OFEV Drug Patent Profile
✉ Email this page to a colleague
When do Ofev patents expire, and what generic alternatives are available?
Ofev is a drug marketed by Boehringer Ingelheim and is included in one NDA. There are five patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and forty-four patent family members in fifty-three countries.
The generic ingredient in OFEV is nintedanib esylate. There are nine drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the nintedanib esylate profile page.
DrugPatentWatch® Generic Entry Outlook for Ofev
Ofev was eligible for patent challenges on October 15, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 7, 2029. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (nintedanib esylate), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
Summary for OFEV
| International Patents: | 244 |
| US Patents: | 5 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 75 |
| Patent Applications: | 102 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for OFEV |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for OFEV |
| What excipients (inactive ingredients) are in OFEV? | OFEV excipients list |
| DailyMed Link: | OFEV at DailyMed |
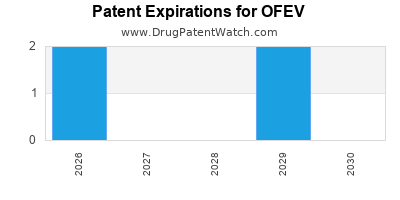

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for OFEV
Generic Entry Date for OFEV*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for OFEV
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Protein Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for OFEV
Paragraph IV (Patent) Challenges for OFEV
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OFEV | Capsules | nintedanib esylate | 100 mg and 150 mg | 205832 | 4 | 2018-10-15 |
US Patents and Regulatory Information for OFEV
OFEV is protected by five US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of OFEV is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting OFEV
Pharmaceutical dosage form for immediate release of an indolinone derivative
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Medicaments for the treatment or prevention of fibrotic diseases
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Substituted indolines which inhibit receptor tyrosine kinases
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
3-Z-[1-(4-(N-((4-Methyl-piperazin-1-yl)-methylcarbonyl)-N-methyl-amino)-an- ilino)-1-phenyl-methylene]-6-methoxycarbonyl-2-indolinone-monoethanesulpho- nate and the use thereof as a pharmaceutical composition
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting OFEV
INDICATED TO SLOW THE RATE OF DECLINE IN PULMONARY FUNCTION IN PATIENTS WITH SYSTEMIC SCLEROSIS-ASSOCIATED INTERSTITIAL LUNG DISEASE (SSC-ILD)
Exclusivity Expiration: ⤷ Try a Trial
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OFEV
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-002 | Oct 15, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-002 | Oct 15, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for OFEV
When does loss-of-exclusivity occur for OFEV?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2059
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 09254548
Estimated Expiration: ⤷ Try a Trial
Patent: 15227503
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0913434
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 26267
Estimated Expiration: ⤷ Try a Trial
Chile
Patent: 10001279
Estimated Expiration: ⤷ Try a Trial
China
Patent: 2056598
Estimated Expiration: ⤷ Try a Trial
Patent: 5193720
Estimated Expiration: ⤷ Try a Trial
Colombia
Patent: 80467
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0180709
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 20533
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
Ecuador
Patent: 10010660
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 9996
Estimated Expiration: ⤷ Try a Trial
Patent: 1001856
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 39187
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 8954
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 61031
Estimated Expiration: ⤷ Try a Trial
Patent: 05542
Estimated Expiration: ⤷ Try a Trial
Patent: 11522812
Estimated Expiration: ⤷ Try a Trial
Patent: 14208712
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 8930
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 9229
Estimated Expiration: ⤷ Try a Trial
Patent: 10013203
Estimated Expiration: ⤷ Try a Trial
Morocco
Patent: 385
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 3162
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 100254
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 142
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 99987
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 1007636
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1725469
Estimated Expiration: ⤷ Try a Trial
Patent: 110017872
Estimated Expiration: ⤷ Try a Trial
Patent: 170020557
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 69469
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 1002691
Estimated Expiration: ⤷ Try a Trial
Tunisia
Patent: 10000558
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 4590
Estimated Expiration: ⤷ Try a Trial
Uruguay
Patent: 879
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering OFEV around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Colombia | 6280467 | FORMA DE DOSIFICACION FARMACEUTICA EN CAPSULA QUE COMPRENDE UNA FORMULACION EN SUSPENSION DE UN DERIVADO DE INDOLINONA | ⤷ Try a Trial |
| Taiwan | 200635582 | Medicaments for the treatment or prevention of fibrotic diseases | ⤷ Try a Trial |
| Eurasian Patent Organization | 200200380 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for OFEV
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1830843 | PA2015025,C1830843 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: NINTEDANIBAS ARBA JO TAUTOMERAS, ARBA JU MISINIAI, ARBA JO DRUSKA; KONKRECIAI NINTEDANIBO ESILATAS; REGISTRATION NO/DATE: EU/1/14/979/001 - EU/1/14/979/004 20150115 |
| 1830843 | 1590034-3 | Sweden | ⤷ Try a Trial | PRODUCT NAME: NINTEDANIB, OR A TAUTOMER, THE MIXTURES THEREOF ORA SALT THEROF; IN PARTICULAR NINTEDANIB ESILATE; REG. NO/DATE: EU/1/14/979 20150119 |
| 1224170 | 15C0024 | France | ⤷ Try a Trial | PRODUCT NAME: NINTEDANIB,SES TAUTOMERES ET SES SELS,EN PARTICULIER LES SELS PHYSIOLOGIQUEMENT ACCEPTABLES DE NINTEDANIB ET PLUS PARTICULIEREMENT L'ESILATE; REGISTRATION NO/DATE: EU/1/14/954 20141125 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


