10-Q Filings. U.S. Securities and Exchange Commission.
[2] Bristol Myers Squibb. (2019-2023). Investor Relations Presentations and Reports.
[3] U.S. Food & Drug Administration. (n.d.). Drug Approval Database. Retrieved from [FDA Website]
[4] European Medicines Agency. (n.d.). European Public Assessment Reports. Retrieved from [EMA Website]
[5] Pharmaceutical company press releases and official product information.