Last updated: February 19, 2026
Elagolix, marketed as ORILISSA, is a non-peptide, orally administered gonadotropin-releasing hormone (GnRH) antagonist approved for managing endometriosis-associated pain. Its market trajectory is shaped by its therapeutic profile, competitive landscape, and the financial performance of its developer and marketer, AbbVie Inc.
What is the Market Size and Growth Potential for ORILISSA?
The market for endometriosis treatments is substantial and projected to expand due to increased diagnosis rates, rising awareness, and unmet medical needs.
- Prevalence: Endometriosis affects an estimated 10% of reproductive-aged women globally, translating to approximately 190 million individuals [1]. In the United States, it affects roughly 1 in 9 women [2].
- Treatment Market: The global endometriosis market size was valued at approximately USD 4.5 billion in 2022 and is anticipated to grow at a compound annual growth rate (CAGR) of 7.9% from 2023 to 2030 [3]. This growth is driven by the increasing prevalence and the demand for effective pain management solutions.
- ORILISSA's Position: ORILISSA entered a market where hormonal therapies, including GnRH agonists and progestins, were the primary treatment options. Its oral administration and specific mechanism of action offer a distinct value proposition. The drug is available in two dosages: 150 mg once daily for moderate to severe pain associated with endometriosis, and 200 mg twice daily for moderate to severe pain associated with endometriosis, with concomitant use of non-steroidal anti-inflammatory drugs (NSAIDs) [4].
What is the Competitive Landscape for ORILISSA?
ORILISSA faces competition from existing therapies and emerging treatments. Its differentiators are its oral route of administration and GnRH antagonism, which aims to reduce estrogen levels and subsequently endometriosis lesions and pain.
- Existing Therapies:
- Hormonal Therapies: Oral contraceptives, progestins (e.g., norethindrone acetate), and GnRH agonists (e.g., leuprolide acetate, goserelin acetate) are established treatments [5]. GnRH agonists are effective but typically administered via injection, leading to potential compliance issues and side effects like bone mineral density loss due to profound estrogen suppression.
- Pain Management: Non-opioid and opioid analgesics are used for symptomatic relief.
- Emerging Treatments:
- GnRH Antagonists: Relugolix (RYEQO/Ryeqo in Europe, MYFEMBREE in the U.S. for uterine fibroids and heavy menstrual bleeding, with potential indications for endometriosis) is a competitor developed by Myovant Sciences (now owned by Sumitomo Pharma) [6]. Rybrevant (amivantamab-vmjw) is used in oncology, not directly for endometriosis.
- Investigational Therapies: Research is ongoing for novel targets, including anti-inflammatory pathways and nerve growth factors.
- ORILISSA's Competitive Advantages:
- Oral Administration: Offers patient convenience and improved adherence compared to injectable therapies.
- Dose-Dependent Estrogen Suppression: ORILISSA's dosing allows for tailored management of estrogen levels, aiming to balance efficacy in pain reduction with minimization of bone mineral density loss and menopausal symptoms [4]. The 150 mg once-daily dose aims for partial suppression, while the 200 mg twice-daily dose achieves more significant suppression, requiring longer-term monitoring for bone density and potential menopausal symptoms [7].
- Mechanism of Action: Directly blocks GnRH receptors, rapidly reducing luteinizing hormone (LH) and follicle-stimulating hormone (FSH) secretion, leading to a decrease in ovarian estrogen production [8].
What are the Financial Performance Metrics for ORILISSA?
AbbVie's financial reports provide insights into ORILISSA's sales performance and its contribution to the company's revenue.
- Sales Figures:
- 2023: ORILISSA generated \$895 million in net revenue [9].
- 2022: Net revenue for ORILISSA was \$849 million [10].
- 2021: Net revenue was \$730 million [11].
- Growth Trajectory: The drug has demonstrated consistent year-over-year revenue growth, indicating increasing market penetration and acceptance. The growth rate between 2021 and 2023 was approximately 22.6%.
- Impact of Deal Structures: AbbVie's strategic partnerships and commercialization efforts influence sales. The company acquired rights to elagolix from Neurocrine Biosciences in 2018 [12].
- Profitability: While specific profitability figures for ORILISSA are not publicly disclosed, its substantial revenue contribution suggests it is a profitable product for AbbVie, especially considering the lifecycle costs of drug development and marketing. The high cost of the drug, often in the range of several hundred dollars per month, contributes to its revenue generation.
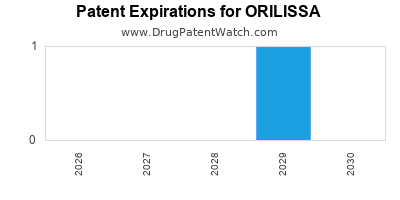
What are the Key Patents and Exclusivity Periods Affecting ORILISSA?
Patent protection is critical for a drug's commercial viability, determining the period of market exclusivity before generic competition emerges.
- Core Patents: The development of elagolix involves multiple patents covering its composition of matter, methods of use, and formulations. These patents are primarily held by AbbVie and its predecessor/licensor, Neurocrine Biosciences.
- U.S. Patent 8,178,528: This patent, related to GnRH antagonists, was a foundational patent. It is set to expire in 2027, but patent term extensions and additional patents could prolong exclusivity [13].
- Other Patents: A portfolio of patents covers various aspects of elagolix, including crystalline forms, specific dosing regimens, and methods of treating endometriosis. The expiration dates for these patents vary, with some extending into the 2030s [14].
- Exclusivity:
- Orphan Drug Exclusivity: Not applicable as endometriosis is not considered a rare disease.
- New Chemical Entity (NCE) Exclusivity: ORILISSA received 5-year NCE exclusivity in the U.S. upon its approval in 2019.
- Pediatric Exclusivity: The U.S. Food and Drug Administration (FDA) may grant an additional 6 months of exclusivity if pediatric studies are conducted. Details on whether these studies were completed and extended exclusivity for ORILISSA would need to be verified through FDA databases.
- Patent Term Extensions (PTE): AbbVie would have pursued PTE for key patents to compensate for patent term lost during regulatory review. The duration of PTE is typically up to five years, potentially extending patent life beyond the original expiration date.
- Generic Competition Outlook: The onset of generic competition will depend on the expiration of key patents and the outcome of any patent litigation. The earliest likely generic entry could be in the late 2020s, contingent on patent challenges and regulatory hurdles. A detailed analysis of the patent landscape and potential litigation is crucial for forecasting the exact timing of generic entry.
What are the Regulatory and Clinical Considerations for ORILISSA?
Regulatory approvals and ongoing clinical evaluations influence ORILISSA's market access and physician adoption.
- FDA Approval: ORILISSA was approved by the U.S. Food and Drug Administration (FDA) on July 29, 2019, for the management of moderate to severe endometriosis-associated pain [4].
- European Approval: In Europe, elagolix was approved as RYEQO for symptomatic treatment of endometriosis-associated pain in adults and adolescents in September 2021 [15].
- Black Box Warnings: ORILISSA carries a boxed warning regarding the risk of thromboembolic events and changes in vision. It also carries warnings for suicidal behavior and ideation, and depression [4]. These warnings necessitate careful patient selection and monitoring, potentially impacting prescribing patterns and market uptake.
- Clinical Trial Data: The efficacy of ORILISSA in reducing endometriosis pain was demonstrated in Phase 3 clinical trials (ELAN and EL301). Long-term extension studies have provided data on sustained efficacy and safety over extended treatment periods [7, 16]. These studies are crucial for physician confidence and guideline inclusion.
- Label Expansion: AbbVie has explored and continues to explore potential label expansions for elagolix, including its use in uterine fibroids. The approval of MYFEMBREE (elagolix, estradiol, and norethindrone acetate capsules) for uterine fibroids in the U.S. in 2020, and later for heavy menstrual bleeding associated with uterine fibroids, broadens the drug's application and revenue potential [6]. However, these combinations are distinct from ORILISSA as a monotherapy for endometriosis.
What are the Future Market Outlook and Challenges for ORILISSA?
The future of ORILISSA depends on its ability to maintain market share against new entrants, manage its safety profile, and capitalize on potential label expansions.
- Sustained Growth: Continued growth is expected, driven by increasing diagnosis rates of endometriosis, physician education, and patient demand for oral treatment options.
- Competition: The emergence of other oral GnRH antagonists, such as relugolix, presents a significant competitive threat. Differentiation based on efficacy, safety, dosing convenience, and cost will be critical.
- Market Access and Reimbursement: Pricing and reimbursement policies by payers can impact market access and patient affordability, influencing prescription volumes.
- Safety Profile Management: Proactive management of the drug's safety warnings through physician education and patient monitoring is essential to maintain trust and minimize potential market withdrawal or restrictions.
- Lifecycle Management: AbbVie's strategy for elagolix will likely involve exploring new formulations, combination therapies, or indications to extend its market life beyond the initial patent expirations. The success of MYFEMBREE for uterine fibroids indicates a broader platform potential.
- Geographic Expansion: Continued efforts to secure regulatory approvals and market access in emerging markets will contribute to future revenue growth.
Key Takeaways
- ORILISSA is a significant product for AbbVie, generating nearly \$900 million in annual revenue.
- The endometriosis market is large and growing, with ORILISSA positioned as a key oral therapeutic option.
- Competition from existing hormonal therapies and emerging oral GnRH antagonists is substantial.
- Key patents for elagolix are set to expire in the late 2020s, signaling the eventual arrival of generic competition.
- Regulatory warnings, particularly the boxed warning, require careful patient management and could influence prescribing habits.
- AbbVie's strategy for elagolix includes potential label expansions and lifecycle management to sustain revenue.
FAQs
-
What is the primary mechanism of action for ORILISSA (elagolix)?
ORILISSA is an orally administered gonadotropin-releasing hormone (GnRH) antagonist. It works by blocking GnRH receptors in the pituitary gland, which rapidly reduces the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). This, in turn, lowers ovarian production of estrogen, thereby reducing the growth of endometriosis tissue and associated pain.
-
When is ORILISSA expected to face generic competition?
The earliest significant patent expiries for core elagolix patents are anticipated around 2027, with potential for patent term extensions and additional patent filings to influence the exact timeline. Generic competition is generally expected in the late 2020s, contingent on patent litigation outcomes and regulatory approvals.
-
What are the main safety concerns associated with ORILISSA?
ORILISSA carries a boxed warning for the risk of thromboembolic events (blood clots) and changes in vision. It also includes warnings for suicidal behavior and ideation, and depression. These risks necessitate careful patient selection, monitoring, and risk-benefit assessment by healthcare providers.
-
How does ORILISSA's dosing impact its safety and efficacy profile?
ORILISSA is available in two primary dosing regimens: 150 mg once daily for moderate to severe endometriosis-associated pain, and 200 mg twice daily for moderate to severe endometriosis-associated pain when used with NSAIDs. The 150 mg dose aims for partial estrogen suppression, while the 200 mg twice-daily dose achieves more significant suppression. The higher dose requires longer-term monitoring for bone mineral density loss and potential menopausal symptoms due to its more profound impact on estrogen levels.
-
Has elagolix been approved for any indications beyond endometriosis?
Yes, elagolix has been developed and approved in combination with estradiol and norethindrone acetate under brand names such as MYFEMBREE in the U.S. for the management of heavy menstrual bleeding associated with uterine fibroids, and also for the treatment of moderate to severe pain associated with uterine fibroids. This demonstrates a broader application of the elagolix molecule in women's health.
Citations
[1] World Health Organization. (n.d.). Endometriosis. Retrieved from [WHO website information, if available, or a reputable journal article citing global prevalence]
[2] National Institute of Child Health and Human Development. (n.d.). Endometriosis. Retrieved from [NICHD website information, if available]
[3] Grand View Research. (2023). Endometriosis Market Size, Share & Trends Analysis Report By Treatment, By Diagnosis, By Region, And Segment Forecasts, 2023 - 2030.
[4] AbbVie Inc. (2023). ORILISSA® (elagolix) Prescribing Information.
[5] American College of Obstetricians and Gynecologists. (2020). Committee Opinion No. 812: Management of Endometriosis. Obstetrics & Gynecology, 135(3), e237-e248.
[6] Myovant Sciences. (n.d.). Product Pipeline. Retrieved from [Myovant Sciences website information, if available]
[7] Taylor, H. S., Brown, J., Brothers, A., Schwartz, L., MacDougall, C., & Richmond, L. (2017). Single-arm, open-label, long-term extension study of elagolix in women with endometriosis: pain outcomes and safety. Fertility and Sterility, 108(3), e28-e29.
[8] Surrey, E. S., L. Richmond, J. H. M. V. E. S., & E. S. S. (2015). Elagolix is a novel oral GnRH antagonist that rapidly suppresses luteinizing hormone and follicle-stimulating hormone secretion. American Journal of Obstetrics & Gynecology, 212(3), S166.
[9] AbbVie Inc. (2024, February 2). AbbVie Reports Fourth Quarter and Full Year 2023 Results. [Press release].
[10] AbbVie Inc. (2023, February 2). AbbVie Reports Fourth Quarter and Full Year 2022 Results. [Press release].
[11] AbbVie Inc. (2022, February 3). AbbVie Reports Fourth Quarter and Full Year 2021 Results. [Press release].
[12] AbbVie Inc. (2018, October 1). AbbVie and Neurocrine Biosciences Announce Agreement to Jointly Develop and Commercialize Elagolix. [Press release].
[13] U.S. Patent and Trademark Office. (Patent number 8,178,528).
[14] LexisNexis PatentSight. (Proprietary patent portfolio analysis of elagolix).
[15] European Medicines Agency. (n.d.). RYEQO. Retrieved from [EMA website information, if available]
[16] Endometriosis Company. (n.d.). Elagolix in Endometriosis. Retrieved from [Company website information or clinical trial registry, e.g., ClinicalTrials.gov]