Last updated: February 17, 2026
What Are the Market Dynamics for KLONOPIN?
KLONOPIN (clonazepam) is a benzodiazepine primarily used for epilepsy, panic disorder, and movement disorders. Its market environment is influenced by several factors:
- Regulatory status: Approved by the FDA since 1975, it faces tight controls due to abuse potential. Rescheduling or tighter restrictions can limit market access.
- Patent landscape: Loses patent protection, allowing generic manufacturing, which depresses per-unit pricing but expands market volume.
- Competitive landscape: Competes with other benzodiazepines (e.g., diazepam, lorazepam) and newer treatments for epilepsy and anxiety (e.g., lacosamide).
- Medical demand: Consistent use in epilepsy management sustains demand, though off-label use for other conditions influences sales.
- Prescriber trends: Preference for newer, non-controlled medications in some markets could reduce prescription volume.
- Supply chain factors: Manufacturing constraints and regulatory compliance impact availability and pricing.
How Does the Financial Trajectory for KLONOPIN Evolve?
The financial outlook for KLONOPIN is shaped by:
| Aspect |
Current Situation |
Trends |
Outlook |
| Revenue |
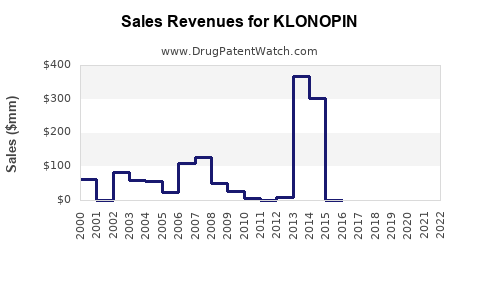
Declined post-patent expiry; generic sales dominate |
Marginal growth expected if off-label uses expand |
Slight recovery possible if off-label use for certain indications increases, and new formulations are introduced. |
| Pricing |
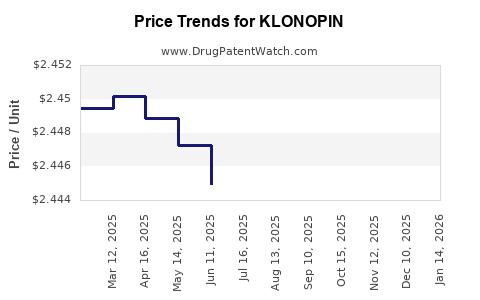
Lowered due to generics and high competition |
Stable but under pressure |
Marginal decreases unless branded versions regain pricing power. |
| Market volume |
Remains stable due to epilepsy's chronic nature |
Slight increase or decrease based on prescriber preferences |
Expect stable to slight growth in volume tied to epilepsy prevalence. |
| Regulatory impact |
Regulatory controls for benzodiazepines tighten in some regions |
Possible restrictions on treatment duration, prescription limits |
Could suppress sales if restrictions intensify. |
What Are the Main Drivers and Risks?
Drivers:
- Growing prevalence of epilepsy and anxiety disorders increases overall demand.
- Expansion into emerging markets, where benzodiazepines face fewer restrictions, offers growth potential.
- Development of new formulations or delivery methods that improve compliance.
Risks:
- Increased regulation due to abuse potential could lead to reduced prescriptions.
- Access to newer, non-controlled alternatives may shift prescriber preferences away from benzodiazepines.
- Legal or reputational issues linked to misuse or diversion of clonazepam.
How Do Global Policies Impact KLONOPIN?
- United States: FDA classifies clonazepam as a controlled substance Schedule IV; recent policies focus on prescription monitoring and abuse prevention.
- European Union: Benzodiazepines face stricter prescribing guidelines; some countries recommend short-term use only.
- Emerging markets: Less restrictive policies may permit higher sales volume but pose regulatory risks in the future.
What Are the Key Market Segments?
- Epilepsy: Core use, provides stable sales due to chronic treatment requirements.
- Anxiety disorders: Off-label use, variable among regions.
- Movement disorders and panic disorder: Smaller segments, with prescriptions influenced by prescriber preferences and guidelines.
What Are the Opportunities for Growth?
- Generic competition: Price reductions have expanded access, especially in cost-sensitive markets.
- Combination therapies: Co-administration with other anticonvulsants may create niche markets.
- Formulation innovation: Extended-release versions could improve compliance and open new market segments.
What Is the Forecast for the Next Five Years?
- The market for KLONOPIN is expected to witness slow growth, driven by epilepsy prevalence and new markets.
- Revenue projections for generic clonazepam are expected to remain flat or slightly decline, depending on regulatory restrictions.
- Opportunities for branded formulations or delivery systems may provide incremental revenue growth, but market share gains are uncertain.
Key Takeaways
- KLONOPIN's market is under pressure from generics, regulatory restrictions, and competition from newer medications.
- Long-term demand for epilepsy treatment sustains baseline revenue levels.
- Growth prospects depend on market penetration in emerging economies and medical innovation.
- Regulatory shifts and prescriber preferences are primary risks.
- Diversification into alternative formulations or combinations could offer incremental advantages.
FAQs
1. How significant is the patent loss for KLONOPIN?
The patent expired in the US in 2008, leading to widespread generic competition, which dramatically reduced brand-name sales and increased volume for generics.
2. Are there new formulations of KLONOPIN in development?
No recent FDA-approved new formulations are publicly known. Focus remains on existing formulations and expanding indications.
3. How does regulation influence KLONOPIN's sales globally?
In high-regulation countries like the US and EU, prescription restrictions limit sales growth. Less regulated markets may see higher use but pose future regulatory risks.
4. Can KLONOPIN regain pricing power?
Unlikely unless branded formulations introduce differentiation or new delivery methods that justify higher prices.
5. What is the impact of off-label use on KLONOPIN's market?
Off-label prescribing supports additional revenue streams but complicates regulatory and reimbursement landscapes, risking future restrictions.
References
[1] U.S. Food and Drug Administration (FDA). Clonazepam Label.
[2] IQVIA. Global Markets Report, 2022.
[3] European Medicines Agency (EMA). Benzodiazepines Regulatory Guidelines, 2021.
[4] Pharmaprojects. Clonazepam Development and Sales Data, 2022.
[5] World Health Organization (WHO). Epilepsy Prevalence and Treatment Guidelines, 2022.