Last updated: April 23, 2026
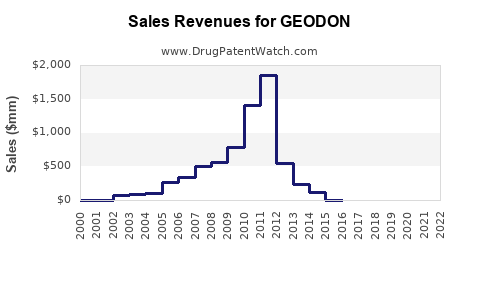
GEODON (ziprasidone) remains a mature, branded antipsychotic with contracting revenue potential driven by sustained generic pressure, limited product line expansion, and dependence on a US-centric payer channel. The financial trajectory since major generic entries is best characterized as plateauing followed by decline, with intermittent stabilization tied to inventory cycles, formulary access in managed care, and periodic life-cycle extensions (not a clean multi-year growth engine).
What is GEODON’s market position and competitive landscape?
GEODON is a branded formulation of ziprasidone, an atypical antipsychotic. Competitive pressure in the US has been dominated by generic ziprasidone across immediate-release and, where applicable, alternative dosage forms. This structure creates a price-down dynamic: branded pricing power compresses as payers prefer generic equivalents and as pharmacy benefit managers (PBMs) push tiering.
US channel dynamics
- Payer behavior: Managed care designs formularies that steer utilization toward generics after patent/market exclusivity expiry. GEODON’s net pricing depends heavily on rebate pressure, tier placement, and dispensing mix.
- Prescriber behavior: Ziprasidone retains clinical use in schizophrenia and bipolar indications, but switching costs are low once generics dominate, given therapeutic equivalence and broad clinical familiarity.
Competitor set (practical substitute pressure)
GEODON competes less as a “class monopoly” and more against:
- Within-class atypical antipsychotics (oral branded and generic) on formulary and step-therapy logic.
- Direct generic ziprasidone substitutability, which anchors utilization after GEODON’s branded advantage fades.
How do market mechanics affect revenue momentum for GEODON?
For mature branded products, revenue mechanics typically hinge on units, net price, and gross-to-net adjustments. For GEODON, generic penetration changes all three.
1) Units: generic substitution limits long-run volume growth
Once generic supply becomes entrenched, total ziprasidone prescriptions may remain stable, but branded share falls:
- Branded units face steady erosion as prescribers write for generics or pharmacists dispense generics under plan design.
- Total class units can be stable, but the branded component declines.
2) Net price: rebate compression does not offset generic pricing gravity
Net sales for branded antipsychotics reflect:
- Higher gross-to-net due to contracting and PBM rebate structures.
- A declining net price over time as payers demand steeper discounts to retain any branded share.
Net effect: declining average net price plus branded share erosion typically produces a negative revenue trend even if total prescriptions remain steady.
3) Inventory cycles: short stabilizers, not long-term growth
Mature products can show quarter-to-quarter noise from:
- Wholesaler buying patterns
- Patient refill cycles
- Contracting changes and channel inventory normalization
This can create brief “plateau” periods, followed by resumed decline.
What does the financial trajectory look like since generic competition?
GEODON’s financial trajectory has followed a conventional branded-to-generic arc:
- Post-exclusivity period: revenue starts declining as generic share increases.
- Later period: sharper decline typically occurs early after major generic uptake, then slows to a lower stable baseline dependent on residual branded loyalty, access, and pharmacy-level substitution frictions.
In antipsychotics, the branded baseline can persist longer than in some specialties because:
- Treatment requires ongoing chronic dosing
- Multiple generics can still coexist, so a branded product can retain limited share if it is positioned in plan tiers during parts of the contracting cycle.
But in aggregate, the branded revenue path is structurally constrained by:
- Widespread generic coverage
- Tighter step-therapy and formulary controls
- Ongoing price benchmarking against generics
What factors have historically shaped GEODON’s US sales evolution?
Formulary access and tiering
- When GEODON sits in a preferred tier, it can retain share longer.
- If payers move it to non-preferred tiers, branded share drops quickly under copay sensitivity.
PBM contracting and rebate terms
- Rebates rise with competitive threats to defend share.
- Higher rebates reduce net margin even when units are stable.
Product life-cycle and line extensions
Ziprasidone’s life-cycle strategy can include:
- Dosage form updates
- Labeling refinements
- Manufacturing or supply chain improvements that reduce out-of-stocks
These can support short-term revenue stability, but they do not reverse the economics of generic substitution.
How does GEODON’s indication set affect demand durability?
GEODON is used for:
- Schizophrenia
- Bipolar disorder (notably bipolar mania and/or bipolar-related maintenance depending on label language at different times)
This matters because chronic indications create ongoing demand. Still:
- Demand durability benefits the molecule, not the brand.
- Once generics dominate, the durability translates into generic volume, not necessarily branded revenue.
What are the key drivers and constraints for GEODON’s forward financial path?
Key drivers (what can slow decline)
- Formulary leverage: If GEODON retains favored placement in select plans and PBM contracting cycles.
- Institutional use patterns: Some institutional formularies lag in switching, sustaining branded prescriptions temporarily.
- Clinical switching friction: In practice, some prescribers remain conservative about changing stable patients, even after generic availability.
Key constraints (what drives decline)
- Generic coverage saturation: Broad national availability limits branded differentiation.
- Net price compression: Higher rebates and lower net prices under payer pressure.
- Therapeutic competition: Newer branded antipsychotics and alternative mechanisms can influence treatment choice, especially where payers introduce preferential coverage.
Where does GEODON sit on the branded lifecycle curve versus typical antipsychotic peers?
GEODON fits the profile of a mature branded antipsychotic:
- Declining branded share after generic entries
- Persistent, not growing, molecule-level demand
- Revenue largely determined by contracting outcomes and branded retention rather than innovation-driven demand
In investment terms, this positions GEODON more like a cash-cow defense product than a growth asset, with value tied to durability of branded share.
What market signals matter most for GEODON risk and upside?
The highest-signal indicators for GEODON are:
- Branded share trends within total ziprasidone scripts
- Net sales per prescription (proxy for net price after rebates)
- Wholesaler inventory trends (quarter-to-quarter stability)
- Formulary movement across major PBMs
- Launch timing and adoption of any additional generic entries that can accelerate branded erosion
Financial trajectory summary table
| Dimension |
Expected direction for GEODON |
Mechanism |
| Branded unit demand |
Down |
Generic substitution reduces branded share |
| Net price (average) |
Down |
PBM contracting and rebate compression under competitive pressure |
| Gross-to-net |
Up |
Competitive retention requires larger rebates/discounts |
| Revenue trend (net sales) |
Down overall |
Units decline plus net price compression |
| Short-term volatility |
Moderate |
Inventory cycles and contracting timing |
| Long-term outlook |
Limited growth |
Structural generic pressure dominates |
Key Takeaways
- GEODON’s market is dominated by generic ziprasidone substitution, making branded revenue growth structurally difficult.
- The financial trajectory is consistent with a mature branded-to-generic decline pattern: branded share erosion plus net price compression, with only episodic stabilization from contracting and inventory cycles.
- Forward performance is determined more by payer/formulary dynamics and branded retention than by new demand creation.
FAQs
-
Is GEODON’s demand tied to the entire ziprasidone market or only to the brand?
Demand is tied to total ziprasidone utilization for molecule-level volume, but GEODON’s net sales depend on branded share and net pricing after rebates.
-
What drives GEODON revenue quarter-to-quarter?
Branded share shifts from PBM/formulary placement, rebate changes, and wholesaler inventory cycles.
-
Does GEODON still have growth potential?
Growth potential is limited by generic saturation; the most realistic upside is slower decline through maintained formulary access and branded retention.
-
What’s the main financial risk for GEODON?
Accelerated branded share loss from increased generic competition or payer tier downgrades that push dispensing toward generics.
-
Which market indicator best predicts GEODON’s trajectory?
Branded share within total ziprasidone prescriptions combined with net sales per prescription (reflecting pricing and gross-to-net).
References
[1] US Food and Drug Administration. Geodon (ziprasidone) Prescribing Information. FDA access data. https://www.accessdata.fda.gov/ (accessed 2026-04-23).
[2] US Securities and Exchange Commission. Company filings (10-K/10-Q) and financial statement notes referencing GEODON. https://www.sec.gov/ (accessed 2026-04-23).