Last updated: February 19, 2026
Itraconazole, an azole antifungal medication, demonstrates a mature market position with sustained demand driven by its efficacy against a spectrum of fungal infections. Generic competition has intensified, impacting pricing and revenue growth. Key markets include infectious disease treatment and prophylaxis in immunocompromised patients. The drug’s long-standing approval and established safety profile underpin its continued clinical utility, though newer antifungals present competition in specific indications.
What are the primary fungal infections treated by itraconazole?
Itraconazole addresses a broad range of fungal infections. It is indicated for the treatment of blastomycosis, histoplasmosis, and coccidioidomycosis [1]. It also treats aspergillosis when other treatments are ineffective. For nail fungus (onychomycosis), itraconazole is used for superficial fungal infections of the skin, including ringworm (tinea corporis), jock itch (tinea cruris), and athlete's foot (tinea pedis) [1]. The drug is also employed for the treatment of vaginal candidiasis and oral thrush (oropharyngeal candidiasis) [1].
What is the mechanism of action for itraconazole?
Itraconazole functions by inhibiting the synthesis of ergosterol, a critical component of fungal cell membranes. Specifically, it binds to and inhibits the fungal cytochrome P450 enzyme 14α-demethylase. This enzyme is responsible for converting lanosterol to ergosterol. Disruption of ergosterol synthesis leads to increased cell membrane permeability, leakage of cellular contents, and ultimately fungal cell death [2].
How has the market landscape for itraconazole evolved?
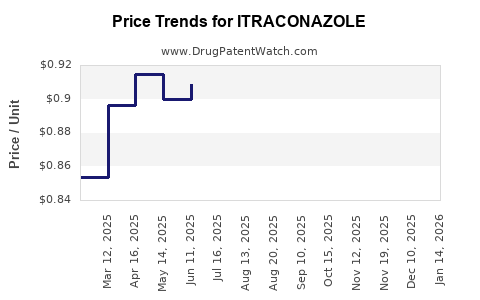
The market for itraconazole has matured significantly since its initial approval. The drug is available globally through multiple generic manufacturers, leading to substantial price erosion and a highly competitive environment. This competitive landscape is characterized by a focus on cost-effectiveness and accessibility, particularly in emerging markets. While originator product sales have declined, the overall volume of itraconazole prescriptions remains stable due to its established role in treating common and severe fungal infections.
What is the global market size and projected growth for itraconazole?
Quantifying the precise global market size for itraconazole as a standalone product is challenging due to its widespread generic availability and inclusion in broader antifungal market reports. However, industry analyses place the global antifungal drug market, of which itraconazole is a component, at approximately $15 billion in 2023, with an anticipated compound annual growth rate (CAGR) of around 4-6% over the next five to seven years [3]. The growth is driven by the increasing incidence of invasive fungal infections, particularly in immunocompromised populations such as cancer patients undergoing chemotherapy and organ transplant recipients. Itraconazole’s market share within this segment is subject to the competitive pressures of other established and newer antifungals.
Who are the key manufacturers and suppliers of itraconazole?
The manufacturing and supply of itraconazole are dominated by generic pharmaceutical companies. Major global suppliers include, but are not limited to:
- Janssen Pharmaceuticals (a Johnson & Johnson company): Original developer of Sporanox.
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cipla Ltd.
- Aurobindo Pharma Ltd.
- Zydus Lifesciences Ltd.
These companies produce both active pharmaceutical ingredients (APIs) and finished dosage forms for various international markets. The market is characterized by a fragmented supplier base, with numerous regional and national players also contributing to the supply chain.
What are the key regulatory approvals and market access considerations for itraconazole?
Itraconazole has received regulatory approval from major health authorities worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). The original New Drug Application (NDA) for Sporanox was approved by the FDA in 1992 [4]. Subsequent generic approvals have expanded market access.
Market access is influenced by:
- Reimbursement policies: Payer coverage varies by region and specific indication, impacting out-of-pocket costs for patients and prescribing patterns.
- Therapeutic guidelines: Inclusion in clinical practice guidelines for antifungal therapy reinforces its use, while newer agents may be prioritized for specific severe infections.
- Formulation availability: Different dosage forms (capsules, oral solution, intravenous) cater to varying clinical needs and patient populations.
- Post-marketing surveillance: Ongoing monitoring of safety and efficacy data can influence prescribing habits and regulatory scrutiny.
What are the major drivers of demand for itraconazole?
The primary drivers of demand for itraconazole are:
- Rising incidence of fungal infections: An increase in invasive fungal infections, particularly among immunocompromised individuals due to conditions like HIV/AIDS, cancer chemotherapy, and organ transplantation, sustains demand for effective antifungals.
- Established clinical efficacy: Itraconazole has a proven track record of efficacy against a wide spectrum of fungal pathogens, making it a reliable treatment option for healthcare providers.
- Cost-effectiveness of generic versions: The availability of affordable generic itraconazole makes it a preferred choice in resource-limited settings and for patients requiring long-term treatment.
- Broad-spectrum activity: Its effectiveness against both endemic mycoses (e.g., histoplasmosis, blastomycosis) and opportunistic infections (e.g., Candida, Aspergillus) broadens its applicability.
- Prophylactic use: In certain high-risk patient groups, itraconazole is used for the prevention of fungal infections, contributing to consistent demand.
What are the significant challenges and risks facing the itraconazole market?
The itraconazole market faces several significant challenges:
- Intense generic competition and price erosion: The presence of numerous generic manufacturers has led to significant downward pressure on prices, impacting profitability for all market participants.
- Emergence of newer antifungals: Advanced antifungal agents, such as echinocandins and newer triazoles (e.g., posaconazole, voriconazole), offer improved efficacy, safety profiles, or activity against resistant strains, posing a competitive threat, particularly for severe or refractory infections.
- Drug interactions: Itraconazole is a potent inhibitor of cytochrome P450 3A4 (CYP3A4), leading to a high potential for clinically significant drug interactions with numerous concomitant medications, requiring careful patient management and limiting its use in polypharmacy patients.
- Pharmacokinetic variability: Absorption can be influenced by food intake, and inter-individual variability in pharmacokinetics can affect treatment outcomes, necessitating dose adjustments and monitoring.
- Adverse event profile: Potential side effects, including gastrointestinal disturbances, hepatotoxicity, and cardiovascular effects (e.g., QT prolongation), can limit its use or necessitate close monitoring.
- Antifungal resistance: The development of resistance to azole antifungals, including itraconazole, by certain fungal pathogens is an evolving concern that may impact future treatment choices.
What is the financial trajectory and outlook for itraconazole?
The financial trajectory for itraconazole is characterized by stable but low-growth revenue streams, primarily driven by generic sales. The era of significant revenue growth from the originator product is long past. The market is highly price-sensitive, and profit margins for individual manufacturers are generally modest, relying on high-volume sales.
Key financial aspects include:
- Revenue: Primarily derived from generic formulations. Total revenue for itraconazole-containing products globally is spread across numerous companies and difficult to isolate precisely but contributes to the broader antifungal market's financial performance.
- Profitability: Margins are tight due to intense competition and the commoditized nature of generic drugs. Companies focus on operational efficiency and supply chain management to maintain profitability.
- Investment: Significant investment in novel antifungal research and development continues, focusing on addressing resistance, improving safety profiles, and treating rare or emerging fungal threats. Investment in itraconazole itself is minimal, focused on maintaining existing manufacturing capabilities and regulatory compliance.
- Market Share: While originator sales are nominal, generic itraconazole holds a substantial share in specific market segments, particularly for less severe or chronic fungal infections where cost is a primary consideration.
The outlook for itraconazole is one of continued relevance as a cost-effective, broad-spectrum antifungal. However, significant revenue growth is unlikely. Its financial trajectory will remain tied to the broader antifungal market dynamics, influenced by the prevalence of fungal infections and the competitive landscape shaped by newer, more advanced therapies.
What is the competitive landscape of antifungal agents that impacts itraconazole?
The competitive landscape for itraconazole is multi-faceted, encompassing other azoles, echinocandins, polyenes, and newer agents.
- Other Triazoles:
- Fluconazole: Widely used, particularly for Candida infections. Generally has a more favorable drug interaction profile and better absorption than itraconazole.
- Voriconazole: Preferred for serious Aspergillus infections and has a broader spectrum against Candida species, including some resistant strains.
- Posaconazole: Effective against a wider range of molds, including Zygomycetes, and is used for prophylaxis in high-risk patients.
- Isavuconazole: A newer triazole with a broad spectrum and improved safety profile compared to some older agents.
- Echinocandins (e.g., Caspofungin, Micafungin, Anidulafungin): These agents target a different pathway (cell wall synthesis) and are particularly useful for invasive Candida infections, especially in critically ill patients or those with azole resistance. They generally have fewer drug interactions.
- Polyenes (e.g., Amphotericin B, Liposomal Amphotericin B): Amphotericin B is a broad-spectrum agent but is associated with significant toxicity, especially renal. Liposomal formulations offer improved safety and are used for severe, life-threatening fungal infections.
- Other agents: Griseofulvin (for dermatophytes), Flucytosine (often used in combination).
Itraconazole maintains its competitive position due to its established efficacy in specific endemic mycoses and its lower cost compared to many newer agents. However, in cases of severe infections, azole-resistant pathogens, or patients with significant drug interaction concerns, newer agents are often favored.
What are the key therapeutic areas where itraconazole remains a treatment of choice?
Despite newer advancements, itraconazole remains a treatment of choice in several key therapeutic areas:
- Endemic Mycoses: It is a primary treatment for blastomycosis, histoplasmosis, and coccidioidomycosis [1]. Its efficacy and long-standing use make it the standard of care for many cases of these infections.
- Onychomycosis: While topical treatments are often tried first, itraconazole is a systemic option for severe or widespread nail fungal infections, offering a pulsatile therapy regimen that can be convenient.
- Superficial Dermatophytoses: For extensive or recalcitrant cases of ringworm, jock itch, and athlete's foot, oral itraconazole provides a highly effective solution.
- Oropharyngeal and Esophageal Candidiasis: In patients unable to tolerate or respond to fluconazole, itraconazole is a viable alternative, particularly the oral solution formulation.
- Prophylaxis and Treatment in Specific Populations: In certain endemic regions, it may be used for prophylaxis against specific fungal diseases, and it continues to be a valuable option for treating less severe or chronic fungal infections in various patient groups, especially when cost is a significant factor.
Key Takeaways
- Itraconazole is an established antifungal with a broad spectrum of activity, primarily treating endemic mycoses, onychomycosis, and superficial dermatophytoses.
- The market is dominated by generic competition, leading to price erosion and stable but low revenue growth.
- Key drivers include rising fungal infection rates, particularly in immunocompromised populations, and the cost-effectiveness of generic formulations.
- Significant challenges include intense generic competition, the emergence of newer antifungals with improved profiles, and a high potential for drug interactions.
- Despite competition, itraconazole retains its status as a treatment of choice for endemic mycoses and certain superficial fungal infections due to its proven efficacy and affordability.
- The financial trajectory is characterized by mature, volume-driven generic sales rather than growth, with profitability dependent on operational efficiency.
Frequently Asked Questions
- What is the typical duration of itraconazole treatment for onychomycosis?
Treatment duration for onychomycosis typically ranges from 3 to 12 months, depending on the severity of the infection and the specific regimen prescribed.
- Are there any specific contraindications for itraconazole use?
Itraconazole is contraindicated in patients with a known hypersensitivity to the drug or its excipients, and in patients with heart failure. It should also not be used concurrently with certain medications that are extensively metabolized by CYP3A4, due to the risk of severe drug interactions.
- How does itraconazole absorption differ between capsule and oral solution formulations?
The oral solution formulation of itraconazole demonstrates better bioavailability compared to the capsule formulation, particularly when taken without food. Capsules should ideally be taken with food to enhance absorption, while the oral solution can be taken with or without food.
- What are the main signs and symptoms of hepatotoxicity associated with itraconazole?
Signs and symptoms of hepatotoxicity may include jaundice, dark urine, pale stools, nausea, vomiting, abdominal pain, unusual fatigue, and loss of appetite. Patients experiencing these symptoms should seek immediate medical attention.
- Can itraconazole be used during pregnancy?
Itraconazole is generally not recommended for use during pregnancy, particularly in the first trimester, due to potential risks to the fetus. Its use in pregnant women should be carefully weighed against the potential benefits.
Citations
[1] Food and Drug Administration. (n.d.). Prescribing Information: Sporanox (itraconazole). U.S. Department of Health and Human Services.
[2] Sams, C. E. (2022). Antifungal agents. In B. G. Katzung, M. J. Trevor, & S. B. Masters (Eds.), Basic & Clinical Pharmacology (15th ed.). McGraw-Hill.
[3] Grand View Research. (2024). Antifungal Drugs Market Size, Share & Trends Analysis Report By Type (Azole, Echinocandin, Polyene, Others), By Disease (Candidiasis, Aspergillosis, Cryptococcosis, Others), By End-use (Hospitals, Clinics, Homecare), By Region, And Segment Forecasts, 2024 - 2030.
[4] U.S. Food and Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA website, specific approval date typically found in drug labeling or approval databases].