FEXOFENADINE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Fexofenadine, and what generic alternatives are available?
Fexofenadine is a drug marketed by Barr, Aurobindo Pharma Ltd, P And L, Contract Pharmacal, Dr Reddys Labs Ltd, L Perrigo Co, Rising, Teva, Granules, Hetero Labs Ltd V, Sciegen Pharms, Sun Pharm Inds, Time Cap Labs, Unique, Aurobindo Pharma, Dr Reddys, Impax Pharms, and Sun Pharm. and is included in twenty-five NDAs.
The generic ingredient in FEXOFENADINE is fexofenadine hydrochloride; pseudoephedrine hydrochloride. There are twenty-three drug master file entries for this compound. Twenty-nine suppliers are listed for this compound. Additional details are available on the fexofenadine hydrochloride; pseudoephedrine hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Fexofenadine
A generic version of FEXOFENADINE was approved as fexofenadine hydrochloride; pseudoephedrine hydrochloride by DR REDDYS LABS LTD on June 22nd, 2011.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for FEXOFENADINE?
- What are the global sales for FEXOFENADINE?
- What is Average Wholesale Price for FEXOFENADINE?
Summary for FEXOFENADINE
| US Patents: | 0 |
| Applicants: | 18 |
| NDAs: | 25 |
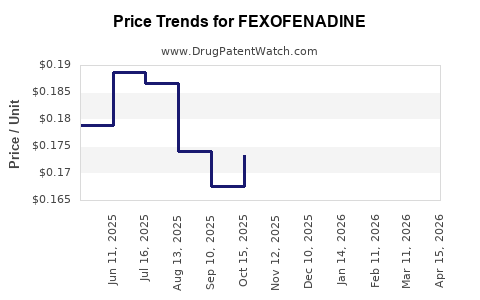
| Drug Prices: | Drug price information for FEXOFENADINE |
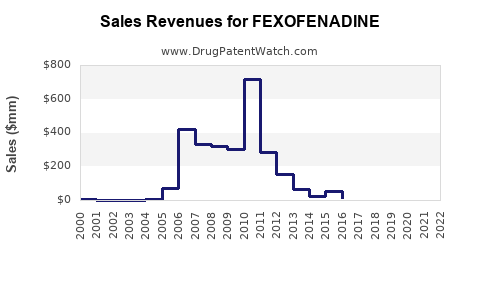
| Drug Sales Revenues: | Drug sales revenues for FEXOFENADINE |
| DailyMed Link: | FEXOFENADINE at DailyMed |