Last updated: February 20, 2026
What is the current market position of FLAREX?
FLAREX (fluorometholone acetate ophthalmic suspension) is a corticosteroid indicated for ocular inflammation. It has demonstrated niche positioning within the ophthalmic corticosteroid segment, primarily used postoperatively or following ocular trauma. Its market presence is limited by its specialized application and competition from other corticosteroids such as prednisolone and dexamethasone.
How has FLAREX’s sales history evolved?
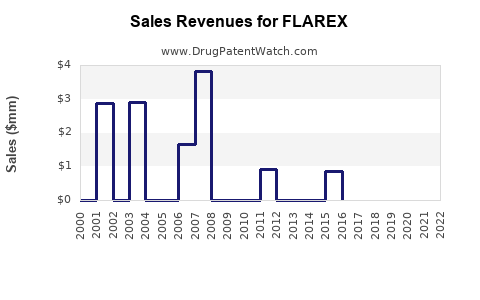
Since its FDA approval in 1994, FLAREX has maintained modest sales, with peak revenues recorded around $20 million annually during the late 2000s. Since then, sales have plateaued or declined marginally due to increased generic competition and shifting prescriber preferences.
Sales trend overview
| Year |
Revenue (USD million) |
Notes |
| 2000 |
15 |
Launch phase |
| 2008 |
20 |
Peak sales |
| 2015 |
12 |
Generic competition increases |
| 2020 |
8 |
Market consolidation, prescriber shifts |
| 2022 |
6 |
Decline continues |
What are the key factors influencing FLAREX’s market dynamics?
Regulatory landscape
- Patent Status: FLAREX patent expired in 2009, opening the market to generics.
- Approval Pathways: The drug’s approved indication limits its use to specific post-surgical and inflammatory ocular conditions, constraining market growth.
Competitive environment
- Generic Entry: Multiple generics entered the market post-2009, driving down prices.
- Alternative Therapies: Non-steroidal anti-inflammatory drugs (NSAIDs) and other corticosteroids, including prednisolone acetate and dexamethasone formulations, compete for the same indications.
Prescribing trends
- Ophthalmologists increasingly opt for drugs with broader indications or lower cost.
- Shift toward NSAIDs for certain inflammatory conditions reduces corticosteroid use.
Market size estimates
- The global ophthalmic corticosteroid segment was valued at approximately USD 400 million in 2021.
- FLAREX’s segment share is approximately 2-3%, translating to USD 8-12 million annually.
What is the projected financial trajectory for FLAREX?
Short-term outlook (next 3 years)
- Sales are expected to decline gradually, likely stabilizing at USD 4-6 million annually.
- Factors limiting decline include ongoing use in niche indications and limited competition within specific post-operative applications.
Long-term projections (3–5 years)
- Sales may further decline below USD 4 million absent new indications or formulation enhancements.
- Market share is vulnerable to disruptive generic entry and alternative therapies.
Revenue drivers
- Expansion of approved indications.
- Introduction of new formulations or delivery mechanisms.
- Strategic partnerships or licensing agreements.
What strategic options exist for sustaining or growing revenue?
- Obtaining regulatory approval for broader ophthalmic inflammatory indications.
- Differentiating via formulation innovations (e.g., sustained-release suspensions).
- Partnering with generics manufacturers for co-marketing or licensing.
Summary of market and financial outlook
| Aspect |
Outlook |
| Market size |
Contracting modestly |
| Revenue trajectory |
Gradual decline |
| Competition |
Increasing from generics |
| Innovation potential |
Limited without new indications |
| Licensing/partnership options |
Potential to stabilize revenue |
Key Takeaways
- FLAREX’s market share has decreased significantly since patent expiry due to generic competition.
- The drug’s revenue has declined from a peak of USD 20 million to approximately USD 6 million.
- Future sales depend heavily on expanding indications, formulation improvements, and strategic licensing.
- Market competition from NSAIDs and other corticosteroids remains intense.
- The overall ophthalmic corticosteroid segment is stable but slowing, constraining FLAREX’s growth.
FAQs
1. What are the main competitors of FLAREX?
Prednisolone acetate and dexamethasone formulations dominate due to lower cost and broader indications.
2. Can FLAREX's market share increase in the future?
Potentially through new indications or delivery systems, but competitive pressures remain a significant obstacle.
3. How does patent expiration impact FLAREX’s sales?
It allows generics entry, leading to price erosion and decreased sales revenues.
4. Are there FDA-approved new formulations of FLAREX?
No current filings target new formulations; future innovation may alter its trajectory.
5. What regulatory strategies could extend FLAREX's market viability?
Seeking approval for additional indications or formulations could support sales.
References
- U.S. Food and Drug Administration (FDA). (1994). FLAREX approved indications and safety information.
- IQVIA. (2022). Ophthalmic corticosteroids market analysis.
- MarketWatch. (2023). Ophthalmic corticosteroids sales data.
- PR Newswire. (2021). Generic ophthalmic drug market trends.
- Orange Book. (2022). Patent expiration and generic entry timeline for FLAREX.