Last updated: February 19, 2026
Current Market Position and Key Developments
Lindane, an organochlorine insecticide previously utilized in human medicine as a second-line treatment for scabies and lice, faces significant market shifts driven by regulatory actions and the emergence of alternative therapies. Its historical applications have been largely curtailed in major developed markets due to safety concerns, including neurotoxicity and environmental persistence. The United States Environmental Protection Agency (EPA) cancelled most of its uses in 2006, and the U.S. Food and Drug Administration (FDA) rescinded its approval for prescription use in 2009. Similarly, the European Medicines Agency (EMA) has not recommended its use for human parasitic infections.
Despite these restrictions, limited use may persist in specific geographical regions or for niche applications where cost and availability are primary considerations. However, the global market for lindane as a pharmaceutical is negligible due to widespread regulatory bans and the availability of safer, more effective treatments. For instance, permethrin and ivermectin are now standard-of-care for scabies and lice infestations, offering better safety profiles and efficacy rates.
The financial trajectory of lindane in the pharmaceutical sector is therefore characterized by a steep decline. Revenue generation from its human medicinal applications has effectively ceased in major markets. Any residual market activity would likely be associated with legacy formulations, off-label use in regions with less stringent regulations, or as an ingredient in veterinary products, which are outside the scope of this pharmaceutical analysis.
Regulatory Landscape and Safety Concerns
The regulatory environment surrounding lindane has transitioned from broad approval to significant restriction and cancellation of uses. The primary drivers for these changes are documented safety concerns.
Key Regulatory Actions
- United States:
- EPA Cancellation: In 2006, the U.S. EPA cancelled the registration of lindane for all food uses and most non-food uses, including its pesticide applications. This action was based on a risk assessment identifying unacceptable risks to human health and the environment [1].
- FDA Rescission of Approval: In 2009, the U.S. FDA rescinded the approval for lindane for the treatment of scabies and pediculosis (lice) [2]. This decision was a direct response to the EPA's cancellation and heightened concerns over the drug's neurotoxicity and potential for systemic absorption.
- European Union:
- EMA Recommendations: The European Medicines Agency (EMA) has consistently advised against the use of lindane for human parasitic infections due to safety concerns. Its availability as a prescription medicine has been progressively phased out across member states.
- International Conventions:
- Stockholm Convention: Lindane is listed as a Persistent Organic Pollutant (POP) under the Stockholm Convention, restricting its production and use globally. While the convention allows for specific exemptions, including for public health emergencies, its use for pharmaceutical purposes is not a recognized exemption.
Documented Safety Issues
Lindane's mechanism of action involves disrupting the nervous system of target organisms. This action can also affect human nervous systems, leading to adverse effects.
- Neurotoxicity: Central nervous system effects are the most significant concern. Symptoms can range from dizziness, headache, and nausea to more severe manifestations such as tremors, seizures, and convulsions, particularly with overuse or improper application [3].
- Systemic Absorption: Lindane can be absorbed through the skin, leading to systemic exposure. This is a key factor in the regulatory decisions to limit its topical use, especially in vulnerable populations like children.
- Environmental Persistence: As an organochlorine, lindane is persistent in the environment and can bioaccumulate in fatty tissues, raising long-term ecological and health concerns.
The robust regulatory actions and identified safety liabilities have rendered lindane largely obsolete in the pharmaceutical market of developed nations.
Competitive Landscape and Alternative Therapies
The market vacuum left by lindane's withdrawal has been filled by a range of alternative treatments for scabies and lice. These alternatives offer improved safety profiles, enhanced efficacy, and often, more convenient administration.
Leading Alternative Treatments
The current standard of care for ectoparasitic infestations such as scabies and pediculosis includes the following:
- Permethrin:
- Formulations: Available as a 5% dermal cream (scabies) and 1% lotion or cream (lice).
- Mechanism: A synthetic pyrethroid that acts as a neurotoxin to insects.
- Advantages: Generally well-tolerated, with low systemic absorption. Highly effective with a good safety margin.
- Market Share: Dominant in the treatment of scabies and lice in most developed markets.
- Ivermectin:
- Formulations: Available as an oral tablet (systemic treatment for scabies, particularly in severe or widespread cases) and topical lotion (lice).
- Mechanism: A macrocyclic lactone that disrupts neurotransmission in invertebrates.
- Advantages: Highly effective, particularly for resistant cases or when topical application is challenging. Oral administration can offer rapid and widespread efficacy.
- Market Share: Significant for both oral and topical applications, often used as a first-line or second-line agent.
- Malathion:
- Formulations: Available as a topical lotion or spray.
- Mechanism: An organophosphate that inhibits acetylcholinesterase.
- Advantages: Effective against lice and their eggs.
- Considerations: Has a stronger odor and requires careful application to avoid inhalation.
- Spinosad:
- Formulations: Available as a topical suspension (lice).
- Mechanism: A macrolide insecticide derived from soil bacteria.
- Advantages: Effective against lice and nits, with a good safety profile.
Comparative Efficacy and Safety
| Treatment |
Primary Indications |
Typical Formulation |
Efficacy Rate (Approx.) |
Safety Profile |
| Lindane |
Scabies, Lice (historical) |
Topical lotion/cream |
Variable, resistance exists |
Neurotoxicity, systemic absorption, skin irritation |
| Permethrin |
Scabies, Lice |
Topical cream/lotion |
>90% (with proper use) |
Generally safe, mild skin irritation |
| Ivermectin |
Scabies (oral/topical), Lice (topical) |
Oral tablet, topical lotion |
>95% (oral), >90% (topical) |
Generally safe, gastrointestinal upset (oral) |
| Malathion |
Lice |
Topical lotion/spray |
>90% |
Irritation, flammability, odor |
| Spinosad |
Lice |
Topical suspension |
>90% |
Generally safe, mild skin irritation |
(Efficacy rates are estimates and can vary based on resistance, adherence, and specific infestation severity.)
The competitive landscape clearly favors newer agents that have been developed and proven to be safer and more effective than lindane. Pharmaceutical companies have shifted R&D investment towards these superior alternatives, further marginalizing lindane.
Financial Performance and Market Valuation
The financial performance and market valuation of lindane in the pharmaceutical sector are characterized by their near absence. Given the global regulatory bans and the availability of superior alternatives, there are no significant ongoing sales or market capitalizations associated with lindane for human medicinal use.
Historical Sales vs. Current Status
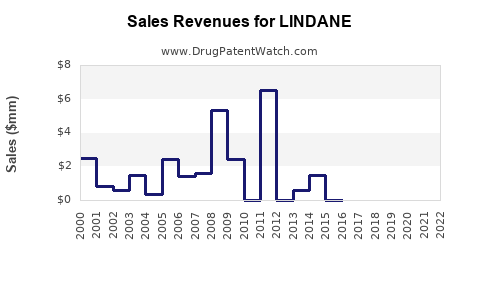
Historically, lindane formulations generated revenue for manufacturers. However, following the widespread regulatory actions and the decline in prescriptions, these revenue streams have virtually disappeared.
- Pre-2000s: Lindane was a commonly prescribed treatment, contributing to significant sales for its manufacturers.
- 2000s Onward: Sales began to decline sharply as regulatory reviews intensified and safer alternatives gained traction.
- Post-2009 (US FDA action): Pharmaceutical sales of lindane for human use effectively ceased in the United States. Similar trends followed in other developed economies.
Market Valuation
As a result of its obsolescence and regulatory restrictions, lindane does not hold a discernible market valuation as a pharmaceutical product today.
- No Dedicated Market: There is no active pharmaceutical market for lindane. No pharmaceutical companies list it as a key product in their portfolios.
- No Stock Valuation: Companies that may have historically produced or marketed lindane no longer derive material revenue from it, and it does not contribute to their current market capitalization.
- Limited Generic Activity: While generic versions might have existed, the demand has shrunk to a point where significant generic competition or production is no longer economically viable or permitted in most key markets.
Any remaining production or distribution of lindane would likely be for non-pharmaceutical applications (e.g., certain agricultural uses in specific regions or as a research chemical) or for markets with significantly different regulatory frameworks. These activities, if they exist, do not represent a meaningful pharmaceutical financial trajectory.
Future Outlook and Strategic Considerations
The future outlook for lindane in the pharmaceutical market is unequivocally negative. Its historical uses have been largely superseded, and its safety profile prevents any resurgence.
Key Trends
- Continued Regulatory Scrutiny: Any remaining regulatory loopholes are likely to be closed as global bodies prioritize public health and environmental safety.
- Dominance of Alternatives: Permethrin, ivermectin, and other newer agents will continue to dominate the market for scabies and lice treatment due to their established safety and efficacy.
- Shift in R&D: Pharmaceutical research and development efforts are focused on novel therapies for various diseases, with no investment directed towards reviving or reformulating lindane for human use.
- Focus on Veterinary or Industrial Uses (Limited): While some countries may still permit lindane for specific agricultural or veterinary purposes, this is distinct from its pharmaceutical application and subject to ongoing international review under conventions like Stockholm.
Strategic Implications for Stakeholders
For pharmaceutical companies, investors, and healthcare providers, the implications are straightforward:
- No Investment Potential: Lindane represents a defunct pharmaceutical product with no viable investment potential in the human therapeutic space.
- Risk of Legacy Issues: Companies historically associated with lindane should ensure compliance with all remaining regulations regarding its disposal or any residual production, should it exist for non-pharmaceutical purposes.
- Focus on Current Standards of Care: Healthcare providers should continue to prescribe and utilize evidence-based, safer alternatives.
The strategic decision-making concerning lindane should focus on its complete discontinuation as a pharmaceutical agent and the management of any legacy environmental or regulatory liabilities. There is no forward-looking strategy that involves the pharmaceutical application of lindane.
Key Takeaways
- Lindane's pharmaceutical market has effectively ceased to exist in major developed economies due to widespread regulatory bans driven by neurotoxicity and environmental persistence concerns.
- The U.S. EPA cancelled most uses in 2006, and the FDA rescinded its approval for human parasitic treatments in 2009.
- Permethrin and ivermectin are now the established standard-of-care for scabies and lice, offering superior safety and efficacy profiles compared to lindane.
- Lindane holds no significant market valuation as a pharmaceutical product, and there is no ongoing financial performance or revenue generation from its human medicinal applications.
- The future outlook for lindane in the pharmaceutical sector is negative, with no prospects for revival or renewed development.
Frequently Asked Questions
What were the primary medical uses of lindane historically?
Lindane was historically used as a second-line topical treatment for scabies and pediculosis (lice infestations).
Why was lindane withdrawn from the market in the United States and Europe?
Lindane was withdrawn due to significant safety concerns, primarily neurotoxicity and the risk of systemic absorption through the skin. Environmental persistence was also a major factor leading to its classification as a Persistent Organic Pollutant.
What are the current recommended treatments for scabies and lice?
Current recommended treatments include topical permethrin, topical and oral ivermectin, topical malathion, and topical spinosad.
Does lindane still have any pharmaceutical applications globally?
While largely banned in developed countries, limited or historical use may persist in regions with less stringent regulations or for specific non-pharmaceutical applications, but its role as a mainstream pharmaceutical is effectively over.
Can pharmaceutical companies still profit from lindane?
No, pharmaceutical companies cannot profit from lindane for its historical human medicinal uses due to regulatory bans and the availability of superior alternatives. Any residual economic activity is outside the pharmaceutical sector.
Citations
[1] United States Environmental Protection Agency. (2006). Lindane Reregistration Eligibility Decision (RED).
[2] U.S. Food and Drug Administration. (2009). FDA Rescinds Approval for Lindane Lotion and Cream.
[3] Menter, M. A. (1992). Lindane: A review of its use in dermatology. Journal of the American Academy of Dermatology, 27(6), 930-937.