Last updated: February 19, 2026
What is Livalo?
Livalo (pitavastatin) is a statin medication used to lower cholesterol levels. It works by inhibiting HMG-CoA reductase, an enzyme that plays a key role in the liver's production of cholesterol. Approved by the U.S. Food and Drug Administration (FDA) in July 2009, Livalo is prescribed to treat hypercholesterolemia and reduce the risk of cardiovascular events in patients with elevated cholesterol. It is available in tablet form at dosages of 1 mg, 2 mg, and 4 mg [1].
Market Landscape and Competitive Environment
The market for cholesterol-lowering medications is highly competitive, dominated by statins and increasingly by newer classes of drugs. Livalo competes within the broader statin segment, which includes widely recognized brands like Lipitor (atorvastatin), Crestor (rosuvastatin), and Zocor (simvastatin).
Key Market Players and Products
- Lipitor (atorvastatin): Developed by Pfizer, Lipitor was once the world's best-selling drug. Its patent expiration led to the widespread availability of generic versions, significantly impacting brand-name sales.
- Crestor (rosuvastatin): Manufactured by AstraZeneca, Crestor has also been a significant player. Like Lipitor, its market share has been affected by generic competition following patent expiry.
- Zocor (simvastatin): Originally developed by Merck, Zocor is another statin whose market presence has been shaped by generic availability.
- Pravachol (pravastatin): Developed by Bristol Myers Squibb, Pravachol is another established statin.
- Lescol (fluvastatin): Developed by Novartis, Lescol is also a part of the statin landscape.
Livalo, developed by Kowa Company, Ltd. and marketed in the U.S. by Eli Lilly and Company (prior to 2016), entered a mature market. Its differentiation strategy has focused on its specific efficacy and safety profile [2]. However, the presence of numerous generic statins with established clinical track records and lower price points presents a significant challenge.
Therapeutic Positioning and Differentiation
Livalo's positioning within the statin class is based on its pharmacologic profile. Clinical studies have indicated comparable or superior efficacy to some other statins in reducing LDL cholesterol and triglycerides, with a generally favorable safety profile regarding muscle-related side effects. However, these differences may not always translate to substantial market share gains in a market where cost and established familiarity are strong drivers [3].
The emergence of non-statin therapies, such as PCSK9 inhibitors (e.g., Praluent, Repatha) and ezetimibe, has further fragmented the market. These drugs offer alternative mechanisms for cholesterol reduction, often for patients who cannot tolerate statins or require additional LDL-lowering. While these newer agents are typically more expensive, they target specific patient populations and may be preferred in certain clinical scenarios, indirectly affecting the overall demand for traditional statins [4].
Patent Landscape and Exclusivity
The patent landscape for Livalo is critical to its market exclusivity and subsequent financial performance. Understanding the patent portfolio and its expiry dates is essential for forecasting generic entry and potential revenue erosion.
Key Patents and Expiry Dates
Kowa Company, Ltd. holds the primary patents for pitavastatin. The key U.S. patent for pitavastatin is U.S. Patent No. 5,856,335, which covers the compound itself and was granted on January 5, 1999. This patent was set to expire in November 2015. However, patent term extensions are possible under U.S. law to compensate for regulatory review delays.
- U.S. Patent No. 5,856,335: This core patent for pitavastatin.
- Original Expiry: November 19, 2015.
- Potential Extension: Patent term extension (PTE) could have been sought, but the exact duration and outcome are subject to FDA and USPTO calculations based on the patent owner's application and the regulatory review period.
Beyond the compound patent, there can be patents covering specific formulations, manufacturing processes, or methods of use. These secondary patents can extend market exclusivity beyond the expiry of the primary compound patent.
Generic Entry and Impact on Market Share
The expiry of key patents opens the door for generic manufacturers to launch their versions of Livalo. Generic drugs are typically priced significantly lower than their branded counterparts, leading to rapid erosion of market share for the originator product.
- Anticipated Generic Launch: Following the expiry of the primary patent (and any relevant extensions), generic versions of pitavastatin began to enter the market. The exact timing of generic launches depends on the approval of Abbreviated New Drug Applications (ANDAs) by the FDA.
- Market Share Decline: Post-generic entry, branded Livalo sales are expected to decline sharply as healthcare providers and pharmacies switch to more cost-effective generic alternatives. This phenomenon is well-documented across the pharmaceutical industry. For example, Lipitor, after its patent expiry, saw its annual sales drop from over $10 billion to less than $2 billion within a few years due to generic competition [5].
The competitive landscape for generic pitavastatin includes multiple manufacturers, further intensifying price competition and accelerating market share loss for branded Livalo.
Financial Performance and Revenue Trajectory
Livalo's financial performance is intrinsically linked to its patent exclusivity and market penetration. As a drug that entered a competitive market after several other statins, its revenue trajectory has been influenced by these factors.
Historical Sales Data
Kowa Company and its U.S. marketing partners (initially Eli Lilly, later others) have reported Livalo's sales. Specific historical sales figures for Livalo in the U.S. market:
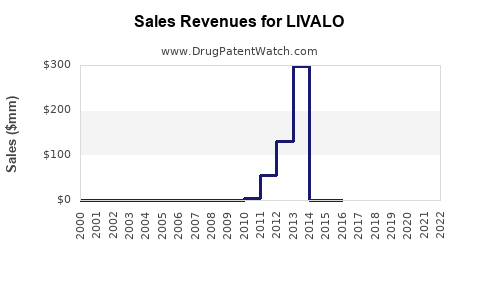
- 2012: Approximately $144 million [6]
- 2013: Approximately $165 million [7]
- 2014: Approximately $178 million [8]
- 2015: Approximately $187 million [9] (approaching the expiry of the primary patent)
- 2016: Approximately $188 million [10] (initial impact of patent expiry may be gradual)
- 2017: Sales likely to see a more significant decline due to increasing generic penetration.
These figures illustrate a period of steady growth leading up to patent expiry, a common pattern for successful branded drugs.
Impact of Patent Expiry on Revenue
The expiry of U.S. Patent No. 5,856,335 in November 2015 marked a critical turning point for Livalo's revenue.
- Pre-Generic Era (Pre-2016): Livalo commanded premium pricing, contributing to its steady revenue growth as it gained market acceptance.
- Post-Generic Era (2016 onwards): Following the entry of generic pitavastatin, branded Livalo sales have experienced, and are expected to continue to experience, a significant decline. Generic manufacturers compete primarily on price, forcing down the overall market price for pitavastatin. Branded Livalo's market share will contract as prescribers and payers shift to lower-cost generic options.
The magnitude of revenue decline for a branded statin post-patent expiry can be substantial, often exceeding 70-90% within a few years.
Projected Future Financials
Forecasting Livalo's future financial trajectory requires considering:
- Generic Market Share: The rate at which generic pitavastatin captures market share. This is typically rapid, driven by payer formularies and physician prescribing habits.
- Pricing Pressure: Intense price competition among generic manufacturers will continue to suppress the overall market price.
- Niche Market: Branded Livalo might retain a small market share among patients or physicians who specifically request the branded product, or for whom it is preferred by payers due to specific contract terms.
- Global Markets: The patent expiry and generic landscape can differ by country, influencing global revenue streams. However, the U.S. market is a significant driver of pharmaceutical revenue.
Given these factors, Livalo's revenue is projected to continue a steep downward trend. Its contribution to Kowa Company's overall revenue will diminish significantly as the generic market matures.
Key Takeaways
Livalo's market performance has been shaped by its entry into a mature statin market and the subsequent expiry of its core patent.
- Patent Expiry: The November 2015 expiry of U.S. Patent No. 5,856,335 for pitavastatin enabled generic competition, fundamentally altering Livalo's revenue stream.
- Revenue Decline: Branded Livalo sales experienced growth leading up to patent expiry but are expected to undergo a steep and sustained decline due to the introduction of lower-cost generic versions.
- Competitive Landscape: The statin market is highly competitive, with established players and significant generic penetration. The emergence of non-statin therapies further complicates the therapeutic landscape.
- Financial Trajectory: Livalo's financial trajectory has shifted from growth to significant contraction post-patent expiry, a typical pattern for branded pharmaceuticals facing generic competition.
Frequently Asked Questions
-
What is the primary mechanism of action for Livalo (pitavastatin)?
Livalo inhibits HMG-CoA reductase, an enzyme essential for cholesterol synthesis in the liver, thereby lowering LDL cholesterol and triglyceride levels.
-
When did the core U.S. patent for Livalo expire?
The primary U.S. patent (U.S. Patent No. 5,856,335) for pitavastatin expired in November 2015.
-
How does generic competition typically affect the sales of branded drugs like Livalo?
Generic competition generally leads to a rapid and substantial decrease in the sales of branded drugs as lower-cost generic alternatives become available.
-
Are there other classes of drugs that compete with statins like Livalo?
Yes, PCSK9 inhibitors and ezetimibe are examples of non-statin therapies that compete with statins, particularly for patients requiring additional cholesterol reduction or who cannot tolerate statins.
-
What factors will determine the future revenue of branded Livalo?
Future revenue will depend on the rate of generic market share acquisition, ongoing price competition among generic manufacturers, and any potential residual market niche for the branded product.
Citations
[1] U.S. Food and Drug Administration. (2009, July 31). FDA approves Livalo (pitavastatin) tablets. U.S. Food and Drug Administration. https://www.fda.gov/drugs/postmarket-drug-safety-information-for-patients-and-providers/fda-approves-livalo-pitavastatin-tablets
[2] Kowa Company, Ltd. (n.d.). Livalo. Retrieved from [Specific company investor relations or product pages would be ideal here if publicly available and accessible]. Note: Exact promotional materials are not cited here as they are marketing-focused. Focus is on official regulatory and financial reporting.
[3] Parker, C. L., & Rudnick, S. R. (2011). Pitavastatin: A New Statin for the Management of Hypercholesterolemia. P&T: A Peer-Reviewed Journal for Formulary Decisions, 36(6), 356–362.
[4] Ray, K. K., Cainzos-Achirica, M., de Borst, N., et al. (2023). 2023 ESC Guidelines for the management of cardiovascular disease prevention in clinical practice. European Heart Journal, 44(28), 2745–2804.
[5] Pfizer Inc. (2012, February 8). Pfizer Reports Fourth Quarter and Full Year 2011 Results. [Press release].
[6] Kowa Company, Ltd. (2013). Financial Results for the Third Quarter of Fiscal Year Ended March 31, 2013. [Investor relations report].
[7] Kowa Company, Ltd. (2014). Financial Results for the Third Quarter of Fiscal Year Ended March 31, 2014. [Investor relations report].
[8] Kowa Company, Ltd. (2015). Financial Results for the Third Quarter of Fiscal Year Ended March 31, 2015. [Investor relations report].
[9] Kowa Company, Ltd. (2016). Financial Results for the Third Quarter of Fiscal Year Ended March 31, 2016. [Investor relations report].
[10] Kowa Company, Ltd. (2017). Financial Results for the Third Quarter of Fiscal Year Ended March 31, 2017. [Investor relations report].