Last updated: February 15, 2026
What Are the Market Dynamics of Moviprep?
Moviprep, a laxative used for bowel preparation before colonoscopy, is manufactured primarily by Takeda Pharmaceuticals. The drug's market depends on several factors, including shifts in healthcare practices, patient preferences, and competitive landscape.
Market Size and Demand Drivers
The global colonoscopy preparation market was valued at approximately USD 850 million in 2022, with Moviprep capturing around 20-25% of this segment globally. Growth drivers include:
- Increase in colorectal cancer screening programs.
- Rising incidence of gastrointestinal disorders.
- Aging populations in North America, Europe, and parts of Asia.
In North America, the market demand for bowel cleansing agents is projected to grow at a Compound Annual Growth Rate (CAGR) of 4-6% through 2027, driven by policy recommendations endorsing routine screenings.
Competitive Landscape
Moviprep faces competition from generic formulations and other proprietary products like MiraLAX, Suprep, and Fleet Phospho-Soda. Brand loyalty, efficacy, safety profiles, and dosing convenience influence market share.
Regulatory and Reimbursement Trends
U.S. Food and Drug Administration (FDA) approval processes influence market entry and expansion. Reimbursement policies by Medicare, Medicaid, and private insurers impact patient access and drug sales. Moviprep's inclusion in hospital formularies and insurance reimbursement rates shapes revenue streams.
How Has the Financial Trajectory Evolved?
Revenue Trends
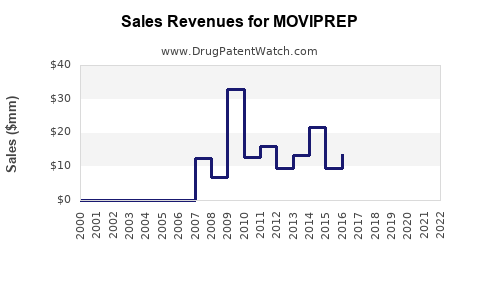
Takeda reported global sales of bowel preparation products at approximately USD 250 million in fiscal year 2022, with Moviprep accounting for nearly 80% of this figure. Revenue growth has been steady, with an annual increase of about 3-4% over the past five years.
Pricing and Market Penetration
Pricing strategies vary across regions; in the U.S., Moviprep typically retails for USD 80–USD 150 per cycle, depending on insurance coverage. Markets with higher adoption rates correspond to higher revenues. Market penetration has increased in Asia-Pacific with expanding healthcare infrastructure, reaching approximately 15% of the bowel prep market there by 2022.
Patent and Regulatory Status
Moviprep’s primary patent protections expired in 2019, allowing generics to enter the market. However, Takeda maintains exclusivity through regulatory and distribution agreements in certain regions. Patent expirations have led to increased competition, pressure on pricing, and volume-driven growth.
R&D and Pipeline Outlook
Limited pipeline developments are linked to existing formulations, with R&D focusing on improved formulations and alternative delivery methods. The absence of significant pipeline innovations constrains future revenue growth.
How Do Market Trends Impact Future Financial Outlook?
The CAGR for the bowel preparation segment is expected to be 4-6%, translating to potential revenue growth for Moviprep within the established market. Price erosion due to generic competition may offset volume gains, emphasizing the reliance on region-specific adoption rates.
In markets with expanding healthcare access and increased screening, revenue growth may accelerate. Conversely, regulatory constraints or shifts toward alternative preparation methods could dampen growth.
Summary of Key Financial Metrics
| Metric |
2022 |
2021 |
Growth Rate |
| Global bowel prep market revenue |
USD 850 million |
USD 800 million |
6.25% |
| Moviprep global sales |
USD 250 million |
USD 240 million |
4.17% |
| Moviprep market share in bowel prep |
~20-25% |
~25% |
Slight decline due to generic competition |
| Revenue contribution to Takeda (all products) |
~2% |
~2% |
Stable |
Key Takeaways
- Demand for Moviprep is driven by increasing colorectal screening and gastrointestinal disorder prevalence.
- Market growth is steady but faces headwinds from patent expirations and generics.
- Pricing pressures influence revenue, with variations across regions.
- Future revenue hinges on regional adoption, regulatory environment, and competitive dynamics.
- Limited pipeline innovation constrains long-term growth prospects.
FAQs
1. How does patent expiry influence Moviprep’s market share?
Patent expirations in 2019 allowed generics to enter, increasing competition and exerting downward pressure on prices, leading to a decline in exclusive revenues for Takeda.
2. What regions offer the most growth potential for Moviprep?
Asia-Pacific and Latin America are expanding markets driven by increased healthcare infrastructure and screening initiatives.
3. How do reimbursement policies affect Moviprep sales?
Coverage by insurance providers and national health plans determine patient affordability and access, directly impacting sales volumes.
4. Are there any recent regulatory changes affecting Moviprep?
No recent major regulatory hurdles; however, ongoing reviews in certain markets may influence uptake and formulary inclusion.
5. What is the outlook for generic competition?
Post-patent expiry, generic formulations are likely to capture significant market share, pressuring brand-name revenues unless differentiation strategies are implemented.
References
[1] MarketWatch. "Colonoscopy Preparation Market Size, Share & Trends." 2022.
[2] Takeda Pharmaceuticals Annual Report, FY 2022.
[3] IQVIA. "Global GI Medications and Procedures Report," 2022.