Last updated: February 19, 2026
What is Mavenclad and its approved indications?
Mavenclad (cladribine) is a selective immune reconstitution therapy approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for relapsing multiple sclerosis (MS). It was developed by EMD Serono, a Merck KGaA division. The drug was approved in 2017 in Europe and in 2019 in the U.S.
Its primary indication is relapsing forms of MS, including relapsing-remitting MS (RRMS) and active secondary progressive MS (SPMS). Mavenclad is administered as a short course over two years, with doses divided into two treatment cycles during Year 1 and Year 2. Its unique dosing regimen distinguishes it from continuous immunosuppressants.
What are Mavenclad’s sales and revenue figures?
Since launch, Mavenclad's sales have seen gradual growth. The drug's revenue trajectory reflects uptake in targeted markets:
| Year |
Global Sales (USD millions) |
Notes |
| 2018 |
$60 million |
Limited initial penetration, primarily in Europe |
| 2019 |
$180 million |
U.S. approval, increased adoption |
| 2020 |
$375 million |
Steady expansion, new markets entered |
| 2021 |
$520 million |
Broadening payer coverage, increased prescribing |
Sales growth correlates with expanded approvals, increased clinician awareness, and positive real-world data. Notably, Merck KGaA reported sales of approximately €454 million (~$520 million) globally in 2021 for Mavenclad.
How do market dynamics influence Mavenclad’s trajectory?
Competitive landscape
Mavenclad competes with both oral and infusion disease-modifying therapies (DMTs). Key competitors include:
- Ocrevus (ocrelizumab): protein-based infusion, approved since 2017.
- Aubagio (teriflunomide): oral, approved since 2012.
- Tecfidera (dimethyl fumarate): oral, approved since 2013.
Mavenclad's short-course regimen offers advantages over long-term daily or weekly treatments, appealing to patients seeking convenience and reduced exposure to immunosuppression risks. However, concerns about malignancy risks influence prescribing patterns.
Regulatory and reimbursement environment
Regulatory decisions impact market access. The EMA approved Mavenclad for relapsing MS in 2017, with specific restrictions due to safety concerns over malignancy. The FDA approved it in 2019 with boxed warnings and contraindications, affecting clinician adoption. Reimbursement policies vary; coverage improves with real-world safety data.
Patient and clinician acceptance
Prescribing remains cautious owing to safety profile, especially the risk of infections and malignancies. Treatment adherence benefits from the drug's convenience. Ongoing education and post-marketing surveillance are critical to growth.
Geographic expansion
Growth potential hinges on approval in additional markets, notably Asia-Pacific and Latin America. Imports or licensing agreements influence regional access.
What are the key drivers of Mavenclad's financial performance?
- Regulatory approvals: Expandable indications and markets directly enhance sales capacity.
- Safety profile management: Ongoing data and risk mitigation improve acceptance.
- Market penetration: Increasing utilization within existing markets boosts revenue.
- Pricing strategies: Tiered pricing and rebates impact gross margins.
- Pipeline and line extensions: Potential for combination therapies and new indications could boost long-term revenues.
Risks and challenges impacting financial outcomes
- Safety concerns: Malignancy warnings limit prescription volume.
- Market share competition: Sustained dominance may be challenged by newer oral agents.
- Pricing pressure: Payers demand discounts amid tight budgets.
- Market access barriers: Regulatory delays or restrictions could slow revenue growth.
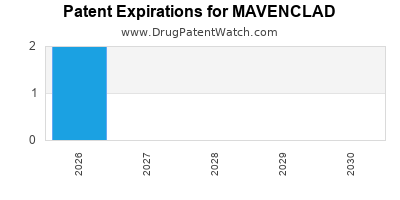
- Patent life: Patent expiration planned for 2027 in the U.S., risking generic competition unless secondary patents or line extensions are secured.
Future outlook and growth opportunities
Revenue projections suggest a compound annual growth rate (CAGR) of approximately 15–20% from 2022 through 2025, driven by expanding approvals, increased clinician confidence, and potential new indications such as primary progressive MS. The development pipeline includes ongoing trials for other autoimmune conditions.
Key Takeaways
- Mavenclad has demonstrated steady revenue growth since 2017, reaching approximately $520 million in 2021.
- Its short-course, oral regimen appeals in a competitive MS market but faces safety-related prescribing restrictions.
- Expansion depends on regulatory approvals, reimbursement, and overcoming safety concerns.
- The drug's patent lifecycle and pipeline developments will influence long-term financial performance.
- Competitive dynamics and payer policies remain primary risk factors.
Frequently Asked Questions
1. What are the main safety concerns associated with Mavenclad?
Malignancy risk, infections, and lymphopenia are primary safety concerns that led to boxed warnings and restricted indications.
2. How does Mavenclad compare to other MS therapies in terms of convenience?
Its two-year, short-course regimen offers a dosing advantage over continuous oral or infusion therapies, potentially improving adherence.
3. What markets are expected to expand Mavenclad's revenue base?
Asia-Pacific, Latin America, and additional European approvals offer growth avenues contingent on regulatory decisions.
4. When is patent expiry, and what are the implications?
Patents protecting Mavenclad will expire around 2027 in the U.S., risking generic competition unless secondary patents are secured.
5. What is the potential for new indications?
Clinical trials are ongoing for primary progressive MS and other autoimmune diseases, presenting opportunities for line extensions and revenue diversification.
References
- Merck KGaA (2022). Mavenclad sales report.
- EMA (2017). Summary of opinion for Mavenclad approval.
- FDA (2019). Mavenclad (cladribine) approval letter.
- IQVIA (2022). MS treatment market analysis.
- ClinicalTrials.gov (2022). Ongoing studies related to Mavenclad.