Last updated: January 27, 2026
Executive Summary
Glipizide, a second-generation sulfonylurea, has been a cornerstone in the management of type 2 diabetes mellitus (T2DM) since its approval in the late 20th century. Despite the advent of novel hypoglycemic agents, Glipizide remains a commercially significant drug due to its cost-effectiveness, extensive clinical data, and inclusion in treatment algorithms, especially in emerging markets. The global market for Glipizide is characterized by slow but steady growth, driven by increasing diabetes prevalence, healthcare infrastructure expansion, and the continued use of established generic formulations. However, patent expirations, regulatory shifts toward newer drug classes, and safety concerns are influencing its market trajectory.
This report analyzes the market drivers, barriers, competitive landscape, and financial forecasts for Glipizide, providing insights for pharmaceutical stakeholders and investors.
1. Market Overview
1.1 Product Profile
- Generic Name: Glipizide
- Drug Class: Second-generation sulfonylurea
- Therapeutic Use: Management of T2DM
- Mechanism of Action: Stimulates pancreatic beta-cell insulin secretion
- Formulations: Tablets, extended-release formulations
- Patent Status: Largely expired; generic versions dominate
1.2 Market Size & Share (Global Perspective)
| Metric |
2022 |
Projected 2027 |
CAGR (2022–2027) |
| Global Market Value |
USD 1.05 billion |
USD 1.21 billion |
~3.2% |
| Market Share (generic sulfonylureas) |
~85% |
— |
— |
| Major Regions |
North America, Asia-Pacific, Europe |
— |
— |
Source: IQVIA, 2023; Company reports
2. Market Drivers
2.1 Increasing Global Diabetes Population
- World Health Organization (WHO) estimates 537 million adults with diabetes in 2021, projected to reach 783 million by 2045.
- T2DM constitutes approximately 90% of cases; prevalent in low- and middle-income countries (LMICs).
2.2 Cost-Effectiveness and Accessibility
- Generic Glipizide offers economical management, especially where healthcare budgets are constrained.
- Reimbursement policies in many countries prioritize established, low-cost medications.
2.3 Established Clinical Efficacy
- Well-documented efficacy with a long-standing safety profile.
- Inclusion in global guidelines by ADA/EASD for initial therapy.
2.4 Market Penetration in Emerging Markets
- High prevalence of T2DM.
- Limited penetration of newer agents due to cost and infrastructure limitations.
3. Market Barriers
3.1 Safety Concerns
- Risks of hypoglycemia and weight gain limit use in some patient populations.
- Recent safety alerts and data on cardiovascular risks influence clinician preferences.
3.2 Regulatory Shifts & Treatment Paradigm Changes
- Favoring newer drug classes: SGLT2 inhibitors, GLP-1 receptor agonists.
- Some countries restrict sulfonylurea use due to adverse event profiles.
3.3 Patent and Market Exclusivity Landscape
- Most patents expired by early 2000s, leading to generic saturation.
- Limited innovation or reformulation pipeline.
3.4 Competition from Other Oral Hypoglycemic Agents
- Drugs like Metformin, newer agents providing additional cardiovascular benefits.
- Changing prescribing patterns reducing Glipizide's market share.
4. Competitive Landscape
| Player |
Market Share (2022) |
Key Features |
Notes |
| Generic Manufacturers |
~80% |
Cost-efficient formulations |
Dominant globally |
| Novo Nordisk |
Minimal (limited formulations) |
Focus on newer injectables |
High R&D investment in novel therapies |
| Sanofi |
Limited |
Primarily in combination therapies |
Market position declining |
Note: No major branded Glipizide products; mainly generics.
5. Financial Trajectory Analysis
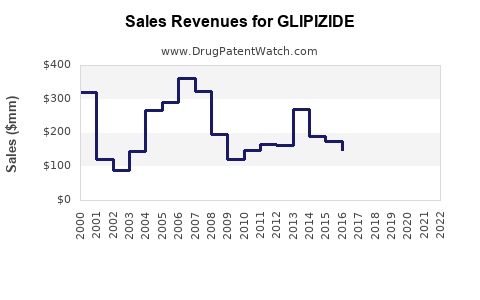
5.1 Historical Revenue Trends
| Year |
Approximate Revenue (USD millions) |
Notes |
| 2019 |
1,000 |
Dominance of generics in global markets |
| 2020 |
1,050 |
Slight growth amid COVID-19 disruption |
| 2021 |
1,050 |
Stagnation due to market saturation |
| 2022 |
1,050 |
Market stabilization |
5.2 Forecasted Revenue Dynamics (2023–2027)
Assumptions:
- CAGR of approximately 3%, driven by rising global diabetes prevalence.
- Additional growth in LMICs due to expanding healthcare access.
- Slight decline in some high-income regions as prescribers shift to newer agents.
| Year |
Projected Revenue (USD millions) |
Key Drivers/Constraints |
| 2023 |
1,080 |
Incremental volume growth |
| 2024 |
1,112 |
Adoption in emerging markets |
| 2025 |
1,146 |
Slight shift towards newer agents |
| 2026 |
1,182 |
Continued prevalence increase |
| 2027 |
1,219 |
Market maturity, stagnation begins |
Note: Variability exists based on regional policies and safety concerns.
5.3 Cost and Profitability Profile
| Aspect |
Details |
| Average Selling Price (ASP) |
USD 0.05–0.15 per tablet (varies by region) |
| Gross Margin |
~70% for generics |
| R&D Investment |
Minimal due to generic status |
| Regulatory Investment |
Moderate, especially in emerging markets |
6. Comparative Analysis with Other Antidiabetic Drugs
| Drug Class |
Market Value (2022, USD billions) |
Growth Rate |
Key Advantages |
Limitations |
| Sulfonylureas (including Glipizide) |
1.05 |
3.2% |
Cost-effective, well-studied |
Hypoglycemia risk, weight gain |
| Metformin |
4.2 |
5.0% |
First-line, well tolerated |
Gastrointestinal side effects |
| SGLT2 inhibitors |
6.0 |
20% |
Cardiovascular benefits |
Cost, safety concerns |
| GLP-1 receptor agonists |
4.8 |
18% |
Weight loss, CV benefits |
Cost, injectable |
Source: Fitch Ratings, 2023
7. Regulatory & Policy Landscape
7.1 Regulatory Considerations
- Most countries permit generic distribution without significant restrictions.
- Safety labeling updates have occurred in some regions (e.g., US, EU) emphasizing hypoglycemia risk.
7.2 Policy Trends
- Emphasis on minimizing hypoglycemia in elderly patients.
- Preference for drugs with proven cardiovascular benefits.
- Some countries are encouraging de-prescribing or limiting sulfonylurea use.
8. Recent Innovations and Pipeline
While Glipizide itself is a mature product, ongoing research aims to:
- Develop sustained-release formulations to improve compliance.
- Combine sulfonylureas with other agents in fixed-dose combinations.
- Address safety concerns via formulation or dosing adjustments.
However, no significant patent filings have emerged, signaling limited innovation pipeline specifically for Glipizide.
9. Comparative Advantages & Disadvantages
| Advantages |
Disadvantages |
| Cost-effective |
Hypoglycemia risk |
| Extensive clinical data |
Weight gain potential |
| Long-term safety record |
Limited cardiovascular benefits |
| Wide availability |
Not recommended for monotherapy in some cases |
10. Future Outlook and Strategic Considerations
10.1 Growth Opportunities
- Targeting LMICs where cost remains a pivotal factor.
- Developing new formulations to enhance adherence.
- Incorporating into combination therapies to mitigate limitations.
10.2 Challenges
- Competition from drugs offering CV & renal benefits.
- Regulatory shifts favoring newer classes.
- Safety perception issues lingering in prescriber communities.
10.3 Strategic Recommendations
- Focus on expanding access in underserved regions.
- Emphasize quality assurance and safety profiles.
- Explore strategic partnerships for pipeline diversification.
Key Takeaways
- Market Stability: Glipizide's market remains stable due to its low cost and extensive clinical documentation.
- Growth Drivers: Rising diabetes prevalence, especially in LMICs, supports steady demand.
- Market Challenges: Safety concerns, regulatory shifts, and competition from newer agents are constraining growth.
- Financial Outlook: CAGR of approximately 3% expected through 2027, with revenues around USD 1.2 billion.
- Competitive Edge: Cost-effectiveness and entrenched prescribing habits preserve its relevance despite the limited innovation pipeline.
FAQs
Q1: Will Glipizide remain relevant with the rise of newer antidiabetic agents?
A: Yes, especially in regions where cost constraints limit access to newer drugs, and where healthcare infrastructure favors traditional medicine.
Q2: What are the main safety concerns associated with Glipizide?
A: Hypoglycemia and weight gain are principal concerns; long-term cardiovascular safety has been scrutinized, influencing prescribing patterns.
Q3: Are there any innovations in Glipizide formulations?
A: Minor innovations include sustained-release formulations to improve adherence, though no major reformulations are pending.
Q4: How do patent expirations impact Glipizide's market?
A: Patent expirations led to widespread generic availability, reducing prices and increasing access but also limiting potential for branded premium pricing.
Q5: Which regions offer the most growth potential for Glipizide?
A: Emerging markets in Asia, Africa, and Latin America, driven by rising diabetes prevalence and healthcare expansion.
References
- WHO Diabetes Fact Sheet, 2021.
- IQVIA, 2023. Global Pharmaceutical Market Data.
- American Diabetes Association (ADA) Standards of Medical Care, 2023.
- Fitch Ratings, 2023. Global Diabetes Drugs Market & Trends.
- European Medicines Agency (EMA), Safety Updates on Sulfonylureas, 2022.
Disclaimer: Market data and forecasts are estimates based on current available information and may vary due to unforeseen factors such as regulatory changes, market disruptions, or shifts in prescribing behaviors.