Last updated: February 19, 2026
Mupirocin, a topical antibiotic, demonstrates a stable yet evolving market presence driven by its established efficacy against gram-positive bacterial infections, primarily Staphylococcus aureus. Patent expirations have led to generic competition, impacting originator pricing but expanding accessibility. The market is segmented by indication and formulation, with nasal and dermatological applications dominating. Growth is supported by increasing prevalence of skin infections and the need for effective treatments against methicillin-resistant Staphylococcus aureus (MRSA).
What is the Current Market Size and Projected Growth for Mupirocin?
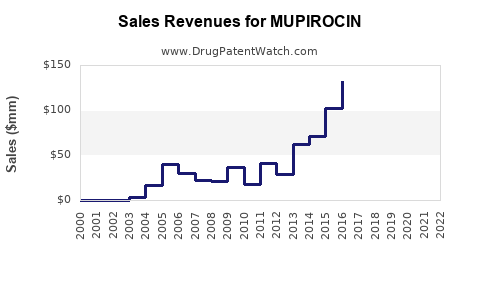
The global mupirocin market was valued at approximately $650 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.0% over the next five years, reaching an estimated $800 million by 2028. This growth is underpinned by persistent demand for topical antimicrobials and emerging applications.
Key market drivers include:
- Prevalence of Skin Infections: Bacterial skin infections, such as impetigo and cellulitis, remain common, requiring effective topical treatments.
- MRSA Activity: Mupirocin's efficacy against MRSA infections, particularly in nasal decolonization protocols, supports its continued use despite the emergence of resistance in some strains [1].
- Generic Availability: The expiration of key patents has facilitated the entry of generic mupirocin, reducing treatment costs and increasing market penetration in various regions.
- Formulation Development: While primarily topical, research into novel delivery systems or combination therapies could potentially expand its utility.
Factors potentially moderating growth include:
- Antibiotic Resistance: Increasing bacterial resistance to mupirocin could limit its long-term effectiveness in certain settings.
- Competition: Development of newer topical antibiotics or alternative treatment modalities presents competitive pressure.
- Regulatory Scrutiny: As with all antibiotics, evolving regulatory landscapes and stewardship initiatives influence prescribing patterns.
What are the Key Indications for Mupirocin Use?
Mupirocin is primarily prescribed for the treatment of bacterial skin infections. Its spectrum of activity is narrow, focusing on gram-positive bacteria.
Major indications include:
- Impetigo: A highly contagious superficial skin infection, often caused by Staphylococcus aureus or Streptococcus pyogenes. Mupirocin ointment is a first-line treatment for non-bullous impetigo.
- Secondary Skin Infections: Treatment of bacterial infections in traumatic skin lesions such as surgical wounds, burns, and abrasions.
- Folliculitis: Inflammation of hair follicles, often bacterial in origin.
- Nasal MRSA Decolonization: A significant off-label use of intranasal mupirocin to eradicate nasal carriage of MRSA in healthcare workers and patients, reducing the risk of transmission and subsequent infection [2].
- Ecthyma: A deeper form of impetigo that penetrates into the dermis.
What is the Patent Landscape for Mupirocin?
The original patent for mupirocin (US Patent 3,883,642) expired in the early 2000s. This expiration allowed for widespread generic manufacturing and distribution.
- Ex-US Patent 3,883,642: Filed in 1973, expired in the early 2000s. This patent covered the compound mupirocin and its initial uses.
- Subsequent Patents: While the core compound is off-patent, secondary patents may exist related to specific formulations (e.g., novel excipients, delivery systems), manufacturing processes, or new indications. However, these are less likely to impact the broad market for standard formulations.
- Generic Entry: The absence of strong, foundational patent protection for the active pharmaceutical ingredient has resulted in a competitive generic market for mupirocin ointment, cream, and nasal formulations.
What is the Competitive Landscape for Mupirocin?
The competitive landscape is characterized by a significant number of generic manufacturers offering mupirocin products. The market for topical antibiotics is crowded, with both broad-spectrum and narrow-spectrum agents vying for clinical use.
Leading generic manufacturers include:
- Teva Pharmaceuticals: A major global producer of generic drugs, offering mupirocin formulations.
- Viatris (formerly Mylan): Another significant player in the generic pharmaceutical market, providing mupirocin products.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company with a strong presence in generics.
- Dr. Reddy's Laboratories: Another major Indian pharmaceutical company with a broad generic portfolio.
- Aurobindo Pharma: A rapidly growing generic and API manufacturer.
Key competitive factors include:
- Price: Generic competition drives significant price sensitivity. Manufacturers compete on cost-effectiveness.
- Availability and Distribution: Wide distribution networks are critical for market penetration.
- Product Quality and Consistency: While generics, manufacturers must meet bioequivalence standards.
- Regulatory Approvals: Obtaining and maintaining regulatory approvals in key markets (e.g., FDA, EMA) is essential.
Mupirocin competes with other topical antibiotics such as:
- Neomycin/Polymyxin B/Bacitracin (Triple Antibiotic Ointment): Broad-spectrum, commonly used for minor cuts and scrapes.
- Clindamycin Topical: Used for acne and certain skin infections.
- Retapamulin: A newer topical antibiotic with activity against gram-positive bacteria, including MRSA.
- Fusidic Acid: Widely used in some international markets for staphylococcal infections.
What are the Financial Implications of Genericization for Mupirocin?
The genericization of mupirocin has profoundly impacted its financial trajectory, shifting revenue generation from originators to a multitude of generic manufacturers.
- Price Erosion: Following patent expiration, the average selling price (ASP) for mupirocin formulations has decreased by an estimated 70-85% compared to originator pricing at the time of patent expiry.
- Volume Expansion: Lower prices have increased accessibility and usage, leading to a higher volume of units sold globally. The overall market value growth is thus driven by volume rather than price increases.
- Profit Margins: For generic manufacturers, profit margins on mupirocin are typically lower compared to branded products, emphasizing efficiency in manufacturing and distribution. Margins are often in the range of 15-25% for established generic products.
- Market Share Stability: Despite price competition, mupirocin has maintained a stable market share in its primary indications due to its proven efficacy and favorable safety profile for topical use.
- Manufacturing Costs: Generic manufacturers focus on optimizing production costs, often through large-scale, integrated manufacturing facilities and efficient supply chain management. The cost of goods sold (COGS) for generic mupirocin can be as low as 20-30% of the selling price.
- Investment Focus: Investment for companies in this space has shifted from novel drug discovery to optimizing manufacturing processes, expanding product portfolios with bioequivalent generics, and securing broad market access through distribution agreements.
What is the Regulatory Status and Future Outlook for Mupirocin?
Mupirocin is approved for topical use in major pharmaceutical markets, including the United States, European Union, and various Asian countries. Regulatory oversight primarily focuses on ensuring product quality, safety, and efficacy through bioequivalence studies for generic approvals.
- FDA Approval: Mupirocin is approved by the U.S. Food and Drug Administration (FDA) for topical treatment of bacterial skin infections. Generic versions require an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence.
- EMA Approval: In the European Union, mupirocin is available through national authorizations and has been reviewed by the European Medicines Agency (EMA) for specific indications.
- WHO Model List: Mupirocin is included in the World Health Organization's (WHO) Model List of Essential Medicines, highlighting its importance in global healthcare [3].
- Antimicrobial Stewardship: The increasing global focus on antimicrobial stewardship could lead to more judicious prescribing of all antibiotics, including mupirocin. However, its topical nature and specific indications for MRSA decolonization are generally considered less prone to systemic resistance development compared to oral or intravenous antibiotics.
- Emerging Resistance: Ongoing monitoring for mupirocin resistance is crucial. While resistance is present, its prevalence and clinical impact are generally lower for topical applications compared to systemic use.
- Future Trends: The future outlook for mupirocin remains stable. Its established role in treating common skin infections and its utility in MRSA decolonization ensure continued demand. Growth will likely be incremental, driven by population increases and the persistent need for effective topical antimicrobials. Research into novel topical formulations or combination therapies could offer avenues for future market expansion, but these would likely require new patentable intellectual property.
Table 1: Mupirocin Market Segmentation (Estimated 2023)
| Segment |
Estimated Market Share |
Key Applications |
| By Application |
|
|
| Dermatological |
70% |
Impetigo, Folliculitis, Secondary Skin Infections, Infected Eczema |
| Nasal |
25% |
MRSA Nasal Decolonization (Off-label in some regions, standard in others) |
| Other Topical |
5% |
Wound care, minor burns, abrasions |
| By Formulation |
|
|
| Ointment |
60% |
Higher occlusion, suitable for dry lesions |
| Cream |
30% |
Lighter feel, suitable for moist or weeping lesions |
| Other |
10% |
Solutions, sprays (less common for mupirocin) |
Source: Industry analysis and market research reports.
Table 2: Mupirocin vs. Competitor Topical Antibiotics (Key Differentiators)
| Antibiotic |
Spectrum of Activity |
Primary Indications |
Key Differentiators/Considerations |
| Mupirocin |
Gram-positive (Staph., Strep.) |
Impetigo, Secondary Skin Infections, MRSA Nasal Decolon. |
Narrow spectrum, effective against MRSA, good safety profile topically, patent expired. |
| Retapamulin |
Gram-positive (including MRSA, MSSA) |
Impetigo |
Newer agent, broader gram-positive coverage than mupirocin, potentially higher cost, patent protected. |
| Clindamycin (Topical) |
Gram-positive (Strep., Staph.), Anaerobes |
Acne, Folliculitis, some Skin/Soft Tissue Infections |
Broader spectrum than mupirocin, can be used for acne, risk of C. difficile colitis with systemic use but lower risk topically. |
| Triple Antibiotic (NeoSporin) |
Broad-spectrum (Gram-positive & negative) |
Minor cuts, scrapes, burns |
Very broad coverage, commonly available OTC, potential for hypersensitivity reactions, lower efficacy against MRSA. |
| Fusidic Acid |
Gram-positive (Staph., Strep.) |
Impetigo, Folliculitis, Wound Infections |
Strong activity against S. aureus, widely used internationally, some resistance observed. |
Source: Clinical literature and product compendia.
Key Takeaways
- The mupirocin market is a mature, genericized segment with stable growth driven by established efficacy in treating bacterial skin infections and MRSA nasal carriage.
- Original patent expirations have led to significant price erosion, with growth now primarily volume-driven.
- Key market players are generic manufacturers focused on cost-efficient production and broad distribution.
- Mupirocin's position is secured by its inclusion on essential medicine lists and its specific utility against MRSA, although ongoing vigilance regarding resistance is necessary.
Frequently Asked Questions
-
What is the primary therapeutic advantage of mupirocin over other topical antibiotics?
Mupirocin's advantage lies in its potent activity against Staphylococcus aureus, including many strains of MRSA, and its narrow spectrum of activity, which theoretically reduces the risk of broad resistance development. Its efficacy in nasal decolonization protocols is a significant niche.
-
How has the expiration of mupirocin's patents impacted investment strategies for pharmaceutical companies?
For originator companies, the impact was a loss of exclusivity and revenue. For generic companies and investors, it opened opportunities for market entry and volume-based sales, shifting investment towards manufacturing optimization and market access rather than R&D for the core compound.
-
What are the main concerns regarding antibiotic resistance in the context of mupirocin?
The primary concern is the emergence of high-level mupirocin resistance (HLMR) in Staphylococcus aureus, which can compromise treatment efficacy. While less prevalent than resistance to systemic antibiotics, it necessitates monitoring and judicious use.
-
Beyond its current indications, are there any emerging therapeutic areas for mupirocin?
While the core indications remain dominant, research occasionally explores mupirocin's potential in combination therapies or for specific wound infections. However, significant new indications are not currently driving market expansion.
-
What is the typical profit margin for a generic mupirocin product compared to a branded topical antibiotic?
Generic mupirocin products typically have lower profit margins, estimated between 15-25%, compared to branded topical antibiotics which might achieve 40-60% or higher margins, especially during their patent-protected period. This is due to intense price competition in the generic market.
Citations
[1] Dryden, M. S. (2008). Mupirocin—the past, the present and the future. International Journal of Antimicrobial Agents, 32(Suppl 2), S69-S74.
[2] Leopold, S. S., & Schoper, J. B. (2000). Mupirocin. Infectious Disease Clinics of North America, 14(2), 337-347.
[3] World Health Organization. (2021). World Health Organization Model List of Essential Medicines. Retrieved from https://www.who.int/publications/i/item/WHO-MVP-MCM-MPP-2021.01