NAYZILAM Drug Patent Profile
✉ Email this page to a colleague
When do Nayzilam patents expire, and when can generic versions of Nayzilam launch?
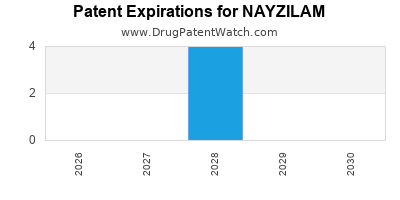
Nayzilam is a drug marketed by Ucb Inc and is included in one NDA. There are four patents protecting this drug and one Paragraph IV challenge.
The generic ingredient in NAYZILAM is midazolam. There are nine drug master file entries for this compound. Seven suppliers are listed for this compound. Additional details are available on the midazolam profile page.
DrugPatentWatch® Generic Entry Outlook for Nayzilam
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are two tentative approvals for the generic drug (midazolam), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for NAYZILAM?
- What are the global sales for NAYZILAM?
- What is Average Wholesale Price for NAYZILAM?
Summary for NAYZILAM
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 48 |
| Patent Applications: | 4,544 |
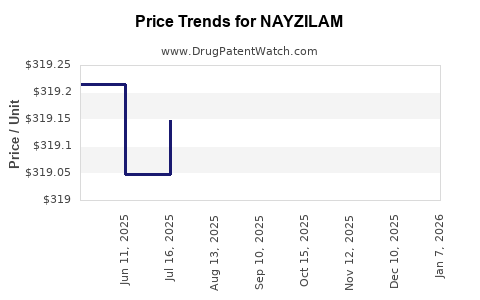
| Drug Prices: | Drug price information for NAYZILAM |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for NAYZILAM |
| What excipients (inactive ingredients) are in NAYZILAM? | NAYZILAM excipients list |
| DailyMed Link: | NAYZILAM at DailyMed |
Pharmacology for NAYZILAM
| Drug Class | Benzodiazepine |
US Patents and Regulatory Information for NAYZILAM
NAYZILAM is protected by four US patents and one FDA Regulatory Exclusivity.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ucb Inc | NAYZILAM | midazolam | SPRAY;NASAL | 211321-001 | May 17, 2019 | RX | Yes | Yes | 9,687,495 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Ucb Inc | NAYZILAM | midazolam | SPRAY;NASAL | 211321-001 | May 17, 2019 | RX | Yes | Yes | 8,809,322 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Ucb Inc | NAYZILAM | midazolam | SPRAY;NASAL | 211321-001 | May 17, 2019 | RX | Yes | Yes | 9,289,432 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Ucb Inc | NAYZILAM | midazolam | SPRAY;NASAL | 211321-001 | May 17, 2019 | RX | Yes | Yes | 8,217,033 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Ucb Inc | NAYZILAM | midazolam | SPRAY;NASAL | 211321-001 | May 17, 2019 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for NAYZILAM
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Neuraxpharm Pharmaceuticals S.L. | Buccolam | midazolam | EMEA/H/C/002267Treatment of prolonged, acute, convulsive seizures in infants, toddlers, children and adolescents (from three months to less than 18 years).Buccolam must only be used by parents / carers where the patient has been diagnosed to have epilepsy.For infants between three and six months of age, treatment should be in a hospital setting where monitoring is possible and resuscitation equipment is available. | Authorised | no | no | no | 2011-09-04 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for NAYZILAM
More… ↓