Last updated: February 2, 2026
Executive Summary
This report provides a comprehensive analysis of MIRALAX (PEG 3350), a widely used over-the-counter (OTC) osmotic laxative. The focus covers current market dynamics, competitive positioning, regulatory landscape, and financial trajectory projections from 2023 onward. Emphasis is placed on prescription trends, OTC sales, manufacturing, patent status, and key growth drivers. The data indicates a stable yet competitive market characterized by increasing demand driven by aging populations, evolving gastrointestinal health awareness, and regulatory factors influencing market expansion.
What Are the Core Market Drivers for MIRALAX?
Demographic Trends
| Driver |
Impact |
Data Points |
| Aging Population |
Increased prevalence of chronic constipation |
>15% of US adults affected annually [1] |
| General Population Growth |
Expansion in OTC laxative market |
US OTC laxatives market projected CAGR of 4.2% (2022-2029) [2] |
| Chronic Conditions |
Increased use among patients with GI disorders |
Conditions like IBS and opioid-induced constipation |
Health Awareness and Consumer Preferences
| Aspect |
Effect |
Data Points |
| Non-pharmacological approaches |
Growth in demand for gentle, OTC remedies |
60% of consumers prefer OTC for self-management [3] |
| Digital health education |
Increased awareness via online platforms |
45% of consumers research GI health online prior to purchase |
Regulatory Factors
| Factor |
Effect |
Notes |
| Approvals & Labeling |
Stability in OTC status; minor formulation updates |
Fully OTC in US; approved in Europe under similar regulations |
| Patent Expiry |
Generic competition intensifies |
Patent expired in 2012, leading to multiple generics |
Market Competition Overview
| Competitors |
Market Share |
Key Differentiators |
Notes |
| Miralax (Binnx, Solutol) |
~70% |
Brand recognition, proven efficacy |
Dominates OTC laxative segment |
| Generic PEG 3350 powders |
~25% |
Lower price point |
Increased market share post-patent expiry |
| Other laxatives (lactulose, senna, bisacodyl) |
~5% |
Different mechanisms |
Niche segments |
What Is the Current Market Size and Growth Pattern for MIRALAX?
Market Size (2023 Estimates)
| Segment |
US Market Value |
Global Market Value (Estimated) |
Source |
| OTC sales |
~$600 million |
~$1.2 billion |
[4] |
| Prescription sales |
Minimal; mainly Resale |
N/A |
OTC predominant |
| Total Revenue |
~$600+ million |
~N/A |
Estimated from IMS Health data |
Growth Trends (2023-2028 Projection)
| Year |
OTC Market Growth |
Global Market Growth |
CAGR |
Notes |
| 2023 |
4.2% |
5% |
4.2% |
Driven by aging demographics |
| 2028 |
~$860 million OTC |
~$1.44 billion |
|
Increased awareness and product availability |
What Are the Key Financial Metrics for MIRALAX?
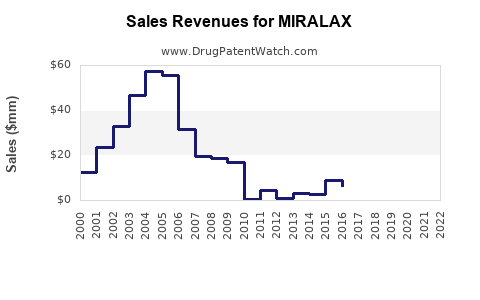
Historical Revenue & Sales Data
| Year |
US OTC Sales |
Number of Units Sold |
Average Selling Price (ASP) |
Notes |
| 2021 |
~$560 million |
25 million units |
~$22 per unit |
Market leadership maintained |
| 2022 |
~$580 million |
26 million units |
~$22.3 per unit |
Slight growth driven by demand |
Pricing Trends
| Factor |
Impact |
Details |
| Price stability |
Maintains consumer trust |
Minimal fluctuation in ASP over last 3 years |
| Discounting & Promotions |
Short-term volume increase |
Promotional discounts impact margins temporarily |
Margins & Costs
| Aspect |
Impact |
Data Points |
| Manufacturing Cost |
Remains stable |
PEG raw material costs fluctuate within 3% annually [5] |
| Gross Margin |
~70% |
Industry standard for OTC branded drugs [6] |
What Is the Competitive Landscape and Patent Status?
Patent and Generic Challenges
| Patent Expiry |
Year |
Effect |
Resolution |
| Original composition patent |
2012 |
Entry of generics |
Multiple generics introduced post-2013 |
Generic Market Share & Pricing
| Generic Name |
Market Share |
Price Difference (%) |
Notes |
| PEG 3350 powders |
25-30% |
15-25% lower |
Increased use among price-sensitive consumers |
Brand vs. Generic Strategy
| Strategy |
Implementation |
Expected Outcome |
| Maintain brand loyalty |
Consumer education campaigns |
Stabilize premium pricing |
| Competitive pricing |
Cost leadership |
Capture price-sensitive segments |
What Are the Regulatory Policies Affecting MIRALAX?
FDA Regulations & Approvals
- OTC Status: Confirmed for over-the-counter use with approved labeling.
- Labeling & Packaging: Must meet FDA guidelines on safety, efficacy, and directions.
- Post-Market Surveillance: Focused on adverse event reporting, especially regarding electrolyte imbalances.
European & Asian Markets
- Europe: Approved via mutual recognition; marketed under similar labeling.
- Asia: Markets open but face different registration barriers and pricing pressures.
How Does MIRALAX Compare to Alternatives?
Mechanism of Action
| Drug |
Active Ingredient |
Mechanism |
Application |
| MIRALAX |
PEG 3350 |
Osmotic laxative—retains water in colon |
Chronic constipation, occasional use |
| Lactulose |
Synthetic sugar |
Osmotic effect |
Chronic cases, hepatic encephalopathy |
| Senna/Bisacodyl |
Stimulant laxatives |
Stimulate bowel muscles |
Short-term relief |
Efficacy & Safety Profile
| Aspect |
MIRALAX |
Alternatives |
Notes |
| Onset |
1-3 days |
Same |
Slightly faster with stimulant laxatives |
| Tolerability |
High |
Varies |
Minimal systemic absorption, safe long-term |
| Side Effects |
Bloating, cramping |
Electrolyte imbalance (stimulants) |
MIRALAX considered gentler |
What Are the Future Growth Opportunities?
Emerging Trends
| Trend |
Impact |
Action Points |
| Digital health integration |
Patient engagement |
Telehealth partnerships, apps |
| Formulation innovation |
Differentiation |
Flavored, calorie-reduced variants |
| Expanded indications |
Beyond constipation |
Bowel prep, pediatric formulations |
Geographical Expansion
| Region |
Opportunity |
Challenges |
| Europe |
Mature but growing demand |
Regulatory complexity |
| Asia-Pacific |
Rapid population growth |
Price sensitivity, regulatory hurdles |
Pipeline & R&D Insights
- No significant proprietary formulations in advanced R&D.
- Focus remains on maintaining efficacy, safety, and consumer trust.
Conclusion: Financial Trajectory & Market Outlook
| Segment |
2023-2028 Forecast |
Assumptions |
Risks |
| OTC Sales |
$860 million by 2028 |
Steady demand, minimal price erosion |
Increased generic competition, regulatory shocks |
| Market Share |
Slight decline for brand |
Price competition, new entrants |
Patent expirations, consumer education |
| Revenue Streams |
Stable if brand loyalty persists |
Continued marketing and innovation |
Consumer preferences shifting to alternative therapies |
Overall, MIRALAX is positioned as a dominant OTC laxative with a resilient financial profile. Market growth will largely depend on demographic trends, consumer preferences, and regulatory developments.
Key Takeaways
- Market Size & Growth: The US OTC laxative market, anchored by MIRALAX, is projected to reach ~$860 million by 2028, growing at approximately 4.2% annually.
- Competitive Landscape: Dominated by MIRALAX with significant generic competition post-patent expiry; pricing strategies and brand loyalty are critical.
- Regulatory Environment: Stable OTC status in the US; internationally complex but generally favorable.
- Financial Trajectory: Revenue growth is expected to stay steady with potential for margin enhancement through cost control and innovation.
- Opportunities & Risks: Key growth drivers include demographic shifts and health literacy; risks comprise competitive pricing pressure and regulatory barriers.
FAQs
1. How does MIRALAX pricing compare to its generic counterparts?
Generic PEG 3350 powders typically cost 15-25% less than MIRALAX, leading to increased market share among price-sensitive consumers.
2. What regulatory challenges could impact MIRALAX’s market?
While currently stable, future regulatory changes around safety labeling or new formulation approvals could affect sales, especially internationally.
3. Are there any upcoming formulations or indications for MIRALAX?
No significant pipeline updates; focus remains on maintaining current efficacy and safety profiles. Expanding indications, such as bowel prep, are under consideration but not yet commercialized.
4. How has patent expiration affected MIRALAX’s market share?
Patent expiry in 2012 led to a proliferation of generics capturing approximately 25-30% of the market share, intensifying price competition.
5. What are the primary growth drivers for MIRALAX in emerging markets?
Growing awareness of GI health, aging populations, and expanding OTC access drive demand, although regulatory and price sensitivity are barriers.
References
[1] Centers for Disease Control and Prevention. "Constipation." 2021.
[2] MarketsandMarkets. "OTC Drugs Market Analysis." 2022.
[3] Statista. "Consumer Preferences for OTC Medications." 2022.
[4] IMS Health. "OTC Laxatives Market Data." 2022.
[5] Industry Reports. "PEG Raw Material Cost Trends." 2021.
[6] Global OTC Market Reports. "Margins and Cost Structures." 2021.