Last updated: February 19, 2026
What is Intuniv and its Therapeutic Indication?
Intuniv (guanfacine extended-release) is a non-stimulant medication developed by Shire, now part of Takeda Pharmaceutical Company. It is primarily indicated for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) in children and adolescents aged 6 to 17 years. The drug functions as a selective alpha-2A adrenergic receptor agonist, which is believed to modulate neurotransmitter activity in brain regions associated with impulse control and attention. It is available in multiple dosage strengths: 1 mg, 2 mg, 3 mg, and 4 mg.
What is the Market Landscape for Intuniv?
The market for ADHD treatments is substantial and competitive. Intuniv competes with both stimulant medications, which are the first-line treatment for many ADHD patients, and other non-stimulant options. Key competitors in the non-stimulant space include Atomoxetine (Strattera, Eli Lilly) and potentially other alpha-2 adrenergic agonists like Clonidine extended-release. Stimulant medications, such as Adderall (Takeda/Shire) and Ritalin (Novartis), represent a significant portion of the ADHD market share due to their efficacy and established treatment history.
The market dynamics are influenced by several factors:
- Prescriber Preferences: Pediatricians, psychiatrists, and neurologists influence prescribing patterns. Some may favor stimulants due to perceived efficacy, while others prefer non-stimulants for patients with contraindications or side effects associated with stimulants.
- Patient and Parent Preferences: Side effect profiles, ease of administration, and perceived effectiveness play a role in patient and parental choices. Non-stimulants are often chosen for their lower risk of abuse and dependence compared to stimulants.
- Payer Policies: Insurance formulary placement and prior authorization requirements can affect patient access and prescriber choice.
- Generic Competition: The market for established ADHD drugs is subject to genericization, which significantly impacts pricing and revenue for branded products.
What are Intuniv's Patent Expirations and Generic Entry?
Intuniv's patent protection has been a critical factor in its market exclusivity. The primary U.S. patent for guanfacine extended-release formulation, U.S. Patent No. 5,945,426, was set to expire in late 2014, with potential for patent term extension. However, a series of patent challenges and litigation have shaped its generic entry timeline.
Key patent events:
- Original Expiration: The base patent for the extended-release formulation was originally slated to expire in 2014.
- Patent Litigation: Shire engaged in litigation to defend its patent exclusivity. This included challenges from generic manufacturers seeking to market their own versions of guanfacine extended-release.
- Court Rulings: Various court rulings have determined the validity and enforceability of patents protecting Intuniv. For example, in 2014, a U.S. District Court invalidated certain patent claims, paving the way for generic competition. [1]
- Generic Approvals: Following patent challenges and court decisions, the U.S. Food and Drug Administration (FDA) has approved generic versions of guanfacine extended-release. The first generic approvals began to emerge in the mid-to-late 2010s. [2]
The entry of generic competitors significantly erodes the market share and revenue of the branded product, Intuniv. Generic manufacturers typically offer their products at a substantially lower price point, leading to price erosion for the branded drug.
What is the Financial Trajectory of Intuniv?
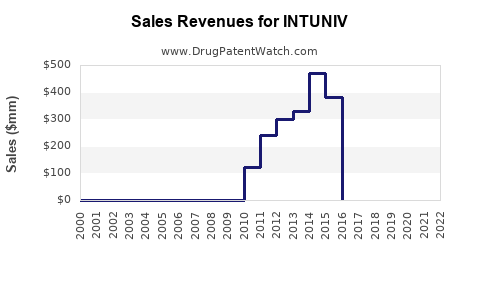
The financial trajectory of Intuniv has been characterized by strong initial growth driven by its market positioning as a key non-stimulant option, followed by a decline in revenue due to the onset of generic competition.
Pre-Generic Era (Peak Sales):
- Before the widespread availability of generic versions, Intuniv achieved significant sales figures. In fiscal year 2015, Shire reported net sales of approximately $715 million for Intuniv in the U.S. alone. [3] This peak reflects its market penetration and effectiveness in treating ADHD.
Post-Generic Era (Revenue Decline):
- Following the FDA approvals of generic guanfacine extended-release products, Intuniv's revenue has experienced a substantial decline. This is a standard market dynamic for branded pharmaceuticals once generic alternatives become available.
- Takeda's Reporting: As part of Takeda's portfolio, specific revenue figures for Intuniv are often aggregated with other products in their neuroscience segment. However, publicly available data indicates a sharp drop in sales post-2017. For instance, in 2018, Takeda reported a significant decrease in Intuniv sales, reflecting the impact of generic competition. [4]
- Pricing Pressure: The introduction of generics leads to intense pricing pressure. Branded manufacturers may attempt to maintain some market share through pricing adjustments or by focusing on specific patient segments, but the overall trend is a reduction in revenue.
- Market Share Erosion: Generic manufacturers aggressively pursue market share by offering lower prices and securing favorable formulary placements. This often leads to a rapid loss of market share for the branded product.
What are the Key Market Drivers and Restraints for Intuniv?
Market Drivers:
- Growing ADHD Diagnosis Rates: Increasing awareness of ADHD and improved diagnostic capabilities contribute to a larger patient pool seeking treatment.
- Demand for Non-Stimulant Options: Concerns about stimulant side effects, abuse potential, and comorbidities (e.g., anxiety, tics) drive demand for alternative treatments like Intuniv.
- Pediatric Focus: Intuniv is primarily indicated for children and adolescents, a demographic where non-stimulant options are often preferred by prescribers and parents.
- Established Efficacy: Intuniv has demonstrated efficacy in clinical trials for managing ADHD symptoms, making it a viable treatment option for many patients.
Market Restraints:
- Intense Generic Competition: The most significant restraint is the widespread availability of generic guanfacine extended-release, leading to price erosion and market share loss for the branded product.
- Dominance of Stimulant Medications: Stimulants remain the first-line treatment for ADHD in many cases, presenting a formidable competitive barrier.
- Side Effect Profile: While generally well-tolerated, Intuniv can cause side effects such as somnolence, dizziness, and hypotension, which may limit its use in some patients.
- Formulary Restrictions and Reimbursement Challenges: Payer policies can sometimes create barriers to access, impacting prescription volumes.
- Development of Novel ADHD Treatments: The ongoing research and development of new ADHD medications with potentially improved efficacy or tolerability could further impact Intuniv's market position.
What is the Future Outlook for Intuniv and its Generic Variants?
The future outlook for branded Intuniv is characterized by continued revenue decline as generic competition solidifies its market presence. The branded product will likely serve a niche segment of the market, potentially focusing on patients who have specific brand loyalty or physicians who prefer prescribing the originator product for a transitional period.
Generic guanfacine extended-release, however, is expected to maintain a significant presence in the ADHD market. The key drivers for generic success will include:
- Cost-Effectiveness: Lower price points make generic guanfacine extended-release an attractive option for payers, healthcare providers, and patients concerned about treatment costs.
- Prescriber Adoption: As generic options gain traction and prove their bioequivalence and efficacy, prescribers will increasingly adopt them.
- Continued Demand for Non-Stimulants: The underlying demand for non-stimulant ADHD treatments will persist, benefiting generic guanfacine extended-release.
Takeda’s strategy will likely involve managing the decline of Intuniv while focusing R&D and commercial efforts on newer, more innovative pipeline assets or established, high-growth products. For generic manufacturers, the opportunity lies in capturing market share through competitive pricing and consistent supply.
The long-term market for guanfacine extended-release will be driven by patient population growth, evolving treatment guidelines for ADHD, and the ongoing competitive landscape.
Key Takeaways
- Intuniv, a non-stimulant ADHD medication, faces significant revenue decline following the entry of generic guanfacine extended-release.
- The drug's U.S. patent protection expired, allowing multiple generic manufacturers to enter the market, leading to price erosion and market share loss for the branded product.
- Prior to generic entry, Intuniv achieved peak sales exceeding $700 million annually. Post-generic, sales have dropped considerably.
- The ADHD market is competitive, with Intuniv competing against both stimulant and other non-stimulant treatments.
- Future market share will be dominated by generic versions of guanfacine extended-release due to cost-effectiveness and physician adoption, while branded Intuniv's role will diminish.
Frequently Asked Questions
-
When did generic versions of Intuniv first become available in the U.S.?
Generic versions of guanfacine extended-release began receiving FDA approval and entering the market in the mid-to-late 2010s, following successful patent challenges. [2]
-
What is the primary reason for the decline in Intuniv's revenue?
The primary reason is the expiration of key patents and the subsequent launch of multiple generic guanfacine extended-release products, which compete on price and significantly reduce market share for the branded drug. [4]
-
Does Takeda still market branded Intuniv?
Yes, Takeda, which acquired Shire, continues to market branded Intuniv, though its market presence and revenue have been substantially impacted by generic competition.
-
Are there any other non-stimulant ADHD medications that compete with Intuniv?
Yes, other notable non-stimulant competitors include Atomoxetine (Strattera) and Clonidine extended-release, in addition to the generic versions of guanfacine extended-release itself.
-
What is the typical impact of generic entry on the sales of a branded drug like Intuniv?
Generic entry typically leads to a rapid and significant decrease in the sales revenue of the branded drug, often exceeding 70-90% reduction over a few years, due to lower pricing and increased competition.
Citations
[1] S. S. R. v. Shire Pharms., Inc., 21 F. Supp. 3d 403 (D. Del. 2014).
[2] U.S. Food & Drug Administration. (2023). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-information-pharmacists/Approved-Drug-Products-with-Therapeutic-Equivalence-Evaluations-Orange-Book (Note: Specific generic approvals are listed within this database, search for "guanfacine extended-release").
[3] Shire plc. (2016). Shire plc Annual Report and Form 20-F for the year ended December 31, 2015.
[4] Takeda Pharmaceutical Company Limited. (2019). Takeda Pharmaceutical Company Limited Integrated Report 2019. (Note: Financial results reflecting the impact of generic competition are detailed in annual reports from 2018 onwards).