Lamictal ODT Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Lamictal Odt, and what generic alternatives are available?
Lamictal Odt is a drug marketed by Glaxosmithkline Llc and is included in one NDA.
The generic ingredient in LAMICTAL ODT is lamotrigine. There are thirty-two drug master file entries for this compound. Fifty-two suppliers are listed for this compound. Additional details are available on the lamotrigine profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Lamictal Odt
A generic version of Lamictal ODT was approved as lamotrigine by DR REDDYS LABS LTD on January 22nd, 2009.
Summary for Lamictal ODT
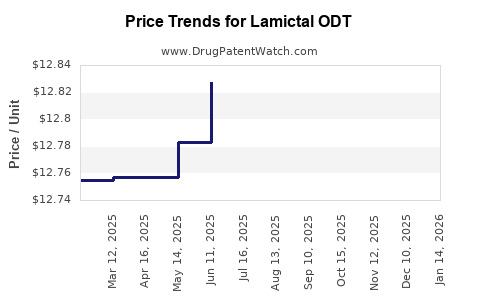
See drug prices for Lamictal ODT
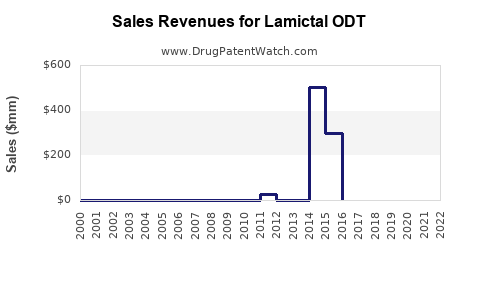
Recent Clinical Trials for Lamictal ODT
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Dr Cipto Mangunkusumo General Hospital | Phase 4 |
| Brown University | Phase 2 |
| National Institute on Alcohol Abuse and Alcoholism (NIAAA) | Phase 2 |
Pharmacology for Lamictal ODT
| Drug Class | Anti-epileptic Agent Mood Stabilizer |
| Mechanism of Action | Dihydrofolate Reductase Inhibitors Organic Cation Transporter 2 Inhibitors |
| Physiological Effect | Decreased Central Nervous System Disorganized Electrical Activity |
Anatomical Therapeutic Chemical (ATC) Classes for Lamictal ODT
Paragraph IV (Patent) Challenges for LAMICTAL ODT
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LAMICTAL ODT | Orally Disintegrating Tablets | lamotrigine | 25 mg, 50 mg, 100 mg, and 200 mg | 022251 | 1 | 2009-12-21 |
US Patents and Regulatory Information for Lamictal ODT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline Llc | LAMICTAL ODT | lamotrigine | TABLET, ORALLY DISINTEGRATING;ORAL | 022251-001 | May 8, 2009 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Glaxosmithkline Llc | LAMICTAL ODT | lamotrigine | TABLET, ORALLY DISINTEGRATING;ORAL | 022251-004 | May 8, 2009 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Glaxosmithkline Llc | LAMICTAL ODT | lamotrigine | TABLET, ORALLY DISINTEGRATING;ORAL | 022251-002 | May 8, 2009 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Glaxosmithkline Llc | LAMICTAL ODT | lamotrigine | TABLET, ORALLY DISINTEGRATING;ORAL | 022251-003 | May 8, 2009 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Lamictal ODT
See the table below for patents covering Lamictal ODT around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Uruguay | 31205 | COMPOSICIONES DE TABLETAS DE DESINTEGRACION ORAL DE LAMOTRIGINA | ⤷ Try a Trial |
| Chile | 2008001966 | Composicion de disolucion oral que comprende lamotrigina,en forma de particulas con lamotrigina recubiertas con una capa para enmascarar el sabor y granulos que comprenden un desinfectante y un alcohol de azucar y/o un hidrato de carbono; metodo de preparacion;uso para tratar trastornos del humor y prevencion de ataques. | ⤷ Try a Trial |
| Saudi Arabia | 08290401 | تركيبات أقراص لاموتريجين تتفتت بالفم (Orally Disintegrating Tablet Compositions of Lamotrigine) | ⤷ Try a Trial |
| Japan | 5346021 | ⤷ Try a Trial | |
| China | 104161733 | Orally disintegrating tablet compositions of lamotrigine | ⤷ Try a Trial |
| Hong Kong | 1201745 | 拉莫三嗪的口服崩解片劑組合物 (ORALLY DISINTEGRATING TABLET COMPOSITIONS OF LAMOTRIGINE) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


