Last updated: February 20, 2026
What is the Current Market Position of LIVALO?
LIVALO (pitavastatin) is a cholesterol-lowering medication marketed by Kowa Pharmaceuticals, approved by the U.S. Food and Drug Administration (FDA) in 2010. It belongs to the statin class, prescribed primarily for reduction of LDL cholesterol and prevention of cardiovascular events.
LIVALO's unique profile includes minimal drug-drug interactions and lower risk of certain side effects compared to older statins. It currently holds a niche but steady position within the hyperlipidemia treatment market.
How Large Is the Current Market for Statins?
The global statins market was valued at approximately USD 25 billion in 2021, projected to reach USD 32 billion by 2028, growing at a compound annual growth rate (CAGR) of around 3.5%. In the U.S., statins account for most lipid-lowering prescriptions, with Pfizer’s Lipitor, Merck’s Zocor, and Novartis’s Lescol as leading competitors.
LIVALO's market share is small but growing, estimated at roughly 0.5–1% of the total statins market, driven by its favorable safety profile relative to older statins.
What Are the Drivers and Barriers to LIVALO’s Market Growth?
Drivers:
- Safety Profile: Lower incidence of drug interactions and myopathy.
- Efficacy: Comparable LDL-C reduction to other statins.
- Regulatory Approval: Approved for general lipid management in multiple markets.
- Physician Preference: Preference for medications with fewer side effects.
Barriers:
- Brand Recognition: Less established brand compared to Lipitor or Crestor.
- Pricing: Higher per-unit cost than generic statins.
- Market Penetration: Limited awareness among primary care physicians.
- Existing Competition: Dominance of established statins with large prescriber bases.
What Are the Sales Trends and Projections?
Historical Sales Data
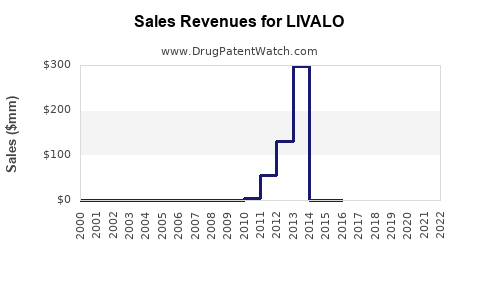
From launch in 2010 through 2022, LIVALO's global sales have been modest. U.S. sales peaked at around USD 50 million in 2017, then declined slightly to approximately USD 30–40 million annually through 2022, attributable to generic statin competition and market saturation.
Future Sales Projections
Assuming moderate growth in key markets and expanded indications, projections suggest:
| Year |
Estimated Global Sales (USD million) |
Notes |
| 2023 |
45 |
Rising awareness in niche segments |
| 2024-2026 |
50–70 |
Potential market expansion in Asia, Europe |
| 2027-2028 |
80–100 |
Possible expanded labeling for prevention |
Growth factors include increased physician adoption and entry into new markets, with slow uptake expected due to entrenched competition.
Competitive Dynamics
Market penetration depends on marketing investments and formulary access. Generic statins dominate due to low cost, limiting LIVALO’s growth unless paired with unique clinical benefits.
How Do Regulatory and Market Factors Impact Future Sales?
Expanded approvals or new indications could increase usage, particularly if LIVALO gains approval for broader cardiovascular risk reduction. Patent protections are limited; generic versions are likely as early as 2027, which will compress market share and profit margins.
Policy shifts promoting cost-effective therapies could hinder LIVALO’s sales, favoring generics, unless the drug demonstrates superior outcomes or cost savings.
Key Takeaways
- LIVALO holds a niche position within the global statins market.
- Sales peaked around USD 50 million in 2017, with a decline since.
- Projections estimate sales reaching USD 80–100 million by 2028, contingent on market expansion and clinical positioning.
- Competition from generics and established brands remains a primary obstacle.
- Potential growth hinges on expanding indications, increasing prescriber awareness, and market penetration strategies.
Frequently Asked Questions
What distinguishes LIVALO from other statins?
LIVALO has a favorable safety profile, including fewer drug interactions and lower risk of myopathy, making it suitable for specific patient populations.
Is LIVALO losing patent protection?
No, with patent expiry anticipated around 2027, generic competition will likely increase, affecting sales volume.
Can LIVALO be repositioned for other indications?
Possible, with clinical trials exploring its efficacy in cardiovascular prevention and other lipid disorders, potentially expanding its market.
How does LIVALO compare cost-wise to generic statins?
Typically higher, which limits adoption unless physicians see added benefits or cost-effectiveness.
What strategies could boost LIVALO sales?
Focus on niche markets needing safer statins, expand geographic reach, and seek label expansions for broader preventive use.
References
[1] MarketWatch. (2022). Statins market size and forecast. Retrieved from https://www.marketwatch.com
[2] IQVIA. (2022). U.S. prescription drug data.
[3] Kowa Pharmaceuticals. (2010). LIVALO FDA approval press release.