JANUMET XR Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Janumet Xr, and when can generic versions of Janumet Xr launch?
Janumet Xr is a drug marketed by Msd Sub Merck and is included in one NDA. There is one patent protecting this drug and two Paragraph IV challenges.
This drug has fifty-two patent family members in forty countries.
The generic ingredient in JANUMET XR is metformin hydrochloride; sitagliptin phosphate. There are forty-nine drug master file entries for this compound. Three suppliers are listed for this compound. Additional details are available on the metformin hydrochloride; sitagliptin phosphate profile page.
DrugPatentWatch® Generic Entry Outlook for Janumet Xr
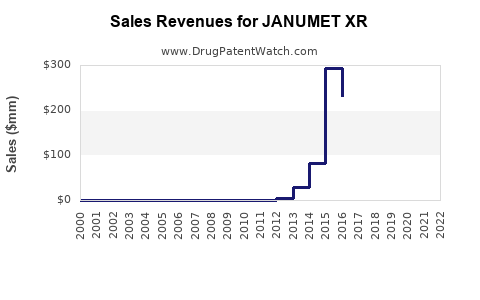
Annual sales in 2022 were $352mm indicating the motivation for generic entry (peak sales were $1.1bn in 2018).
There have been thirty-three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for JANUMET XR?
- What are the global sales for JANUMET XR?
- What is Average Wholesale Price for JANUMET XR?
Summary for JANUMET XR
| International Patents: | 52 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 8 |
| Clinical Trials: | 20 |
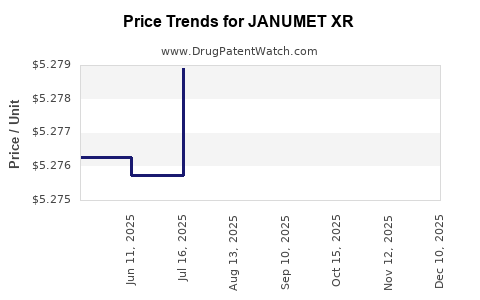
| Drug Prices: | Drug price information for JANUMET XR |
| Drug Sales Revenues: | Drug sales revenues for JANUMET XR |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for JANUMET XR |
| What excipients (inactive ingredients) are in JANUMET XR? | JANUMET XR excipients list |
| DailyMed Link: | JANUMET XR at DailyMed |
Recent Clinical Trials for JANUMET XR
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Galenicum Health | Phase 1 |
| Eva Pharma | Phase 1 |
| Genuine Research Center, Egypt | Phase 1 |
Pharmacology for JANUMET XR
| Drug Class | Biguanide Dipeptidyl Peptidase 4 Inhibitor |
| Mechanism of Action | Dipeptidyl Peptidase 4 Inhibitors |
Paragraph IV (Patent) Challenges for JANUMET XR
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| JANUMET XR | Extended-release Tablets | metformin hydrochloride; sitagliptin phosphate | 100 mg/1000 mg | 202270 | 1 | 2012-10-22 |
| JANUMET XR | Extended-release Tablets | metformin hydrochloride; sitagliptin phosphate | 50 mg/500 mg and 50 mg/1000 mg | 202270 | 1 | 2012-03-16 |
US Patents and Regulatory Information for JANUMET XR
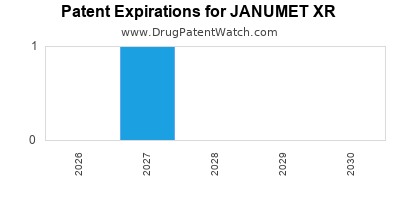
JANUMET XR is protected by one US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msd Sub Merck | JANUMET XR | metformin hydrochloride; sitagliptin phosphate | TABLET, EXTENDED RELEASE;ORAL | 202270-002 | Feb 2, 2012 | RX | Yes | No | 7,326,708*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Msd Sub Merck | JANUMET XR | metformin hydrochloride; sitagliptin phosphate | TABLET, EXTENDED RELEASE;ORAL | 202270-003 | Feb 2, 2012 | RX | Yes | Yes | 7,326,708*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Msd Sub Merck | JANUMET XR | metformin hydrochloride; sitagliptin phosphate | TABLET, EXTENDED RELEASE;ORAL | 202270-001 | Feb 2, 2012 | RX | Yes | No | 7,326,708*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for JANUMET XR
International Patents for JANUMET XR
See the table below for patents covering JANUMET XR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Bulgaria | 108493 | ⤷ Start Trial | |
| Montenegro | 00439 | BETA-AMINO TETRAHIDROMIDAZO (1,2-A) PIRAZINI I TETRAHIDROTRIAZOLO (4,3-A) PIRAZINI KAO INHIBITORI DIPEPTIDIL PEPTIDAZE U LIJEČENJU ILI PREVENCIJI DIJABETESA (BETA-AMINO TETRAHYDROIMIDAZO (1, 2-A) PYRAZINES AND TETRAHYDROTRIOAZOLO (4, 3-A) PYRAZINES AS DIPEPTIDYL PEPTIDASE INHIBITORS FOR THE TREATMENT OR PREVENTION OF DIABETES) | ⤷ Start Trial |
| Cyprus | 1106936 | ⤷ Start Trial | |
| Luxembourg | 91382 | ⤷ Start Trial | |
| Spain | 2505665 | ⤷ Start Trial | |
| Georgia, Republic of | P20053734 | Beta-Amino Tetrahydroimidazo (1, 2-A)Pyrazines And Tetrahydrotrioazolo (4, 3-A)Pyrazines as Dipeptidyl Peptidase Inhibitors for Treatment or Prevention of Diabetes | ⤷ Start Trial |
| Hong Kong | 1095144 | PHOSPHORIC ACID SALT OF A DIPEPTIDYL PEPTIDASE-IV INHIBITOR | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for JANUMET XR
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2498758 | PA2020003,C2498758 | Lithuania | ⤷ Start Trial | PRODUCT NAME: METFORMINO HIDROCHLORIDAS; SAKSAGLIPTINAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; DAPAGLIFLOZINAS ARBA FARMACINIU POZIURIU PRIIMTINAS JO SOLVATAS; REGISTRATION NO/DATE: EU/1/19/1401 20191111 |
| 1084705 | C300707 | Netherlands | ⤷ Start Trial | PRODUCT NAME: SAXAGLIPTIN; REGISTRATION NO/DATE: EU/1/09/545/001-010 20091001 |
| 1506211 | C300677 | Netherlands | ⤷ Start Trial | PRODUCT NAME: COMBINATIE VAN DAPAGLIFLOZINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN EN METFORMINE OF EEN FARMACEITISCH AANVAARDBAAR ZOUT DAARVAN, ZOALS BESCHERMD DOOR HET BASISOCTROOI EP 1506211B1; REGISTRATION NO/DATE: EU/1/13/900 20140121 |
| 2498758 | PA2020003 | Lithuania | ⤷ Start Trial | PRODUCT NAME: METFORMINAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; SAKSAGLIPTINAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; DAPAGLIFLOZINAS ARBA FARMACINIU POZIURIU PRIIMTINAS JO SOLVATAS; REGISTRATION NO/DATE: EU/1/19/1401 20191111 |
| 0896538 | 300308 | Netherlands | ⤷ Start Trial | 300308, 20170424, EXPIRES: 20220423 |
| 1412357 | SPC/GB07/046 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: SITAGLIPTIN PHOSPHATE MONOHYDRATE; REGISTERED: UK EU/1/07/383/001 20070323; UK EU/1/07/383/002 20070323; UK EU/1/07/383/003 20070323; UK EU/1/07/383/004 20070323; UK EU/1/07/383/005 20070323; UK EU/1/07/383/006 20070323; UK EU/1/07/383/007 20070323; UK EU/1/07/383/008 20070323; UK EU/1/07/383/009 20070323; UK EU/1/07/383/010 20070323; UK EU/1/07/383/011 20070323; UK EU/1/07/383/012 20070323; UK EU/1/07/383/013 20070323; UK EU/1/07/383/014 20070323; UK EU/1/07/383/015 20070323; UK EU/1/07/383/016 20070323; UK EU/1/07/383/017 20070323; UK EU/1/07/383/018 20070323 |
| 1084705 | C01084705/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: SITAGLIPTIN; REGISTRATION NO/DATE: SWISSMEDIC 57863 18.04.2007 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for JANUMET XR
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.