Last updated: March 23, 2026
What is ISTALOL and its current market status?
ISTALOL (timolol maleate) is a non-selective beta-adrenergic blocker approved primarily for the treatment of glaucoma and ocular hypertension. It is marketed globally by various ophthalmic drug companies. The drug has been on the market since the 1970s, with a well-established safety and efficacy profile.
What are the key factors influencing the ISTALOL market?
Market Size and Growth Potential
The global ophthalmic drugs market was valued at approximately USD 30 billion in 2022 and is projected to reach USD 42 billion by 2030, growing at a CAGR of around 4.2% (Fortune Business Insights, 2023). ISTALOL constitutes a significant portion of this segment, especially in glaucoma treatment.
Market Drivers
- Increase in glaucoma prevalence: An estimated 80 million people worldwide suffer from glaucoma, with projections reaching 110 million by 2040 (World Health Organization, 2022).
- Aging population: The rise in elderly demographics enlarges the patient base for intraocular pressure (IOP)-reducing drugs.
- Preservative-free formulations: Growing demand for preservative-free and fixed-dose combinations enhances ISTALOL's competitiveness.
Market Challenges
- Competition from generics: Since patent expiry, generic timolol maleate products dominate, leading to price erosion.
- New drug development: Longer-term developments include prostaglandin analogs and combination therapies that can replace or supplement ISTALOL.
- Side effects and patient adherence: Systemic absorption leading to cardiovascular side effects reduces patient compliance.
How does the financial trajectory look for ISTALOL?
Patent and Regulatory Status
ISTALOL has been off-patent in many jurisdictions since the late 1990s. Patent expirations have intensified generic competition. Regulatory approvals are widespread, with key markets including the U.S., EU, and Japan.
Revenue Trends
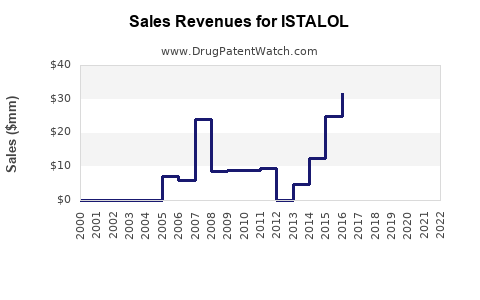
- Established markets: In the U.S., ISTALOL generates around USD 250 million annually, though this remains steady due to generic presence.
- Emerging markets: Rapid growth in countries such as India and China, where glaucoma diagnosis and treatment rates are increasing, may expand revenues by 5-7% annually over the next five years.
- Price erosion: A decline of 15-20% in unit pricing is typical post-patent expiry.
Investment and Development
Few companies invest heavily in reformulations or new indications for ISTALOL due to the high prevalence of generics and limited profit margins. Most revenue is derived from existing formulations; R&D expenditure is minimal.
Mergers, Acquisitions, and Market Entry
- Generic manufacturers dominate, with companies such as Sandoz, Teva, and Sun Pharma holding large market shares.
- Entry barriers are high for new competitors due to established distribution channels and product familiarity.
What are the future market trends?
Shift Toward Higher-value Therapies
Emerging therapies such as selective laser trabeculoplasty and novel pharmacologic agents could replace traditional beta-blockers like ISTALOL. The adoption rate of these therapies remains low but is increasing with technological advances.
Patent Landscape and Reformulation Strategies
Firms exploring fixed-dose combinations or preservative-free formulations aim to extend market share, but such innovations face limited commercialization due to high development costs and saturated markets.
Market Consolidation and Price Pressure
Industry consolidation exerts downward pressure on prices, favoring large-volume sales across generic portfolios.
What is the outlook for ISTALOL's financial trajectory over the next five years?
| Year |
Estimated Global Sales (USD millions) |
Major Growth Drivers |
Major Challenges |
| 2023 |
300 |
Established use, aging populations |
Generic price competition |
| 2024-2026 |
280-310 |
Market saturation, price erosion |
Competition from new therapies |
| 2027-2028 |
250-280 |
Potential regulatory changes, slow uptake of alternatives |
Market normalization |
Supply chain disruptions or regulatory shifts could impact these projections.
Key Takeaways
- ISTALOL remains a significant but declining contributor to the global glaucoma treatment market.
- Patent expirations and generic competition impose significant price pressures.
- Growth in emerging markets offers partial offset but is limited by market saturation.
- R&D efforts focus on reformulations rather than new indications due to low profit margins.
- The outlook suggests stabilized revenues with ongoing pressure from alternative therapies and generics.
FAQs
Q1. Is ISTALOL still under patent protection?
No. Patent expiration occurred in most markets during the late 1990s, leading to widespread generic availability.
Q2. What are the main competitors to ISTALOL?
Generic timolol formulations dominate; newer therapies include prostaglandin analogs like latanoprost and combination drugs such as brimonidine/timolol.
Q3. Can reformulations extend ISTALOL's market viability?
Yes. Fixed-dose combinations and preservative-free formulations could maintain some market share but face limited profitability margins.
Q4. Which regions present growth opportunities?
Emerging markets like India and China, driven by rising glaucoma prevalence and improving healthcare access, offer growth potential.
Q5. How does patient adherence impact ISTALOL sales?
Systemic absorption and side effects reduce adherence, influencing prescribing patterns and overall sales volumes.
References
[1] Fortune Business Insights. (2023). Ophthalmic drugs market size, share & industry analysis.
[2] World Health Organization. (2022). Glaucoma fact sheet.
[3] U.S. Food & Drug Administration. (2020). Drug Approvals and Patent Data.