NEXLETOL Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Nexletol, and what generic alternatives are available?
Nexletol is a drug marketed by Esperion Theraps Inc and is included in one NDA. There are seven patents protecting this drug and one Paragraph IV challenge.
This drug has eighty-one patent family members in twenty-four countries.
The generic ingredient in NEXLETOL is bempedoic acid. One supplier is listed for this compound. Additional details are available on the bempedoic acid profile page.
DrugPatentWatch® Generic Entry Outlook for Nexletol
Nexletol was eligible for patent challenges on February 21, 2024.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 19, 2040. This may change due to patent challenges or generic licensing.
There have been five patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (bempedoic acid), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for NEXLETOL?
- What are the global sales for NEXLETOL?
- What is Average Wholesale Price for NEXLETOL?
Summary for NEXLETOL
| International Patents: | 81 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 64 |
| Patent Applications: | 393 |
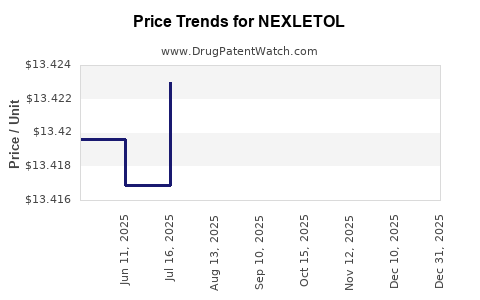
| Drug Prices: | Drug price information for NEXLETOL |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for NEXLETOL |
| What excipients (inactive ingredients) are in NEXLETOL? | NEXLETOL excipients list |
| DailyMed Link: | NEXLETOL at DailyMed |
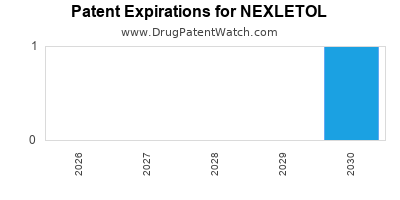
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for NEXLETOL
Generic Entry Date for NEXLETOL*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for NEXLETOL
| Drug Class | Adenosine Triphosphate-Citrate Lyase Inhibitor |
| Mechanism of Action | Adenosine Triphosphate-Citrate Lyase Inhibitors |
Paragraph IV (Patent) Challenges for NEXLETOL
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| NEXLETOL | Tablets | bempedoic acid | 180 mg | 211616 | 9 | 2024-02-21 |
US Patents and Regulatory Information for NEXLETOL
NEXLETOL is protected by seven US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of NEXLETOL is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for NEXLETOL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| Esperion Theraps Inc | NEXLETOL | bempedoic acid | TABLET;ORAL | 211616-001 | Feb 21, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for NEXLETOL
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Daiichi Sankyo Europe GmbH | Nilemdo | bempedoic acid | EMEA/H/C/004958Nilemdo is indicated in adults with primary hypercholesterolaemia (heterozygous familial and non familial) or mixed dyslipidaemia, as an adjunct to diet:in combination with a statin or statin with other lipid-lowering therapies in patients unable to reach LDL C goals with the maximum tolerated dose of a statin (see sections 4.2, 4.3, and 4.4) or,alone or in combination with other lipid-lowering therapies in patients who are statin intolerant, or for whom a statin is contraindicated. | Authorised | no | no | no | 2020-04-01 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for NEXLETOL
When does loss-of-exclusivity occur for NEXLETOL?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 20295503
Estimated Expiration: ⤷ Start Trial
Patent: 20296094
Estimated Expiration: ⤷ Start Trial
Patent: 25203232
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2021025928
Estimated Expiration: ⤷ Start Trial
Patent: 2021025964
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 44371
Estimated Expiration: ⤷ Start Trial
Patent: 44372
Estimated Expiration: ⤷ Start Trial
Patent: 43601
Estimated Expiration: ⤷ Start Trial
China
Patent: 2437765
Estimated Expiration: ⤷ Start Trial
Patent: 2437766
Patent: 制备贝派地酸及其组合物的方法 (METHODS OF MAKING BEMPEDOIC ACID AND COMPOSITIONS OF THE SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 5429784
Patent: 制备贝派地酸及其组合物的方法 (Method for preparing bepiridic acid and composition thereof)
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 86859
Patent: PROCÉDÉS DE PRÉPARATION D'ACIDE BEMPÉDOÏQUE ET COMPOSITIONS DE CELUI-CI (METHODS OF MAKING BEMPEDOIC ACID AND COMPOSITIONS OF THE SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 86860
Patent: FORMES SALINES D'ACIDE BEMPEDOÏQUE ET LEURS PROCÉDÉS D'UTILISATION (SALT FORMS OF BEMPEDOIC ACID AND METHODS FOR USING THE SAME)
Estimated Expiration: ⤷ Start Trial
Patent: 38114
Patent: PROCÉDÉS DE FABRICATION D'ACIDE BEMPÉDOÏQUE ET COMPOSITIONS ASSOCIÉES (METHODS OF MAKING BEMPEDOIC ACID AND COMPOSITIONS OF THE SAME)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 72061
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 8909
Patent: שיטות להכנה של חומצה במפדואית ותכשירים המכילים אותה (Methods of making bempedoic acid and compositions of the same)
Estimated Expiration: ⤷ Start Trial
Patent: 8997
Patent: צורות מלח של חומצה במפדואית ושיטות לשימוש בהם (Salt forms of bempedoic acid and methods for using the same)
Estimated Expiration: ⤷ Start Trial
Patent: 4076
Patent: שיטות להכנה של חומצה במפדואית ותכשירים המכילים אותה (Methods of making bempedoic acid and compositions of the same)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 22536979
Patent: 塩形態のベンペド酸及びそれを使用する方法
Estimated Expiration: ⤷ Start Trial
Patent: 22537049
Patent: ベンペド酸の製造方法及びその組成物
Estimated Expiration: ⤷ Start Trial
Patent: 25032197
Patent: ベンペド酸を含む製薬材料、医薬製剤及び医薬組成物 (PHARMACEUTICAL MATERIAL, MEDICINAL PREPARATION AND PHARMACEUTICAL COMPOSITION THAT CONTAIN BEMPEDOIC ACID)
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 21015935
Patent: FORMAS DE SAL DE ACIDO BEMPEDOICO Y METODOS PARA UTILIZAR EL MISMO. (SALT FORMS OF BEMPEDOIC ACID AND METHODS FOR USING THE SAME.)
Estimated Expiration: ⤷ Start Trial
Patent: 21015936
Patent: METODOS PARA PREPARAR ACIDO BEMPEDOICO Y COMPOSICIONES DEL MISMO. (METHODS OF MAKING BEMPEDOIC ACID AND COMPOSITIONS OF THE SAME.)
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 220024815
Patent: 벰페도산의 염 형태 및 그의 사용 방법
Estimated Expiration: ⤷ Start Trial
Patent: 220024816
Patent: 벰페도산 및 그의 조성물을 제조하는 방법
Estimated Expiration: ⤷ Start Trial
Patent: 250143358
Patent: 벰페도산 및 그의 조성물을 제조하는 방법 (METHODS OF MAKING BEMPEDOIC ACID AND COMPOSITIONS OF THE SAME)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 32779
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering NEXLETOL around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 112437766 | 制备贝派地酸及其组合物的方法 (METHODS OF MAKING BEMPEDOIC ACID AND COMPOSITIONS OF THE SAME) | ⤷ Start Trial |
| China | 112437765 | ⤷ Start Trial | |
| Brazil | 0318046 | Compostos de hidroxila e composições para controle de colesterol e empregos relacionados | ⤷ Start Trial |
| Mexico | 2017011499 | ⤷ Start Trial | |
| Mexico | 2021015935 | FORMAS DE SAL DE ACIDO BEMPEDOICO Y METODOS PARA UTILIZAR EL MISMO. (SALT FORMS OF BEMPEDOIC ACID AND METHODS FOR USING THE SAME.) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for NEXLETOL
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2404890 | 122020000048 | Germany | ⤷ Start Trial | PRODUCT NAME: BEMPEDOINSAEURE ODER EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ, HYDRAT, SOLVAT ODER EIN GEMISCH DAVON; REGISTRATION NO/DATE: EU/1/20/1424 20200327 |
| 2404890 | 132020000000112 | Italy | ⤷ Start Trial | PRODUCT NAME: ACIDO BEMPEDOICO, O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE, IDRATO, SOLVATO, O LORO MISCELA(NUSTENDI); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/20/1424, 20200331 |
| 2404890 | C02404890/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: BEMPEDOINSAEURE; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 67583 14.12.2020 |
| 2404890 | 301062 | Netherlands | ⤷ Start Trial | PRODUCT NAME: BEMPEDONZUUR, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT, HYDRAAT, SOLVAAT OF MENGSEL DAARVAN; REGISTRATION NO/DATE: EU/1/20/1424 20200331 |
| 2404890 | 640 | Finland | ⤷ Start Trial | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for NEXLETOL
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.