LONSURF Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Lonsurf, and when can generic versions of Lonsurf launch?
Lonsurf is a drug marketed by Taiho Oncology and is included in one NDA. There are six patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and eighteen patent family members in thirty-three countries.
The generic ingredient in LONSURF is tipiracil hydrochloride; trifluridine. There are two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the tipiracil hydrochloride; trifluridine profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Lonsurf
A generic version of LONSURF was approved as tipiracil hydrochloride; trifluridine by NATCO on June 13th, 2023.
Summary for LONSURF
| International Patents: | 118 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 13 |
| Clinical Trials: | 55 |
| Patent Applications: | 27 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for LONSURF |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for LONSURF |
| What excipients (inactive ingredients) are in LONSURF? | LONSURF excipients list |
| DailyMed Link: | LONSURF at DailyMed |
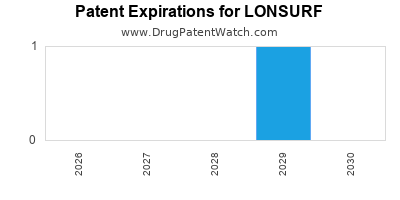

Paragraph IV (Patent) Challenges for LONSURF
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LONSURF | Tablets | tipiracil hydrochloride; trifluridine | 15 mg/6.14 mg and 20 mg/8.19 mg | 207981 | 4 | 2019-09-23 |
US Patents and Regulatory Information for LONSURF
LONSURF is protected by ten US patents and one FDA Regulatory Exclusivity.
Patents protecting LONSURF
Method for treating cancer patients with severe renal impairment
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC COLORECTAL CANCER ALONE OR WITH BEVACIZUMAB IN SEVERELY RENALLY IMPAIRED PATIENTS PREVIOUSLY TREATED WITH FLUOROPYRIMIDINE-, OXALIPLATIN- AND IRINOTECAN THERAPY, AN ANTI-VEGF BIOLOGIC, AND IF RAS WILD-TYPE, ANTI-EGFR THERAPY
Method for treating cancer patients with severe renal impairment
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC GASTRIC OR GJA IN SEVERELY RENALLY IMPAIRED PATIENTS TREATED WITH AT LEAST TWO LINES OF CHEMOTHERAPY THAT INCLUDED A FLUOROPYRIMIDINE, A PLATINUM, A TAXANE OR IRINOTECAN, AND IF APPROPRIATE, HER2/NEU-TARGETED THERAPY
Stable crystal form of tipiracil hydrochloride and crystallization method for the same
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Method for treating cancer patients with severe renal impairment
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC COLORECTAL CANCER ALONE OR WITH BEVACIZUMAB IN SEVERELY RENALLY IMPAIRED PATIENTS PREVIOUSLY TREATED WITH FLUOROPYRIMIDINE-, OXALIPLATIN- AND IRINOTECAN THERAPY, AN ANTI-VEGF BIOLOGIC, AND IF RAS WILD-TYPE, ANTI-EGFR THERAPY
Method for treating cancer patients with severe renal impairment
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC GASTRIC OR GJA IN SEVERELY RENALLY IMPAIRED PATIENTS TREATED WITH AT LEAST TWO LINES OF CHEMOTHERAPY THAT INCLUDED A FLUOROPYRIMIDINE, A PLATINUM, A TAXANE OR IRINOTECAN, AND IF APPROPRIATE, HER2/NEU-TARGETED THERAPY
Stable crystal form of tipiracil hydrochloride and crystallization method for the same
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC COLORECTAL CANCER WITH BEVACIZUMAB IN PATIENTS PREVIOUSLY TREATED WITH FLUOROPYRIMIDINE-, OXALIPLATIN- AND IRINOTECAN-BASED CHEMOTHERAPY, AN ANTI-VEGF BIOLOGICAL THERAPY, AND IF RAS WILD-TYPE, AN ANTI-EGFR THERAPY
Method of administrating an anticancer drug containing .alpha., .alpha., .alpha.-trifluorothymidine and thymidine phosphorylase inhibitor
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF ADULTS WITH METASTATIC GASTRIC OR GJA PREVIOUSLY TREATED WITH AT LEAST TWO PRIOR LINES OF CHEMOTHERAPY THAT INCLUDED A FLUOROPYRIMIDINE, A PLATINUM, EITHER A TAXANE OR IRINOTECAN, AND IF APPROPRIATE, HER2/NEU-TARGETED THERAPY
Method of administrating an anticancer drug containing .alpha., .alpha., .alpha.-trifluorothymidine and thymidine phosphorylase inhibitor
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF PATIENTS WITH METASTATIC COLORECTAL CANCER WHO HAVE BEEN PREVIOUSLY TREATED WITH FLUOROPYRIMIDINE-, OXALIPLATIN- AND IRINOTECAN-BASED CHEMOTHERAPY, AN ANTI-VEGF BIOLOGICAL THERAPY, AND IF RAS WILD-TYPE, AN ANTI-EGFR THERAPY
Method of administrating an anticancer drug containing .alpha., .alpha., .alpha.-trifluorothymidine and thymidine phosphorylase inhibitor
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC COLORECTAL CANCER ALONE OR WITH BEVACIZUMAB IN PATIENTS PREVIOUSLY TREATED WITH FLUOROPYRIMIDINE-, OXALIPLATIN- AND IRINOTECAN-BASED CHEMOTHERAPY, AN ANTI-VEGF BIOLOGICAL THERAPY, AND IF RAS WILD-TYPE, AN ANTI-EGFR THERAPY
FDA Regulatory Exclusivity protecting LONSURF
TREATMENT OF ADULT PATIENTS WITH METASTATIC GASTRIC OR GASTROESOPHAGEAL JUNCTION ADENOCARCINOMA PREVIOUSLY TREATED WITH AT LEAST TWO PRIOR LINES OF CHEMOTHERAPY, AND IF APPROPRIATE, HER2/NEU-TARGETED THERAPY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-002 | Sep 22, 2015 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-002 | Sep 22, 2015 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for LONSURF
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-002 | Sep 22, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-002 | Sep 22, 2015 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for LONSURF
When does loss-of-exclusivity occur for LONSURF?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 14282281
Estimated Expiration: ⤷ Try a Trial
Patent: 17208215
Estimated Expiration: ⤷ Try a Trial
Patent: 18219967
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 2015031619
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 14999
Estimated Expiration: ⤷ Try a Trial
Patent: 85006
Estimated Expiration: ⤷ Try a Trial
China
Patent: 4395307
Estimated Expiration: ⤷ Try a Trial
Patent: 6967051
Estimated Expiration: ⤷ Try a Trial
Patent: 9912573
Estimated Expiration: ⤷ Try a Trial
Patent: 0746403
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0211903
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 24868
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 12255
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 12255
Estimated Expiration: ⤷ Try a Trial
Patent: 05779
Estimated Expiration: ⤷ Try a Trial
Georgia, Republic of
Patent: 0186841
Estimated Expiration: ⤷ Try a Trial
Patent: 0186875
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 03071
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 57352
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 2014203877
Estimated Expiration: ⤷ Try a Trial
Patent: 64409
Estimated Expiration: ⤷ Try a Trial
Patent: 26883
Estimated Expiration: ⤷ Try a Trial
Patent: 18145204
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 12255
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 3410
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 15016986
Estimated Expiration: ⤷ Try a Trial
Morocco
Patent: 668
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 4090
Estimated Expiration: ⤷ Try a Trial
Philippines
Patent: 015502809
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 12255
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 12255
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 40417
Estimated Expiration: ⤷ Try a Trial
Patent: 74441
Estimated Expiration: ⤷ Try a Trial
Patent: 16100972
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 691
Estimated Expiration: ⤷ Try a Trial
Singapore
Patent: 201710916Y
Estimated Expiration: ⤷ Try a Trial
Patent: 201912293U
Estimated Expiration: ⤷ Try a Trial
Patent: 202007643S
Estimated Expiration: ⤷ Try a Trial
Patent: 201509189S
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 12255
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1806346
Estimated Expiration: ⤷ Try a Trial
Patent: 160020560
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 98408
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 39592
Estimated Expiration: ⤷ Try a Trial
Patent: 1534601
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 2626
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering LONSURF around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2594713 | MEDICAMENTS ANTICANCEREUX CONTENANT DE LA A,A,A-TRIFLUOROTHYMIDINE ET UN INHIBITEUR DE LA THYMIDINE PHOSPHORYLASE (ANTICANCER DRUG CONTAINING A,A,A-TRIFLUOROTHYMIDINE AND THYMIDINE PHOSPHORYLASE INHIBITOR) | ⤷ Try a Trial |
| European Patent Office | 1080726 | AGENTS ATTENUANT LES EFFETS SECONDAIRES (AGENTS FOR RELIEVING SIDE EFFECTS) | ⤷ Try a Trial |
| Japan | 2018145204 | チピラシル塩酸塩の安定形結晶及びその結晶化方法 (STABLE FORM CRYSTAL OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD THEREFOR) | ⤷ Try a Trial |
| Japan | WO2006080327 | α,α,α−トリフルオロチミジンとチミジンホスホリラーゼ阻害剤とを配合した抗癌剤 | ⤷ Try a Trial |
| China | 109912573 | 地匹福林盐酸盐的稳定型晶体及其结晶化方法 (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for LONSURF
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1849470 | 17C1028 | France | ⤷ Try a Trial | PRODUCT NAME: TRIFLURIDINE/TIPIRACIL; REGISTRATION NO/DATE: EU/1/16/1096 20160427 |
| 1849470 | 132017000102371 | Italy | ⤷ Try a Trial | PRODUCT NAME: TRIFLURDINA IN COMBINAZIONE CON TIPIRACILE O UN SALE DI TIPIRACILE QUALE TIPIRACILE IDROCLORURO(LONSURF); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/16/1096, 20160427 |
| 1849470 | 34/2017 | Austria | ⤷ Try a Trial | PRODUCT NAME: TRIFLURIDIN / TIPIRACIL ODER DEREN PHARMAZEUTISCH WIRKSAMEN SALZE; REGISTRATION NO/DATE: EU/1/16/1096 (MITTEILUNG) 20160427 |
| 1849470 | PA2017024 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: TRIFLURIDINAS/TIPIRACILAS; REGISTRATION NO/DATE: EU/1/16/1096 20160425 |
| 1849470 | SPC/GB17/049 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: TRIFLURIDINE WITH TIPACRIL HYDROCHLORIDE; REGISTERED: UK EU/1/16/1096/001(NI) 20160427; UK EU/1/16/1096/002(NI) 20160427; UK EU/1/16/1096/003(NI) 20160427; UK EU/1/16/1096/004(NI) 20160427; UK EU/1/16/1096/005(NI) 20160427; UK EU/1/16/1096/006(NI) 20160427; UK PLGB 05815/0112 20160427; UK PLGB 05815/0113 20160427 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


