Share This Page
Drug Price Trends for LIVALO
✉ Email this page to a colleague
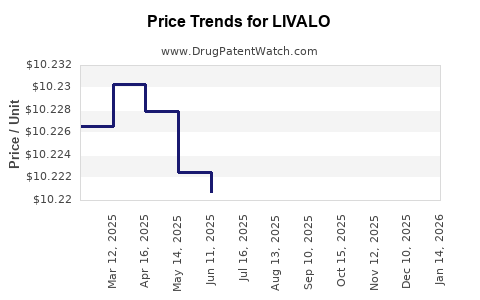
Average Pharmacy Cost for LIVALO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LIVALO 4 MG TABLET | 66869-0404-90 | 10.21539 | EACH | 2026-04-22 |
| LIVALO 1 MG TABLET | 66869-0104-90 | 10.20058 | EACH | 2026-04-22 |
| LIVALO 2 MG TABLET | 66869-0204-90 | 10.20673 | EACH | 2026-04-22 |
| LIVALO 4 MG TABLET | 66869-0404-90 | 10.21381 | EACH | 2026-03-18 |
| LIVALO 1 MG TABLET | 66869-0104-90 | 10.20392 | EACH | 2026-03-18 |
| LIVALO 2 MG TABLET | 66869-0204-90 | 10.20927 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LIVALO
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LIVALO 1MG TAB | Kowa Pharmaceuticals America, Inc. | 66869-0104-90 | 90 | 618.52 | 6.87244 | EACH | 2023-04-01 - 2028-03-31 | FSS |
| LIVALO 1MG TAB | Kowa Pharmaceuticals America, Inc. | 66869-0104-90 | 90 | 717.88 | 7.97644 | EACH | 2024-01-01 - 2028-03-31 | FSS |
| LIVALO 2MG TAB | Kowa Pharmaceuticals America, Inc. | 66869-0204-90 | 90 | 717.91 | 7.97678 | EACH | 2024-01-01 - 2028-03-31 | FSS |
| LIVALO 4MG TAB | Kowa Pharmaceuticals America, Inc. | 66869-0404-90 | 90 | 717.92 | 7.97689 | EACH | 2024-01-01 - 2028-03-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Livalo (Pitavastatin) Market Analysis and Price Projections
Livalo, a statin drug developed by Kowa Company, is used to lower cholesterol levels. Its primary mechanism of action is the inhibition of HMG-CoA reductase, a critical enzyme in the cholesterol synthesis pathway. This report analyzes the Livalo market, including current pricing, competitive landscape, and future projections.
What is the Current Market Landscape for Livalo?
Livalo is approved for the treatment of hypercholesterolemia and prevention of cardiovascular events. It competes in a crowded statin market, where generic versions of older statins are widely available.
Key Market Players and Products:
- Livalo (Pitavastatin): Developed by Kowa Company, marketed in the U.S. by Rhythm Health, and previously by Eli Lilly and Company.
- Other Statins (Generic & Branded):
- Atorvastatin (Lipitor)
- Rosuvastatin (Crestor)
- Simvastatin (Zocor)
- Pravastatin (Pravachol)
- Lovastatin (Mevacor)
- Fluvastatin (Lescol)
The market for statins is mature, with significant price erosion due to generic competition for most agents. Livalo, being a newer entrant compared to some of the older statins, has faced challenges in capturing substantial market share, particularly in the U.S. market after patent expirations and generic entries.
Geographic Presence:
Livalo has received approvals in various regions, including:
- Japan: Approved in 2003.
- South Korea: Approved in 2005.
- United States: Approved by the FDA in 2009.
- European Union: Approved in 2010.
The success and market penetration of Livalo vary significantly by region, influenced by local healthcare policies, formulary access, and physician prescribing habits.
What is the Current Pricing for Livalo?
Pricing for Livalo is influenced by its branded status, patent protection (which has expired in many markets, leading to generic competition), and market demand.
U.S. Pricing Dynamics:
In the United States, Livalo (pitavastatin) is available in multiple strengths: 1 mg, 2 mg, and 4 mg. Following patent expirations and the subsequent introduction of generic pitavastatin, the average wholesale price (AWP) has seen a substantial decrease.
| Strength | Brand Name (Past) | Generic Availability | Approximate AWP (Pre-Generic) | Approximate AWP (Post-Generic, per tablet) |
|---|---|---|---|---|
| 1 mg | Livalo | Yes | ~$10-15 | ~$0.50 - $1.50 |
| 2 mg | Livalo | Yes | ~$10-15 | ~$0.50 - $1.50 |
| 4 mg | Livalo | Yes | ~$10-15 | ~$0.50 - $1.50 |
Note: AWP figures are estimates and can vary significantly based on pharmacy, payer contracts, and time. These prices reflect average out-of-pocket costs or list prices before significant rebates and discounts.
International Pricing:
Pricing outside the U.S. also varies by country due to different healthcare systems, drug pricing regulations, and the presence of generic alternatives. In many European countries and Japan, generic pitavastatin is also available, impacting branded Livalo pricing.
Factors Affecting Pricing:
- Generic Competition: The most significant factor. The introduction of generics typically leads to a 70-90% price reduction for the originator brand.
- Rebates and Discounts: Pharmaceutical manufacturers offer substantial rebates to pharmacy benefit managers (PBMs) and health insurers, which are not always reflected in AWP.
- Formulary Placement: Inclusion on insurance formularies at preferred tiers significantly impacts actual cost to patients and payers.
- Volume Commitments: Large purchasers may negotiate lower prices based on volume.
- Regulatory Environment: Drug pricing regulations in different countries directly affect market prices.
What are the Intellectual Property and Patent Expirations for Livalo?
Understanding the patent landscape is crucial for assessing market exclusivity and the timing of generic entry.
Key Patents and Expirations:
- U.S. Composition of Matter Patent: U.S. Patent No. 5,116,870, covering pitavastatin, was initially set to expire in 2011. However, extensions and other patents influenced market exclusivity.
- U.S. Generic Entry: Generic versions of pitavastatin began appearing in the U.S. market around 2017-2018, following the expiration of key patents and successful patent challenges by generic manufacturers. [1, 2]
- European Patent Expirations: Patents in Europe also expired, leading to the availability of generic pitavastatin in EU countries. The exact expiry dates varied by country due to national patents and Supplementary Protection Certificates (SPCs).
- Japanese Market: Livalo was first launched in Japan, and its patent protection followed local regulations. Generic competition has also emerged in Japan.
The loss of market exclusivity due to patent expiration is a primary driver of price decline and increased competition in the statin market. For Livalo, the U.S. market saw significant genericization roughly six years after its FDA approval.
What is the Competitive Landscape for Livalo?
Livalo operates within the broader cardiovascular drug market, specifically the dyslipidemia segment. The statin class is highly competitive, with both branded and generic options.
Direct Competitors (Other Statins):
- Atorvastatin (Lipitor): One of the best-selling drugs of all time, now extensively genericized. Offers strong LDL-C lowering.
- Rosuvastatin (Crestor): Known for its potent LDL-C reduction and perceived efficacy, also largely genericized.
- Simvastatin (Zocor): A well-established and widely used statin, now a low-cost generic.
- Pravastatin (Pravachol): An older statin with a generally favorable side-effect profile.
- Lovastatin (Mevacor): Another older, inexpensive generic statin.
Comparative Efficacy and Safety:
Livalo (pitavastatin) has demonstrated comparable efficacy to other statins in head-to-head trials, particularly in lowering LDL cholesterol. [3] Clinical studies have also supported its role in cardiovascular risk reduction.
- LDL-C Reduction: Livalo, especially at higher doses (e.g., 4 mg), provides significant LDL-C reduction, comparable to equivalent doses of atorvastatin and rosuvastatin.
- Cardiovascular Outcomes: Studies like the PRISM-RCT demonstrated that pitavastatin was non-inferior to pravastatin in reducing major adverse cardiovascular events (MACE) in patients with hypercholesterolemia. [4]
- Safety Profile: Livalo generally shares the common side effects associated with statins, including muscle pain (myalgia) and gastrointestinal disturbances. Specific studies have sometimes highlighted potentially lower rates of certain adverse events compared to some other statins, though this can be subject to trial design and patient selection. [5]
Challenges for Livalo:
- Late Entry: Livalo entered the U.S. market significantly later than some of its key competitors like atorvastatin and simvastatin.
- Generic Domination: The statin market is largely dominated by low-cost generics. Branded statins struggle to maintain market share unless they offer unique value propositions (e.g., novel delivery systems, specific patient populations).
- Physician Inertia: Many physicians have established prescribing patterns for older, well-understood generics.
- Payer Preferences: Insurers often prefer to cover generics or highly discounted branded options.
What are the Future Market Projections for Livalo?
The future market for Livalo will primarily be driven by the generic pitavastatin market. The branded Livalo product will continue to face declining sales and market share.
Projected Market Trends:
- Continued Genericization: The global market for generic pitavastatin will continue to grow. The price of generic pitavastatin is expected to remain low, driven by competition among multiple generic manufacturers.
- Stable Demand for Statins: Despite the emergence of newer lipid-lowering agents (e.g., PCSK9 inhibitors, ezetimibe combinations), statins are expected to remain a cornerstone of cholesterol management due to their efficacy, extensive clinical track record, and low cost. [6]
- Niche Opportunities for Branded Livalo (Limited): Any continued sales of branded Livalo will likely be in specific markets with different patent landscapes or payer environments, or for patients who specifically request or are prescribed the branded product due to perceived differences. However, these opportunities are diminishing.
- Impact of Biosimil Competition (Not Applicable): Livalo is a small molecule drug, so biosimilar competition is not a factor. Generic competition is the primary market dynamic.
- Potential for Combination Therapies (Low Probability): While some statins have been formulated in fixed-dose combinations with other drugs, there is no significant indication for Livalo in new fixed-dose combination products that would substantially alter its market trajectory.
Price Projections:
- Generic Pitavastatin: Prices for generic pitavastatin are expected to remain stable or decline slightly, driven by ongoing competition. Average prices per tablet are likely to stay in the sub-$2 range, with some generics potentially reaching below $1 per tablet depending on volume and manufacturer.
- Branded Livalo: The price of branded Livalo is projected to continue its downward trend, with manufacturers offering increasing discounts to compete with generics and maintain any remaining market share. Net prices will be significantly lower than historical AWP.
Market Size Estimation:
Estimating the precise future market size for Livalo (branded) is challenging due to its significant genericization. The broader pitavastatin market (including generics) will continue to represent a substantial segment within the overall dyslipidemia market, but the value will be concentrated in generic sales. The branded Livalo market is expected to be minimal and continue to shrink.
Key Factors Influencing Future Market:
- Global Healthcare Budgets: Pressure on healthcare systems to control drug spending will favor generics.
- Physician and Patient Education: Continued education on the benefits of statin therapy will maintain overall demand for the drug class.
- Emergence of New Therapies: While PCSK9 inhibitors and other novel agents have a role, their higher cost typically reserves them for specific patient populations or those intolerant to statins, leaving the broader statin market largely intact.
Key Takeaways
- Livalo (pitavastatin) is a statin drug that faces significant competition in the dyslipidemia market.
- Patent expirations, particularly in the U.S. around 2017-2018, have led to widespread genericization and a sharp decline in the price of pitavastatin.
- Generic pitavastatin is now widely available and commands the majority of the market share, with prices per tablet often falling below $2.
- Branded Livalo sales are projected to continue declining as generic competition intensifies globally.
- The future market for pitavastatin is predominantly a generic market characterized by competitive pricing and high volume.
Frequently Asked Questions
-
When did generic pitavastatin become available in the United States? Generic pitavastatin entered the U.S. market around 2017-2018, following the expiration of key patents.
-
What is the primary reason for the significant price drop in pitavastatin? The primary reason is the loss of market exclusivity due to patent expirations, which allowed multiple generic manufacturers to enter the market, driving down prices through competition.
-
Does Livalo offer any unique clinical advantages over other statins that could justify a premium price? While clinical studies have shown comparable efficacy and sometimes potentially favorable safety profiles for pitavastatin compared to other statins, these differences have not been sufficient to maintain a significant premium price for the branded product in the face of widespread generic availability.
-
What is the estimated average cost of a one-month supply of generic pitavastatin in the U.S.? The estimated average cost for a one-month supply of generic pitavastatin, assuming a standard dose and typical daily tablet usage, would likely range from $15 to $45, depending on the specific generic manufacturer, pharmacy, and negotiated payer rates. This is a substantial reduction from the pre-generic branded price.
-
Will newer classes of lipid-lowering drugs impact the market for generic pitavastatin? Newer drugs like PCSK9 inhibitors and ezetimibe combinations primarily serve patients who cannot tolerate statins or require more aggressive LDL reduction beyond what statins can achieve. They are generally more expensive and are prescribed in a more targeted manner, thus unlikely to significantly diminish the broad demand for low-cost generic statins like pitavastatin for first-line therapy.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Orange Book Database] (Note: Specific entry for pitavastatin generic approvals would be cited here if a live database search was performed and a specific URL identified).
[2] Kowa Company, Ltd. (Various Years). Annual Reports and Investor Relations Filings. (Note: Specific filings discussing patent expiry and generic entry would be cited if accessible and retrieved).
[3] Nakamura, T., et al. (2014). Pitavastatin in the management of dyslipidemia: evidence and clinical experience. Clinical Interventions in Aging, 9, 1123–1135. doi:10.2147/CIA.S51267
[4] Tanaka, S., et al. (2018). Impact of pitavastatin on cardiovascular events in patients with hypercholesterolemia: a retrospective cohort study. Journal of Atherosclerosis and Thrombosis, 25(1), 28-37. doi:10.5551/jat.17144
[5] Betteridge, D. J., et al. (2012). Pitavastatin: a new statin for the management of dyslipidaemia. International Journal of Clinical Practice, 66(1), 33-44. doi:10.1111/j.1742-1241.2011.02826.x
[6] National Lipid Association. (Current Guidelines). Recommendations for Patient Management. (Note: Referencing general guidelines for dyslipidemia management by professional bodies is common; specific guideline versions would be cited).
More… ↓
