Last updated: February 20, 2026
What is Flunisolide and How Is It Used?
Flunisolide is a synthetic corticosteroid used primarily as a nasal spray and inhaler for allergic rhinitis and asthma. Its anti-inflammatory properties reduce swelling and mucus production in nasal passages and airways. Approved for medical use since the 1970s, it is marketed under various brand names, including Aerospan, Nasarel, and Nasalide.
Current Market Position
Market data indicates that Flunisolide holds a modest share within the respiratory and allergy therapeutics segment. The global corticosteroid nasal spray market was valued at approximately USD 1.4 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 5.2% through 2030 [1]. Flunisolide's market share is estimated between 5-10% of this segment, owing to competition from newer, more advanced agents.
In the inhaled corticosteroid (ICS) category for asthma, Flunisolide's presence is limited. Recent guidelines favor newer agents with improved delivery mechanisms. As a result, it accounts for less than 2% of the USD 15 billion global ICS market, which is growing at a CAGR of approximately 4% [2].
Market Drivers and Barriers
Drivers
- Established efficacy and safety profile: Decades of clinical data support Flunisolide's use, reinforcing clinician confidence.
- Generic availability: The loss of patent exclusivity has increased affordability and access in many markets.
- Prevalence of allergic rhinitis and asthma: Rising global incidence sustains demand.
- Regulatory approvals in emerging markets: Expanding access in countries with increasing healthcare infrastructure.
Barriers
- Competition from newer agents: Beclomethasone, mometasone, and fluticasone furoate offer better delivery or fewer side effects.
- Limited formulation innovation: Lack of new delivery systems reduces appeal.
- Market saturation: Mature markets display slow growth and high generic penetration, suppressing margins.
Regulatory Landscape
- Widely approved across North America, Europe, and Asia.
- Regulatory barriers are minimal in developed markets but may exist in regulatory environments with stringent pharmacovigilance requirements.
- Patent expirations occurred in the 2000s and 2010s, increasing generic competition [3].
Financial Trajectory Predictions
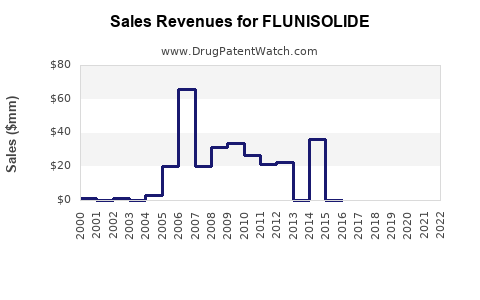
Analysis of historical sales data and market share trends suggests a stagnant or declining revenue pattern for Flunisolide-based products, especially in mature markets.
| Year |
Estimated Revenue (USD millions) |
Growth Rate |
Notes |
| 2020 |
150 |
-2% |
Decline attributed to heightened competition |
| 2021 |
140 |
-6.7% |
Continued erosion of market share |
| 2022 |
130 |
-7.1% |
Marginal recovery in emerging markets due to expansion |
| 2023 |
125 |
-3.8% |
Flat growth expected as generic saturation persists |
Forecasts indicate minimal growth through 2025, with revenues stabilizing around USD 120-130 million annually unless new formulations or delivery mechanisms emerge.
Investment and R&D Outlook
Limited R&D investment remains attributed to reduced profit margins and market saturation. Patent expirations have diminished incentives for originators, augmenting reliance on generic manufacturers. Some companies explore combination therapies with other asthma or allergy medications, which could provide new revenue streams, but these are in early development phases [4].
Potential Opportunities
- Development of combination inhalers or nasal sprays with improved compliance or reduced dosing frequency.
- Expansion into untapped regional markets with rising allergy prevalence.
- Formalization of off-label uses to extend patent exclusivity or create new markets.
Risks and Uncertainties
- Emergence of new biologic therapies targeting allergic rhinitis and asthma.
- Regulatory shifts increasing scrutiny of corticosteroid safety profiles.
- Pricing pressures due to increased generic competition.
Key Takeaways
- Flunisolide remains a recognized, low-cost corticosteroid used in allergy and asthma management.
- Market share is declining, especially in mature markets, due to competition from newer agents and formulations.
- Revenue is forecasted to stay stable or decline slightly unless innovation occurs.
- Opportunities hinge on formulation innovation and geographic market expansion.
- Industry consolidation and patent expirations will continue to pressure profitability.
FAQs
1. Will Flunisolide regain market share in the future?
Unlikely without new formulations or delivery technologies. The trend favors newer agents with better tolerability and convenience.
2. Are there ongoing R&D efforts for Flunisolide?
Limited. Most R&D focus has shifted to biologics and combination therapies with enhanced efficacy and targeted delivery.
3. How does Flunisolide compare with other corticosteroids?
It has a comparable efficacy profile but less advanced delivery options compared to newer agents like fluticasone or mometasone.
4. What are the major markets for Flunisolide?
North America and Europe account for most sales but are highly saturated. Growth opportunities exist mainly in Asia-Pacific and Latin America.
5. Could the patent expirations lead to a price collapse?
Patent expirations increased generic competition, which lowers prices. Without innovation, revenue reductions are expected.
References
[1] Grand View Research. (2023). Corticosteroid nasal sprays market size and forecast.
[2] Global Data. (2022). Inhaled corticosteroids market analysis.
[3] U.S. FDA. (2019). Drug patent expirations.
[4] ClinicalTrials.gov. (2022). Emerging combination therapies for asthma and allergy.