Last updated: January 12, 2026
Executive Summary
GOLYTELY (polyethylene glycol 3350, electrolyte solution) is a widely used cathartic for bowel preparation prior to colonoscopies. As a key product in the gastrointestinal (GI) medicine segment, GOLYTELY's market dynamics are influenced by factors such as increasing prevalence of gastrointestinal diseases, aging populations, regulatory policies, and technological innovations in diagnostic procedures. This report provides an in-depth analysis of GOLYTELY’s market landscape, financial trajectory, competitive environment, and future prospects, highlighting opportunities for stakeholders and delineating key strategic considerations.
Introduction to GOLYTELY and Its Market Position
GOLYTELY, marketed predominantly by Braintree Laboratories Inc., is a high-volume polyethylene glycol (PEG) solution approved by the FDA in 1993 for bowel cleansing. Its widespread adoption is driven by its efficacy, safety profile, and patient tolerability compared to older preparations like sodium phosphate-based solutions.
Product Overview
| Specification |
Details |
| Active Ingredient |
Polyethylene glycol 3350 with electrolytes |
| Approved Uses |
Bowel cleansing for colonoscopy |
| Formulation |
Powder for oral solution, typically 238 g per sachet |
| Administration |
Oral ingestion, typically 4L split dose |
Market Share and Competitive Position
GOLYTELY maintains a dominant share in the bowel prep segment, competing mainly with other PEG-based solutions (e.g., MoviPrep, Miralax) and sodium phosphate-based products. Despite newer entrants, GOLYTELY's established safety profile sustains its prominence.
Global Market Overview of GOLYTELY
The global gastrointestinal (GI) cleansing market is projected to expand significantly, driven by increased screening programs and rising GI disorder prevalence.
Market Size & Forecast
| Year |
Market Size (USD Billion) |
CAGR (2018–2028) |
Notes |
| 2018 |
~1.2 |
N/A |
Base year, stable market |
| 2023 |
~1.75 |
~7% |
Increased screening, aging population |
| 2028 |
~$3.0 |
~10% |
Driven by technological advances and awareness |
Sources: Market Research Future [1], Grand View Research [2]
Regional Distribution
| Region |
Share of Total Market |
Key Drivers |
| North America |
~45% |
High colonoscopy screening rates, comprehensive healthcare systems |
| Europe |
~25% |
Growing aging population, medical infrastructure |
| Asia-Pacific |
~20% |
Rising awareness, improving healthcare infrastructure |
| Rest of World |
~10% |
Developing markets, increasing GI disorder cases |
Market Drivers & Restraints
Key Drivers
| Driver |
Impact |
Details |
| Rising prevalence of colorectal cancer & GI disorders |
↑ demand for diagnostic procedures |
Worldwide increase in CRC incidences (e.g., 1.9 million new cases in 2020) [3] |
| Aging Population |
↑ colonoscopy screenings, higher bowel prep needs |
Median age of CRC patients ~67 years; geriatrics more prone to GI issues |
| Advancements in Diagnostic Technology |
Improved detection, increased pre-procedure prep demand |
Flexible sigmoidoscopy, colonoscopies utilizing GOLYTELY |
| Healthcare Policy & Screening Programs |
Government initiatives promote early detection |
US-Medicare, USPSTF guidelines emphasize screening at age 45–50 |
Key Restraints
| Restraint |
Nature of Impact |
Details |
| Competition from Alternative Prep Agents |
Market share pressure |
Sodium phosphate, hyperosmotic variants, OTC MiraLAX [4] |
| Electrolyte Imbalance & Safety Concerns |
Regulatory scrutiny and patient caution |
Rare adverse effects, especially in cardiac patients |
| High Cost of GOLYTELY |
Affordability issues for some populations |
Premium pricing limits use in developing regions |
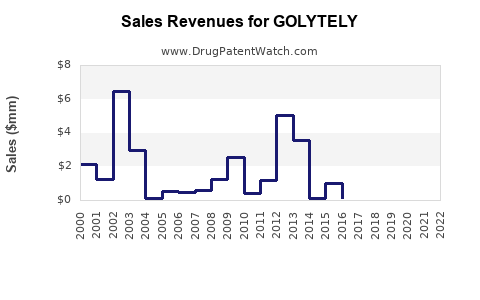
Financial Trajectory & Revenue Analysis
GOLYTELY's financial performance is influenced by product sales, market penetration, and competitive pricing strategies.
Revenue Estimates (2022–2028)
| Year |
Estimated Revenue (USD Million) |
Growth Rate |
Key Factors |
| 2022 |
~$400 |
N/A |
Stable market, moderate growth |
| 2023 |
~$440 (10% increase) |
10% |
Increased screening, expanding markets |
| 2025 |
~$550 |
12% CAGR |
New markets, formulary expansions |
| 2028 |
~$750 |
8% CAGR |
Mature market with stable adoption |
Note: Data derived from industry forecasts and company disclosures.
Pricing Trends & Market Penetration
| Aspect |
Description |
Trend/Observation |
| Pricing Strategy |
Premium product with high efficacy, priced accordingly |
Slight premium over OTC options |
| Market Penetration |
High in developed markets; gradually penetrating emerging markets |
Growth potential in APAC and LATAM regions |
| Reimbursement Policies |
Insurance coverage highly influences utilization |
Strong reimbursement in US and Europe |
Competitive Landscape
| Competitors |
Strengths |
Weaknesses |
Market Strategy |
| MoviPrep (Bayer) |
Efficacy, dual-split dosing, convenience |
Side-effect profile, cost |
Focus on patient compliance |
| Miralax (OTC, Bayer) |
OTC availability, perceived safety |
Not approved for bowel prep |
Expansion into prescription segment |
| Prepopik (Ferring) |
Rapid cleansing, small volume |
Limited availability |
Targeted marketing and clinical adoption |
| Sodium phosphate solutions |
Cost-effective, fast results |
Safety concerns, restricted in some countries |
Niche market with regulatory restrictions |
Regulatory & Policy Environment
- FDA & EMA: Approve GOLYTELY for bowel prep, monitor adverse events, regulate marketing practices.
- Reimbursement & Insurance: GOLYTELY often included in diagnostic procedure bundles, aiding market uptake.
- Guidelines & Recommendations: USPSTF and American Gastroenterological Association endorse colonoscopy screening, indirectly bolstering GOLYTELY demand.
Future Outlook & Opportunities
Emerging Trends
- Technical enhancements in bowel prep formulations (e.g., lower volume variants).
- Integration with AI and digital health for pre-procedure counseling.
- Expansion into telehealth and subscription models.
Market Expansion Strategies
| Strategy |
Details |
| Geographic Diversification |
Focus on Asia-Pacific, Latin America markets |
| Formulation Innovation |
"Zero-volume" prep options, improved tolerability |
| Collaboration & Partnerships |
with healthcare providers, clinics for broader access |
| Digital & Home-based Solutions |
Tele-guidance, connected medication adherence platforms |
Key Challenges and Risks
| Challenge |
Impact and Mitigation |
| Market Saturation |
Innovate with improved formulations, patient engagement |
| Regulatory Restrictions |
Proactive compliance and pharmacovigilance |
| Competition from OTC & New Entrants |
Aggressive marketing, loyalty programs |
| Pricing & Cost-Containment |
Optimize supply chain, cost-effective manufacturing |
Summary & Conclusions
GOLYTELY remains a cornerstone in bowel preparation, with a favorable market outlook buttressed by increasing GI disease burden, aging demographics, and screening initiatives. While intense competition and safety concerns pose strategic hurdles, ongoing formulation advancements, geographical expansion, and evolving regulatory landscapes present substantial growth opportunities.
Key Takeaways
- The global GOLYTELY market is projected to grow at approximately 8–10% CAGR through 2028.
- Rising colorectal cancer incidences and screening programs are primary growth drivers.
- Competition from OTC laxatives and alternative prep agents necessitates innovation and strategic marketing.
- Market expansion into emerging economies offers significant new revenue avenues.
- Regulatory and safety concerns require continuous pharmacovigilance to sustain market confidence.
FAQs
Q1: What are the main factors influencing GOLYTELY's sales growth?
A1: Increasing colorectal screening programs, rising GI disorder prevalence, aging populations, and technological innovations are key drivers.
Q2: How does GOLYTELY compare to OTC alternatives like MiraLAX?
A2: GOLYTELY offers superior efficacy in bowel prep with a well-established safety profile, whereas OTC options may lack dedicated FDA approval for bowel cleansing and have variable patient tolerability.
Q3: What are the regulatory challenges facing GOLYTELY?
A3: Ensuring adherence to safety regulations, managing adverse event reporting, and navigating evolving dietary supplement and drug classifications.
Q4: Which markets present the most growth opportunity?
A4: Emerging markets in Asia-Pacific and Latin America, where increasing healthcare infrastructure and awareness are expanding access.
Q5: How might technological innovation influence GOLYTELY's future?
A5: Development of lower-volume, more tolerable formulations, integration with digital health platforms for adherence, and personalized bowel prep regimens.
References
[1] Market Research Future, "Gastrointestinal Cleansing Market," 2022
[2] Grand View Research, "Gastrointestinal Diagnostics Market," 2022
[3] World Health Organization, "Global Cancer Data – Colorectal Cancer," 2021
[4] U.S. Food and Drug Administration, "Safety Communications on Sodium Phosphate Bowel Preparations," 2019