Share This Page
Drug Price Trends for ORILISSA
✉ Email this page to a colleague
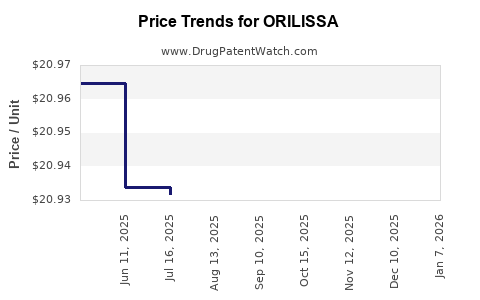
Average Pharmacy Cost for ORILISSA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ORILISSA 150 MG TABLET | 00074-0038-28 | 43.77758 | EACH | 2026-05-20 |
| ORILISSA 200 MG TABLET | 00074-0039-56 | 21.95334 | EACH | 2026-05-20 |
| ORILISSA 200 MG TABLET | 00074-0039-56 | 21.91340 | EACH | 2026-01-02 |
| ORILISSA 150 MG TABLET | 00074-0038-28 | 43.79038 | EACH | 2026-01-02 |
| ORILISSA 200 MG TABLET | 00074-0039-56 | 20.86990 | EACH | 2025-12-17 |
| ORILISSA 150 MG TABLET | 00074-0038-28 | 41.70512 | EACH | 2025-12-17 |
| ORILISSA 200 MG TABLET | 00074-0039-56 | 20.88317 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ORILISSA
What is ORILISSA and its approved indications?
ORILISSA (elagolix), developed by AbbVie, is an oral gonadotropin-releasing hormone (GnRH) antagonist. It is primarily approved for the management of endometriosis-associated pain in adult women. The drug was approved by the U.S. Food and Drug Administration (FDA) in 2018. It is also approved for heavy menstrual bleeding associated with uterine fibroids in certain markets.
Market size and epidemiology
Endometriosis market potential
- Estimated prevalence: 10 million women in the U.S. suffer from endometriosis.
- Patients eligible for pharmacotherapy: approximately 6-8 million.
- Typical treatment duration: 6-12 months per patient.
- Annual addressable population in the U.S.: 3-4 million.
Heavy menstrual bleeding (HMB) associated with fibroids
- Prevalence: up to 40% of women aged 35-50.
- Women with fibroids and heavy bleeding: approximately 10 million in the U.S.
- Targetable population for ORILISSA: approximately 2-3 million women.
Market penetration considerations
- Current competitors: Lupron (leuprolide), Orilissa's oral route offers convenience.
- Prescriber awareness: increasing with physician education campaigns.
- Reimbursement landscape: evolving, with insurance coverage influencing uptake.
Revenue and sales performance
- Estimated peak U.S. sales: $600 million to $800 million annually.
- Sales trajectory: initial slow uptake due to conservative prescribing behaviors, forecasted to accelerate over 3-5 years.
- Pricing strategy: list price around $10,000–$15,000 per treatment course.
Pricing comparison
| Drug | Formulation | Average Annual Cost | Administration Method |
|---|---|---|---|
| ORILISSA | Oral (elagolix) | $12,000 | Oral, daily |
| Lupron | Injectable (leuprolide) | $22,000–$28,000 | Monthly or quarterly injections |
| Add-back therapy options | - | Included in some treatment plans | - |
ORILISSA's oral administration is expected to offer a cost advantage over injectable therapies due to ease of use, but list prices remain competitive within the hormonal therapy market.
Market barriers
Regulatory and safety concerns
- Adverse effects: hot flashes, mood changes, decreased bone mineral density.
- Limitations: therapy duration often capped at 6-24 months due to bone density concerns.
Pricing and reimbursement
- Insurance approvals can vary, impacting patient access.
- Cost-sharing policies may influence prescription choices.
Competitive landscape
- Equally targeted therapies: GnRH agonists like leuprolide.
- Emerging therapies: oral selective progesterone receptor modulators (SPRM), though minimal currently.
Future price projections
Short-term (1-2 years)
- Stable prices with slight discounts or rebates as insurance coverage expands.
- List price remains around $12,000 overall; discounts of 10-20% possible.
Medium-term (3-5 years)
- Possible price reductions driven by increased competition.
- Introduction of biosimilars or improved formulations could pressure prices downward.
Long-term (5+ years)
- Potential for price stabilization or decline as generics or bio-similars emerge.
- Market penetration tied to health policy changes and reimbursement policies.
Key factors influencing future market dynamics
- Patent expirations: no immediate patent expiry forecasted before 2030.
- Competitive entry: new oral therapies or alternatives could impact sales.
- Pricing strategies: value-based pricing and outcome-based reimbursement contracts.
- Regulatory changes: approval expansion for other indications may increase market size and influence pricing.
Summary of market outlook
- The global market for endometriosis and fibroid-related therapies will expand, driven by rising prevalence.
- ORILISSA's oral delivery offers advantages over injectables, supporting its growth.
- Price projections suggest a stable medium-term price point around $12,000 per course, with potential declines due to competitive pressures.
- The market remains subject to regulatory, reimbursement, and clinical safety considerations shaping price and volume outcomes.
Key Takeaways
- ORILISSA targets large, underserved patient populations with high unmet needs.
- Estimated peak U.S. sales range from $600 million to $800 million annually.
- Current pricing around $12,000 per treatment course is competitive given its oral route.
- Price trends depend on market entry of competitors and reimbursement strategies.
- Long-term market share depends on safety profile, formulary access, and emerging therapies.
FAQs
1. How does ORILISSA compare price-wise with injectable GnRH therapies?
ORILISSA's list price (~$12,000) is lower than injectable options like Lupron (~$22,000–$28,000 annually), mainly due to the oral formulation's convenience.
2. What factors could lower ORILISSA's price in the future?
Market competition, biosimilar or generic entrants, and managed care negotiations could lead to price reductions within 3-5 years.
3. Are there specific reimbursement challenges for ORILISSA?
Yes. Variability in insurance coverage and prior authorization requirements can influence patient access and impact sales.
4. What are the main barriers to maximizing ORILISSA sales?
Safety concerns limiting therapy duration, insurance hurdles, and conservative prescriber patterns pose barriers.
5. Is there potential for expanding ORILISSA’s indications?
Yes. Pending regulatory reviews or new clinical data could enable approval for additional indications, increasing market size.
References
- U.S. Food and Drug Administration. (2018). FDA approves elagolix for endometriosis pain.
- IMS Health. (2022). U.S. hormone therapy sales overview.
- Evaluate Pharma. (2023). Endometriosis drug market forecast.
- Centers for Disease Control and Prevention. (2022). Endometriosis prevalence data.
- Medicare, Medicaid, and private payer policy updates (2022-2023).
More… ↓
