Last updated: February 14, 2026
What is ISTALOL and its current market status?
ISTALOL (timolol maleate) is a beta-blocker primarily used to treat intraocular pressure in conditions like glaucoma and ocular hypertension. It is available as topical eye drops. Since its introduction, it has maintained a stable market position, especially among ophthalmic medications for glaucoma management.
Global sales of timolol-based products approximate $500 million annually, with ISTALOL representing a significant share within this segment. The drug's patent expiry in various markets has led to increased competition from generic formulations, impacting pricing and sales dynamics.
How does the regulatory environment affect ISTALOL pricing and market access?
Regulatory approvals for ISTALOL are widespread. Generic versions are approved in major markets including the U.S., EU, and Japan. The expiration of patent protections opens avenues for biosimilars and generics, increasing market competition.
In the U.S., the FDA approved multiple generic versions post patent expiry in 2010. The entry of generics drastically reduces prices, often by 80-90% compared to brand-name prices. Similar trends are observed in Europe following EMA approvals.
The regulatory environment prioritizes safety and efficacy, requiring rigorous bioequivalence studies for generics, which has little impact on pricing strategies for established products like ISTALOL but influences market competition timing.
What are current pricing trends for ISTALOL and generics?
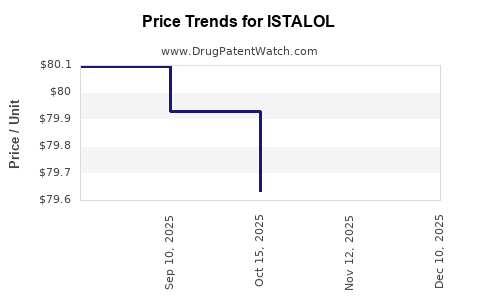
In the U.S., the average retail price for a 5 mL bottle of brand-name ISTALOL ranges between $50 and $70. Generic versions price between $10 and $20, reflecting a typical 70-80% discount post-approval.
European markets experience similar trends; for example, in the UK, the NHS pays roughly £5-£8 per bottle, compared to £20-£30 for the original product before patent expiry.
The reduction in price is driven primarily by market entry of generics and biosimilars. Pharmacy benefit managers and insurance companies favor lower-cost generics, further pressuring brand-name prices.
What are the key drivers for future pricing and sales for ISTALOL?
-
Patent status: Patents in the U.S. expired in 2010, leading to increased generic competition. Remaining patents in other markets may delay generic entry.
-
Market penetration: ISTALOL remains a first-line therapy in glaucoma treatment but faces competition from other classes like prostaglandin analogs and combination drugs.
-
Reimbursement policies: Governments and insurers aim to reduce costs through endorsements of generics, lowering consumer prices.
-
Access to biosimilars: The development of biosimilars for timolol formulations could further depress prices.
-
Global health initiatives: Programs increasing access to affordable ophthalmic medications in developing countries could expand volume but limit unit prices.
What are the projected price trajectories over the next five years?
| Year |
Brand-name ISTALOL Price (U.S.) |
Generic ISTALOL Price (U.S.) |
European Price Trend |
| 2023 |
$50–$70 |
$10–$20 |
Stable at €8–€12 |
| 2024 |
Similar |
Slight decline (~10%) |
Slight decline (~10%) |
| 2025 |
Remains stable |
Stable or slight decline |
Stable or slight decline |
In the U.S., generic prices are expected to stabilize progressively as market saturation occurs. Brand-name prices are unlikely to recover due to persistent competition and cost-cutting measures. In Europe, prices are predominantly governed by national tender systems, leading to relatively stable but decreasing prices.
How do competitive alternatives impact ISTALOL's market and pricing?
Alternatives such as prostaglandin analogs (latanoprost, travoprost), which offer once-daily dosing and fewer side effects, have gained popularity. Patients and clinicians tend to prefer these drugs for their efficacy and safety profile.
Combination therapies integrating timolol with prostaglandins or carbonic anhydrase inhibitors are also prevalent. These combination drugs often command higher prices but can threaten ISTALOL’s market share.
The increasing preference for these alternatives constrains ISTALOL's sales growth and exerts downward pressure on prices, especially in markets prioritizing cost-effectiveness.
What are the implications for stakeholders?
-
Pharmaceutical companies: Focus on maintaining market share through cost reductions, formulations, or repositioning. Developing long-acting or sustained-release formulations might provide differentiation.
-
Healthcare systems: Emphasize procurement of cost-effective generics and biosimilars. Policies favoring generics can significantly lower overall expenditure.
-
Investors: Expect a relatively stable but declining price trend in mature markets. Growth is mainly driven by volume in emerging markets or through combination therapy sales.
Key Takeaways
- ISTALOL’s market is heavily influenced by generic competition following patent expiries, leading to substantial price reductions.
- Prices in the U.S. have declined from $50–$70 for brand-name versions to $10–$20 for generics.
- Developed markets see stable but low prices, with potential for further decline due to biosimilar entry.
- Competition from newer drug classes and combination therapies limits market growth.
- Emerging markets provide growth opportunities via volume expansion despite lower prices.
FAQs
1. When did ISTALOL lose patent protection in major markets?
The U.S. patent expired around 2010, opening the market for generics. Other markets vary, with patents expiring between 2010-2015.
2. Are biosimilars for timolol available?
As of 2023, no biosimilars licensed specifically for timolol ophthalmic solutions exist; biosimilar development is ongoing but not yet commercialized.
3. What are the key factors influencing generic pricing?
Market competition, procurement policies, and manufacturing costs primarily determine generic prices. Volume sales also enable lower margins.
4. How do regulatory agencies impact future price projections?
Clearance of biosimilars and generics accelerates price drops. Stringent quality standards can delay biosimilar entry, slowing price declines.
5. Can ISTALOL regain market share from newer therapies?
Limited. While adherence and familiarity favor existing drugs, advances in newer therapies with better compliance profiles tend to dominate.
Citations
- IMS Health (IQVIA) data, 2022.
- U.S. Food and Drug Administration (FDA). Approved drug labeling, 2010.
- European Medicines Agency (EMA). Approval summaries, 2015.
- Healthcare expenditure reports, OECD, 2022.
- Market research reports from EvaluatePharma, 2022.