OFEV Drug Patent Profile
✉ Email this page to a colleague
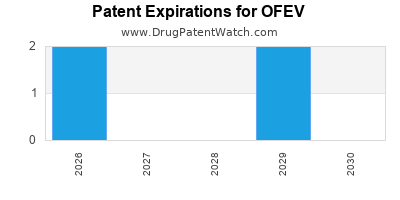
When do Ofev patents expire, and when can generic versions of Ofev launch?
Ofev is a drug marketed by Boehringer Ingelheim and is included in one NDA. There are four patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and ninety-seven patent family members in fifty-three countries.
The generic ingredient in OFEV is nintedanib esylate. There are nine drug master file entries for this compound. Eight suppliers are listed for this compound. Additional details are available on the nintedanib esylate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Ofev
A generic version of OFEV was approved as nintedanib esylate by APOTEX on April 2nd, 2026.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for OFEV?
- What are the global sales for OFEV?
- What is Average Wholesale Price for OFEV?
Summary for OFEV
| International Patents: | 197 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 76 |
| Patent Applications: | 360 |
| Drug Prices: | Drug price information for OFEV |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for OFEV |
| What excipients (inactive ingredients) are in OFEV? | OFEV excipients list |
| DailyMed Link: | OFEV at DailyMed |
Pharmacology for OFEV
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Protein Kinase Inhibitors |
Paragraph IV (Patent) Challenges for OFEV
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OFEV | Capsules | nintedanib esylate | 100 mg and 150 mg | 205832 | 4 | 2018-10-15 |
US Patents and Regulatory Information for OFEV
OFEV is protected by four US patents and two FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-002 | Oct 15, 2014 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-002 | Oct 15, 2014 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OFEV
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-002 | Oct 15, 2014 | ⤷ Start Trial | ⤷ Start Trial |
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | ⤷ Start Trial | ⤷ Start Trial |
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-002 | Oct 15, 2014 | ⤷ Start Trial | ⤷ Start Trial |
| Boehringer Ingelheim | OFEV | nintedanib esylate | CAPSULE;ORAL | 205832-001 | Oct 15, 2014 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for OFEV
When does loss-of-exclusivity occur for OFEV?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2059
Patent: FORMA DE DOSIFICACION FARMACEUTICA EN CAPSULA QUE COMPRENDE UNA FORMULACION EN SUSPENSION DE UN DERIVADO DE INDOLINONA
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09254548
Patent: Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative
Estimated Expiration: ⤷ Start Trial
Patent: 15227503
Patent: Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0913434
Patent: forma de dosagem farmacêutica em cápsula compreendendo uma formulação de um derivado de indolinona em supensão
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 26267
Patent: FORME POSOLOGIQUE PHARMACEUTIQUE EN CAPSULE COMPRENANT UNE FORMULATION EN SUSPENSION D'UN DERIVE D'INDOLINONE (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 10001279
Patent: Forma de dosificacion que comprende 3-z-[1-(4-(n-((4-metil-piperazin-1-il)-metilcarbonil)-n-metil-amino)-anilino)-1-fenil-metileno]-6-metoxicarbonil-2-indolinona-monoetanosulfonato, un vehiculo lipidico, un espesante y un agente de deslizamiento/solubilizante, de grupos definidos.
Estimated Expiration: ⤷ Start Trial
China
Patent: 2056598
Patent: Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative
Estimated Expiration: ⤷ Start Trial
Patent: 5193720
Patent: CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 80467
Patent: FORMA DE DOSIFICACION FARMACEUTICA EN CAPSULA QUE COMPRENDE UNA FORMULACION EN SUSPENSION DE UN DERIVADO DE INDOLINONA
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0180709
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 20533
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 99987
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 10010660
Patent: FORMA DE DOSIFICACIÓN FARMACÉUTICA EN CÁPSULA QUE COMPRENDE UNA FORMULACIÓN EN SUSPENCIÓN DE UN DERIVADO DE INDOLINONA
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 9996
Patent: КАПСУЛЯРНАЯ ЛЕКАРСТВЕННАЯ ФОРМА, СОДЕРЖАЩАЯ СУСПЕНЗИОННУЮ КОМПОЗИЦИЮ ПРОИЗВОДНОГО ИНДОЛИНОНА (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Patent: 1001856
Patent: КАПСУЛЯРНАЯ ЛЕКАРСТВЕННАЯ ФОРМА, СОДЕРЖАЩАЯ СУСПЕНЗИОННУЮ КОМПОЗИЦИЮ ПРОИЗВОДНОГО ИНДОЛИНОНА
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 99987
Patent: FORME POSOLOGIQUE PHARMACEUTIQUE EN CAPSULE COMPRENANT UNE FORMULATION EN SUSPENSION D'UN DÉRIVÉ D'INDOLINONE (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 39187
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 8954
Patent: צורת מינון רוקחית שהינה קפסולה המכילה הרכב תרחיפי של נגזרת אינדולינון (Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 61031
Estimated Expiration: ⤷ Start Trial
Patent: 05542
Estimated Expiration: ⤷ Start Trial
Patent: 11522812
Estimated Expiration: ⤷ Start Trial
Patent: 14208712
Patent: インドリノン誘導体の懸濁液製剤を含むカプセル医薬投薬形態 (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING SUSPENSION FORMULATION OF INDOLINONE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 99987
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 8930
Patent: CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 9229
Patent: FORMA DE DOSIFICACIÓN FARMACÉUTICA EN CÁPSULA QUE COMPRENDE UNA FORMULACIÓN EN SUSPENSIÓN DE UN DERIVADO DE INDOLINONA. (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE.)
Estimated Expiration: ⤷ Start Trial
Patent: 10013203
Patent: FORMA DE DOSIFICACION FARMACEUTICA EN CAPSULA QUE COMPRENDE UNA FORMULACION EN SUSPENSION DE UN DERIVADO DE INDOLINONA. (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 385
Patent: شكل جرعات صيدلانية لكبسولة تتألف من صياغة مستعلقة من مشتقة الإيندولينون
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3162
Patent: Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 99987
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 100254
Patent: FORMA DE DOSIFICACION FARMACEUTICA EN CAPSULA QUE COMPRENDE UNA FORMULACION EN SUSPENSION DE UN DERIVADO DE INDOLINONA
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 99987
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 99987
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 142
Patent: FARMACEUTSKI DOZNI OBLIK U VIDU KAPSULE KOJI SADRŽI FORMULACIJU SUSPENZIJE INDOLINON DERIVATA (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 99987
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1007636
Patent: CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1725469
Estimated Expiration: ⤷ Start Trial
Patent: 110017872
Patent: CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE
Estimated Expiration: ⤷ Start Trial
Patent: 170020557
Patent: 인돌리논 유도체의 현탁 제형을 포함하는 캡슐 약제학적 투여 형태 (Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 69469
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1002691
Patent: Capsule pharmaceutical dosage form comprising a suspension formulation of an indolinone derivative
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 10000558
Patent: CAPSULE PHARMACEUTICAL DOAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 4590
Patent: КАПСУЛИРОВАННАЯ ЛЕКАРСТВЕННАЯ ФОРМА, СОДЕРЖАЩАЯ СУСПЕНЗИОННУЮ КОМПОЗИЦИЮ ПРОИЗВОДНОЙ ИНДОЛИНОНА;КАПСУЛЬОВАНА ЛІКАРСЬКА ФОРМА, ЩО МІСТИТЬ СУСПЕНЗІЙНУ КОМПОЗИЦІЮ ПОХІДНОЇ ІНДОЛІНОНУ (CAPSULE PHARMACEUTICAL DOSAGE FORM COMPRISING A SUSPENSION FORMULATION OF AN INDOLINONE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 879
Patent: FORMA DE DOSIFICACIÓN FARMACÉUTICA EN CÁPSULA QUE COMPRENDE UNA FORMULACIÓN EN SUSPENSIÓN DE UN DERIVADO DE INDOLINONA
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering OFEV around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Montenegro | P53708 | 3-Z-[1-(4-(N-((4-METIL-PIPERAZIN-1-IL) -METILKARBONIL) -N-METIL-AMINO) -ANILINO)-1- FENIL-METILEN] -6-METOKSIKARBONIL-2 -INDOLINON- MONOETANSULFONAT I NJEGOVA PRIMJENA U FARMACEUTSKOJ KOMPOZICIJI (3-Z-[1-(4-(N-((4-METHYL-PIPERAZIN-1-YL)-METHYLCARBONYL) -N-METHYL -AMINO) -ANILINO)-1- PHENYL-METHYLENE] -6-METHOXYCARBONYL -2-INDOLINONE -MONOETHANESULPHONATE AND THE USE THEREOF AS A PHARMACEUTICAL COMPOSITION) | ⤷ Start Trial |
| Hungary | E050998 | ⤷ Start Trial | |
| Singapore | 191607 | PHARMACEUTICAL DOSAGE FORM FOR IMMEDIATE RELEASE OF AN INDOLINONE DERIVATIVE | ⤷ Start Trial |
| South Africa | 200703601 | Medicaments for the treatment or prevention of fibriotic diseases | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for OFEV
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1224170 | 15C0024 | France | ⤷ Start Trial | PRODUCT NAME: NINTEDANIB,SES TAUTOMERES ET SES SELS,EN PARTICULIER LES SELS PHYSIOLOGIQUEMENT ACCEPTABLES DE NINTEDANIB ET PLUS PARTICULIEREMENT L'ESILATE; REGISTRATION NO/DATE: EU/1/14/954 20141125 |
| 1224170 | PA2015015 | Lithuania | ⤷ Start Trial | PRODUCT NAME: NINTEDANIBUM; REGISTRATION NO/DATE: EU/1/14/954/001-004 20141121 |
| 1224170 | 122015000023 | Germany | ⤷ Start Trial | PRODUCT NAME: NINTEDANIB, DESSEN TAUTOMERE UND DESSEN SALZE, INSBESONDERE NINTEDANIB UND PHYSIOLOGISCH VERTRAEGLICHE SALZE DAVON; REGISTRATION NO/DATE: EU/1/14/954/001-004 20141121 |
| 1830843 | 2015C/038 | Belgium | ⤷ Start Trial | PRODUCT NAME: NINTEDANIB, OU UN TAUTOMERE DE CELUI-CI, DES MELANGES DE CEUX-CI OU UN SEL DE CELUI-CI, SPECIFIQUEMENTNINTEDANIB SOUS FORME D'ESILATE; AUTHORISATION NUMBER AND DATE: EU/1/14/979/001 20150119 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
OFEV: Patent Landscape and Financial Performance Analysis
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.