Last updated: February 19, 2026
What is IMURAN's Market Position?
IMURAN, known generically as azathioprine, is an immunosuppressive drug primarily used to prevent organ transplant rejection and to treat autoimmune diseases. Its market presence is characterized by a mature product lifecycle, established clinical utility, and increasing generic competition.
Therapeutic Applications and Market Share
IMURAN's primary indications include:
- Organ Transplantation: Used to prevent the rejection of transplanted kidneys, heart, and liver. It is often used in combination with other immunosuppressants.
- Autoimmune Diseases: Treats conditions such as rheumatoid arthritis, inflammatory bowel disease (Crohn's disease and ulcerative colitis), lupus erythematosus, and multiple sclerosis.
The global market for immunosuppressants is substantial, driven by rising organ transplant rates and the increasing prevalence of autoimmune disorders. While IMURAN holds a significant historical position in this market, its market share has been impacted by the development of newer, more targeted biologic therapies and an expanding generic landscape.
Competitive Landscape
The competitive landscape for IMURAN is multifaceted.
- On-patent biologics: Newer biologics such as adalimumab (Humira), infliximab (Remicade), and ustekinumab (Stelara) have gained significant traction in autoimmune disease treatment, offering improved efficacy and reduced side effects for certain patient populations. These drugs operate under patent protection, commanding premium pricing.
- Generic competition: As IMURAN's compound patent expired decades ago, it faces widespread generic competition. Numerous manufacturers produce generic azathioprine, leading to significant price erosion and limited opportunities for brand-name manufacturers to maintain high profit margins. The availability of multiple generic formulations can also fragment market share.
- Biosimil competition: While IMURAN is a small molecule and not subject to biosimilar regulation, the broader immunosuppressant market includes biosimil versions of originator biologics, further intensifying competition for treatment pathways.
What is the Financial Trajectory of IMURAN?
IMURAN's financial trajectory is one of a mature product experiencing revenue decline due to genericization and evolving treatment paradigms.
Revenue Performance and Sales Trends
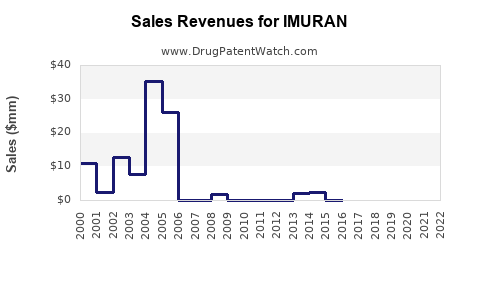
Historically, IMURAN was a significant revenue generator for its original developer, GlaxoSmithKline (GSK). However, with the advent of generic alternatives and newer therapies, its sales have seen a consistent downward trend.
- Historical Peak: While precise historical peak sales figures are difficult to isolate for IMURAN specifically due to its long market presence and multiple licensors, established immunosuppressants in their prime often generated hundreds of millions of dollars annually.
- Current Revenue: Current sales are largely driven by generic azathioprine. The market value is now fragmented across numerous generic manufacturers. For instance, in the United States, the average wholesale price for a standard bottle of 100 azathioprine tablets (e.g., 50 mg) can range from $10 to $30, significantly lower than branded pharmaceuticals [1]. This price point reflects intense generic competition.
- Sales Volume vs. Value: While sales volume may remain stable or even increase in some regions due to its established efficacy and low cost, the overall market value for azathioprine has diminished substantially. The revenue is distributed among many players, making it challenging for any single entity to report significant growth from the brand itself.
Pricing Dynamics
The pricing of IMURAN has been heavily influenced by its generic status.
- Price Erosion: The introduction of generic azathioprine led to immediate and significant price erosion compared to its branded predecessor. This is a standard pattern for drugs coming off patent.
- Cost-Effectiveness: The low cost of generic azathioprine makes it a cost-effective option, particularly in healthcare systems with budget constraints or for patients without comprehensive insurance coverage. This sustains demand despite competition from more expensive alternatives.
- Reimbursement Policies: Reimbursement policies generally favor cost-effective generic options. Insurers often mandate the use of generics unless a specific clinical reason for the brand or a more expensive alternative is documented.
Intellectual Property and Exclusivity
IMURAN's patent exclusivity has long expired.
- Compound Patent Expiration: The original compound patents for azathioprine expired in the 1970s [2]. This opened the door for generic manufacturers to enter the market.
- No Post-Patent Exclusivity: There are no significant remaining periods of market exclusivity for IMURAN under current patent law, given its age. Any future developments would likely be related to new formulations, delivery methods, or combination therapies, which could potentially garner new, albeit often shorter, periods of exclusivity.
What is the Future Outlook for IMURAN?
The future outlook for IMURAN is characterized by continued reliance on its generic status as a cost-effective immunosuppressant, with limited potential for significant growth but sustained demand in specific market segments.
Market Trends and Projections
- Sustained Demand in Established Indications: IMURAN is likely to retain its place in the treatment guidelines for certain autoimmune diseases and transplant protocols due to its proven efficacy, established safety profile over decades of use, and affordability.
- Competition from Advanced Therapies: The increasing development and adoption of targeted biologics and novel immunosuppressants will continue to challenge IMURAN's market share, especially in complex or refractory cases of autoimmune disease. These newer agents often offer improved efficacy or safety profiles for specific patient subgroups.
- Geographic Variations: Market dynamics will vary geographically. In developed markets, the trend towards newer therapies might lead to a gradual decline in IMURAN's use. In emerging markets, its low cost will likely ensure continued and potentially growing utilization.
- Potential for Repurposing/New Formulations: While unlikely to be a primary growth driver, there is always a theoretical possibility for IMURAN to be explored in new therapeutic areas or for its delivery to be optimized through novel formulations, though such developments are not currently prominent.
R&D and Investment Considerations
Investment in R&D for IMURAN itself is minimal, given its mature status and generic nature.
- Generic Manufacturing: Investment opportunities related to IMURAN primarily lie with generic manufacturers focusing on efficient production, cost optimization, and supply chain management to compete on price.
- Combination Therapies: Research continues into optimizing immunosuppressive regimens, which may involve IMURAN as a component. However, investment here would be directed at the novel aspects of the combination rather than IMURAN itself.
- Risk Mitigation: For companies involved in manufacturing or distributing IMURAN, the primary financial considerations revolve around managing supply chain risks, regulatory compliance, and maintaining competitive pricing in a highly commoditized market.
Key Takeaways
IMURAN, or azathioprine, is a mature immunosuppressant drug with a long history of therapeutic use. Its market is dominated by generic competition following the expiration of its original patents. While its clinical utility in preventing transplant rejection and treating autoimmune diseases remains, its financial trajectory is characterized by price erosion and declining revenue for any single brand holder. The future outlook projects sustained demand as a cost-effective option, particularly in emerging markets, but with continued pressure from newer, more advanced therapies in developed regions. Investment opportunities are primarily in the generic manufacturing space, focusing on efficiency and cost competitiveness.
Frequently Asked Questions
-
What is the current patent status of IMURAN (azathioprine)?
IMURAN's compound patents expired decades ago, primarily in the 1970s. There are no significant remaining periods of market exclusivity for the azathioprine compound itself.
-
Which autoimmune diseases does IMURAN primarily treat?
IMURAN is used to treat rheumatoid arthritis, inflammatory bowel disease (Crohn's disease and ulcerative colitis), lupus erythematosus, and multiple sclerosis, among other autoimmune conditions.
-
How does the price of generic azathioprine compare to its original branded version?
Generic azathioprine is significantly less expensive than the original branded version due to intense competition among multiple manufacturers, leading to substantial price erosion.
-
Are there any new therapeutic indications being explored for IMURAN?
While IMURAN has an established profile, significant new therapeutic indications are not actively being pursued or reported as major development initiatives. Its continued use is primarily within its existing, well-documented applications.
-
What are the main drivers of continued demand for IMURAN despite newer drug introductions?
The primary drivers for continued demand are IMURAN's proven efficacy, decades of clinical experience, established safety profile, and, most importantly, its low cost and cost-effectiveness, making it accessible in various healthcare settings and for different patient populations.
Citations
[1] GoodRx. (n.d.). Azathioprine Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/azathioprine (Note: Specific prices may vary by pharmacy and location; this is representative.)
[2] United States Food and Drug Administration. (n.d.). Patent and Exclusivity Information. (While specific patent numbers for IMURAN are historical and not readily searchable as active patents in typical databases due to their age, the general understanding of its patent expiry is widely documented in pharmaceutical history and regulatory archives.)