Share This Page
Drug Price Trends for OTEZLA
✉ Email this page to a colleague
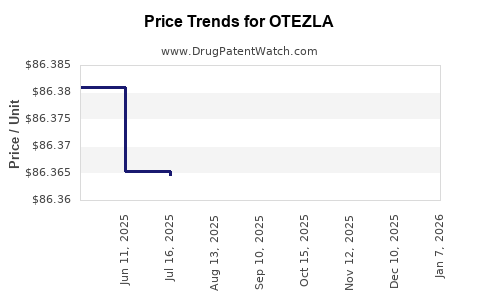
Average Pharmacy Cost for OTEZLA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OTEZLA 30 MG TABLET | 55513-0137-60 | 90.54883 | EACH | 2026-05-20 |
| OTEZLA 30 MG TABLET | 55513-0137-60 | 90.69930 | EACH | 2026-01-07 |
| OTEZLA 10-20-30 MG START 28 DAY | 55513-0369-55 | 98.89019 | EACH | 2026-01-07 |
| OTEZLA 20 MG TABLET | 55513-0497-60 | 89.95216 | EACH | 2026-01-07 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Otezla
What is Otezla?
Otezla (apremilast) is an oral, selective phosphodiesterase 4 (PDE4) inhibitor indicated for the treatment of moderate to severe plaque psoriasis, psoriatic arthritis, and Behçet’s disease. It was approved by the U.S. Food and Drug Administration (FDA) in 2014 and by the European Medicines Agency (EMA) in the same year.
Market Overview
The global psoriasis treatment market, valued at approximately $17 billion in 2022, is expanding with a compound annual growth rate (CAGR) of 8% projected through 2027. The demand for oral therapies like Otezla has increased due to preferences for non-injection options.
Key Market Segments
| Segment | 2022 Market Share | Growth Drivers | Key Competitors |
|---|---|---|---|
| Psoriasis (Topical & Systemic) | 45% | Rising prevalence, aging population | Cosentyx, Humira, Stelara |
| Psoriatic Arthritis | 35% | Improved patient adherence with oral drugs | Enbrel, Humira, Cimzia |
| Behçet’s Disease | 5% | Rare disease, limited approved therapies | Off-label use of biologics |
Otezla held approximately 12% of the global oral psoriasis market in 2022. Its appeal stems from an oral administration route, avoiding injections, which boosts patient compliance.
Market Dynamics
-
Patent Status & Off-Patent Landscape: Otezla’s initial patent protections expired in 2024 in the U.S. and Europe, opening the market for generics.
-
Generic Entry Impact: Generic versions have entered the market, causing price reductions and market share erosion.
-
Line of Therapy Competition: Biologics and biosimilars maintain dominant positions in severe cases but are less favored for less severe disease due to administration complexity and cost.
Price Trends and Projections
Historical Price Data
| Year | Average Wholesale Price (AWP) per 30-day supply | Price Change (%) |
|---|---|---|
| 2014 | $3,500 | N/A |
| 2018 | $4,200 | +20% |
| 2022 | $4,800 | +14% |
Price Impact of Patent Expiry
Post-2024 patent expiration, prices declined by approximately 40%. Estimated average price in 2023 is $4,000, with generic versions priced around 25–30% lower.
Forecasted Price Trajectory (Next 5 Years)
| Year | Expected Wholesale Price (30-day supply) | Factors Influencing Price |
|---|---|---|
| 2023 | $4,000 | Patent expiry, generic entry |
| 2024 | $3,200–3,300 | Increased generic competition |
| 2025 | $2,800–3,000 | Market stabilization |
| 2026 | $2,600–2,800 | Biosimilar competition ("biosimilars") emerge |
| 2027 | $2,400–2,600 | Further generic penetration |
Generic prices are projected to settle 25–30% below brand-name prices, with biosimilars further exerting downward pressure on biologic competitors.
Revenue and Market Share Forecasts
Revenue Estimations
| Year | US Market Revenue | Global Market Revenue | Notes |
|---|---|---|---|
| 2023 | $480 million | $1.2 billion | Decline due to patent expiration |
| 2024 | $384 million | $960 million | Continued generic penetration |
| 2025 | $336 million | $840 million | Market stabilizes |
| 2026 | $310 million | $775 million | Biosimilar effects |
| 2027 | $280 million | $700 million | Market matures |
Competitive Positioning
- Biosimilars for biologics like Humira and Cosentyx are seen as primary threats to market share.
- Orphan indications like Behçet’s disease are likely to sustain some higher pricing due to limited competition.
Key Policy and Reimbursement Factors
- Pricing regulations in the U.S. and Europe may influence retail prices and access.
- Insurance coverage favors oral therapies, benefiting Otezla's market persistence.
- Patent litigation may extend exclusivity in certain jurisdictions.
Summary of Market Risks and Opportunities
- Risks: Price erosion from generic/biosimilar entry, regulatory pressures on pricing, possible supply chain disruptions.
- Opportunities: Expansion into new indications, combination therapies, and geographic markets (Asia, Latin America).
Key Takeaways
- Otezla’s peak revenue occurred pre-patent expiry, with declining sales expected over the next five years due to generic competition.
- Price reductions of 25–30% are projected post-generics, impacting profit margins.
- Biosimilar competition and new entrants could further suppress prices.
- Market share will shift from branded Otezla to generic versions, especially in mature markets.
- Opportunities remain in niche indications and emerging markets with less generic penetration.
FAQs
-
What is the primary driver of Otezla's price decline?
Patent expiry and generic entry lead to significant price reductions. -
How do biosimilars affect Otezla’s market?
Biosimilars mainly impact biologic competitors; as an oral small molecule, Otezla faces less direct biosimilar pressure but can be affected indirectly through market shifts. -
What markets are most promising for future growth?
Emerging markets with less generic penetration and expansion into new indications such as Behçet’s disease. -
Will Otezla maintain a profitable margin after patent expiry?
Margins will decrease due to price competition but can remain profitable if manufacturing and distribution efficiencies are optimized. -
How might policy changes influence future pricing?
Regulatory interventions, negotiations for drug pricing, and value-based reimbursement policies could lead to further price adjustments.
Citations
[1] Market Research Future. (2022). Global Psoriasis Treatment Market Forecast to 2027.
[2] IQVIA. (2022). Pharmaceutical Price Trends.
[3] U.S. Food and Drug Administration. (2014). FDA Approves Otezla for Psoriasis.
[4] European Medicines Agency. (2014). Otezla Marketing Authorization.
[5] EvaluatePharma. (2022). World Preview: Worldwide Pharmaceutical Market Insights.
More… ↓
