Last updated: February 19, 2026
What is Salix Pharmaceuticals’ current market position?
Salix Pharmaceuticals operates primarily in the gastrointestinal (GI) disorder segment, specializing in prescription drugs for constipation, diarrhea, and other gut-related conditions. In 2022, Salix generated approximately $1.7 billion in net revenue, making it a leading player in the GI therapeutics market, which was valued at $17.5 billion globally in 2022 (Grand View Research, 2022). The company holds about 8-10% market share in the prescription GI drugs segment.
Salix’s growth stems from its focus on niche, high-margin products like Xifaxan (rifaximin), a treatment for irritable bowel syndrome with diarrhea (IBS-D) and traveler’s diarrhea. Xifaxan accounts for about 70% of total revenue. While dominated by several large pharmaceutical companies, its specialty positioning allows Salix to sustain a competitive edge in its core markets.
How does Salix’s portfolio compare to competitors?
| Company |
Key Products |
Market Focus |
Estimated Revenue (2022) |
Market Share in GI Segment |
| Salix Pharmaceuticals |
Xifaxan, Relistor, Apriso |
Gastrointestinal, IBS, hepatic encephalopathy |
$1.7 billion |
8-10% |
| Pfizer (merged with BioDelivery) |
Viberzi, Zenvia |
Functional bowel disorders, diarrhea |
$850 million |
4-6% |
| Allergan (part of AbbVie) |
Linzess (under its broader portfolio) |
Chronic constipation, IBS-C |
$2.3 billion |
13% |
| Ferring Pharmaceuticals |
Dimpax, Ferring GI products |
Gastroenterology |
$900 million |
5-7% |
Salix’s key differentiator remains the efficacy and oral administration of Xifaxan, alongside its suitability for specific GI conditions. Its reliance on fewer blockbuster products enhances its revenue stability but also exposes it to risks if patents face challenges.
What are Salix’s core strengths?
- Product Specialization: Focus on GI disorders yields deep expertise, enabling targeted R&D and clinical development.
- Xifaxan Portfolio: The dominant product, with multiple indications, supports sustained revenue streams and global licensing deals.
- Market Penetration: Strong presence in the U.S. with coast-to-coast distribution channels and partnerships with major healthcare providers.
- Regulatory Approvals: Multiple FDA approvals for Xifaxan and other drugs allow diverse indications, reducing vulnerability to market shifts.
What strategic opportunities exist for Salix?
- Expansion of Indications: Developing new uses for Xifaxan, such as hepatic encephalopathy, to broaden revenue sources.
- International Growth: Increasing market footprint in Europe and Asia, where GI disorder treatments are underpenetrated.
- Biosimilars and Generics: Exploiting patent expirations for Xifaxan if applicable, or introducing biosimilar options.
- Partnerships & Licensing: Engaging with biotech firms to co-develop new therapies specifically for niche GI sub-indications.
What threats could influence Salix’s market position?
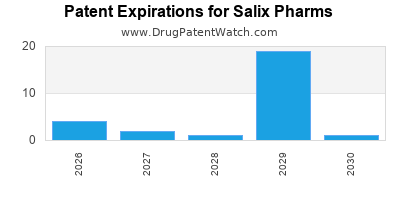
- Patent Challenges: The Xifaxan patent expired in 2029, and generic competition could erode margins.
- Market Consolidation: Larger pharmaceutical companies may acquire or outcompete Salix via increased R&D activity.
- Regulatory Hurdles: Lengthy approval processes for new indications or formulations can slow growth.
- Pricing Pressures: Increasing scrutiny on drug prices in the U.S. can compress profit margins.
How are competitors positioning themselves?
- AbbVie’s Linzess: With sales of $2.3 billion in 2022, AbbVie's strategy emphasizes broad indications and enhanced formulations.
- Pfizer’s Viberzi: Focuses on functional GI disorders but faces stiff competition from established products and emerging therapies.
- Ferring’s Biotech Initiatives: Investing in novel biologics for GI indications, aiming to complement traditional small-molecule therapies.
Conclusion
Salix maintains a strong position within the GI market through its flagship product, Xifaxan, and focused therapeutic area. Its predominantly niche focus presents opportunities for growth via expansion of indications and international markets but also exposes it to patent expiration risks and industry consolidation. Strategic partnerships and innovation remain essential for maintaining its competitive edge.
Key Takeaways
- Salix's 2022 revenue centered on Xifaxan, which dominates its portfolio.
- Competition includes AbbVie, Pfizer, and Ferring, with varying market shares.
- Growth opportunities include expanding indications, geographic expansion, and biosimulants.
- Patent expiration in 2029 poses a significant risk for revenue erosion.
- Strategic alliances and pipeline diversification are necessary to sustain long-term competitiveness.
FAQs
1. What is the primary revenue driver for Salix Pharmaceuticals?
Xifaxan is the main source of revenue, accounting for approximately 70% of total sales.
2. How does patent expiration impact Salix’s future sales?
The patent for Xifaxan expires in 2029, potentially opening the market to generic competitors and reducing margins.
3. Which markets are most promising for Salix’s international expansion?
Europe and Asia exhibit growing demand for GI therapeutics, with unmet needs in chronic GI disorder management.
4. What are the major competitors to Salix in the GI space?
AbbVie (Linzess), Pfizer (Viberzi), and Ferring Pharmaceuticals (biologics) present the strongest competition.
5. How can Salix sustain its market position amid industry consolidation?
Through innovation, expanding indications, strategic licensing, and expanding international presence.
References
- Grand View Research. (2022). Gastrointestinal therapeutics market size, share & trends analysis. Retrieved from https://www.grandviewresearch.com/industry-analysis/gastrointestinal-therapeutics-market
- Salix Pharmaceuticals. (2022). Annual report.
- MarketWatch. (2022). Top GI drug manufacturers market share analysis. Retrieved from https://www.marketwatch.com/