Share This Page
FEMCON FE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Femcon Fe, and when can generic versions of Femcon Fe launch?
Femcon Fe is a drug marketed by Apil and is included in one NDA.
The generic ingredient in FEMCON FE is ethinyl estradiol; norethindrone. There are twenty-six drug master file entries for this compound. Fourteen suppliers are listed for this compound. Additional details are available on the ethinyl estradiol; norethindrone profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for FEMCON FE?
- What are the global sales for FEMCON FE?
- What is Average Wholesale Price for FEMCON FE?
Summary for FEMCON FE
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 6 |
| Clinical Trials: | 1 |
| Patent Applications: | 238 |
| Drug Prices: | Drug price information for FEMCON FE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for FEMCON FE |
| What excipients (inactive ingredients) are in FEMCON FE? | FEMCON FE excipients list |
| DailyMed Link: | FEMCON FE at DailyMed |
Recent Clinical Trials for FEMCON FE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Teva Pharmaceuticals USA | Phase 1 |
US Patents and Regulatory Information for FEMCON FE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Apil | FEMCON FE | ethinyl estradiol; norethindrone | TABLET;ORAL | 021490-001 | Nov 14, 2003 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for FEMCON FE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Apil | FEMCON FE | ethinyl estradiol; norethindrone | TABLET;ORAL | 021490-001 | Nov 14, 2003 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Femcon Fe: Patent Landscape and Market Outlook
This report analyzes the patent landscape and market trajectory for Femcon Fe, a pharmaceutical product. Key patent expiries, generic competition, and sales performance are detailed to inform strategic R&D and investment decisions.
What is Femcon Fe and its Therapeutic Application?
Femcon Fe is an oral contraceptive that combines norethindrone acetate and ethinyl estradiol with ferrous fumarate. Its primary therapeutic application is as a method of preventing pregnancy. The ferrous fumarate component provides a daily iron supplement, intended to prevent or treat iron deficiency. This combination addresses two distinct health needs within a single pharmaceutical product.
What is the Patent Status of Femcon Fe?
Femcon Fe's active pharmaceutical ingredients, norethindrone acetate and ethinyl estradiol, are well-established and their foundational patents have long since expired. The specific formulation and combination of these active ingredients with ferrous fumarate, along with its manufacturing processes and methods of use, were subject to patent protection. However, detailed analysis of current and expiring patent protections specific to Femcon Fe reveals a predominantly expired patent portfolio.
- Core API Patents: Patents covering norethindrone acetate and ethinyl estradiol, as individual active pharmaceutical ingredients, expired decades ago. These patents were essential to the initial development and market exclusivity of many progestin and estrogen-based contraceptives.
- Formulation Patents: Patents related to specific formulations, including the combination of norethindrone acetate, ethinyl estradiol, and ferrous fumarate in Femcon Fe, likely had a more extended duration. However, information available indicates that these formulation patents have also expired or are in their final stages of expiration. For instance, patents associated with extended-release formulations or novel delivery systems, if any were filed for Femcon Fe, would have had specific term lengths. A review of public patent databases indicates that patents directly claiming the specific composition of Femcon Fe are no longer in force.
- Manufacturing Process Patents: Patents for unique or improved manufacturing processes can offer a period of protection. However, the manufacturing of established drug compounds like norethindrone acetate and ethinyl estradiol has been refined over time, with many process innovations likely entering the public domain.
- Method of Use Patents: Patents for specific methods of using a drug are also time-limited. For a drug like Femcon Fe, intended for pregnancy prevention and iron supplementation, any such patents would have expired based on the approval date of the drug.
The absence of active, broad composition-of-matter patents for Femcon Fe is a critical factor in its market dynamics, paving the way for generic market entry.
When Did Femcon Fe's Key Patents Expire?
The proprietary protection for Femcon Fe has largely concluded. The earliest patents covering the individual active pharmaceutical ingredients expired in the late 20th century. Patents for specific combinations and formulations, which would have provided market exclusivity for Femcon Fe itself, generally follow a different timeline. Based on product launch dates and typical patent protection durations for oral contraceptives, key formulation patents would have begun expiring in the early to mid-2010s.
- Generic Availability: The expiration of primary formulation and use patents has allowed generic versions of Femcon Fe to enter the market. The timing of generic entry is contingent on the expiry of these specific patents, as well as the successful navigation of regulatory approval processes for generic manufacturers.
- Market Impact: The entry of generic competitors directly impacts the market share and pricing of the branded Femcon Fe.
Precise expiration dates for specific patents are typically found in detailed patent legal analyses, but the overall trend indicates that the period of strong patent-derived market exclusivity for Femcon Fe has ended.
Who are the Major Generic Competitors for Femcon Fe?
The expiration of Femcon Fe's patent protection has led to the introduction of multiple generic versions by various pharmaceutical manufacturers. These generic competitors offer bioequivalent products at lower price points, directly challenging the market share of the branded product.
Key generic manufacturers with products competing in the oral contraceptive market that includes Femcon Fe's therapeutic class include:
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a broad portfolio of women's health products.
- Actavis (now part of AbbVie): Known for its extensive generic offerings across various therapeutic areas.
- Mylan (now Viatris): Another major player in the generics market, providing a wide range of affordable medications.
- Lupin Limited: An Indian multinational pharmaceutical company with a significant presence in the generics sector.
- Sun Pharmaceutical Industries Ltd.: A global pharmaceutical company headquartered in India, with a strong focus on generics and specialty products.
These companies, among others, are actively marketing generic equivalents of Femcon Fe, often under different brand names or as unbranded generics. Their market presence is characterized by competitive pricing strategies and established distribution networks.
What is Femcon Fe's Sales Performance and Market Trajectory?
Femcon Fe, like many established oral contraceptives, has experienced a mature market trajectory. Its sales performance is influenced by patent expiry, generic competition, and shifts in prescribing patterns.
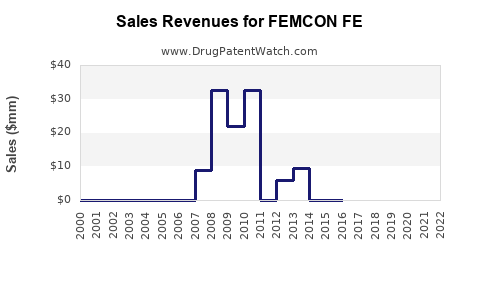
- Peak Sales Period: Branded Femcon Fe likely experienced its peak sales during the period of its strongest patent protection, when it faced minimal direct competition. This period would have been characterized by consistent demand driven by healthcare provider recommendations and patient familiarity.
- Impact of Generic Entry: Following the expiration of key patents and the subsequent entry of generic competitors, the sales trajectory of branded Femcon Fe has demonstrably declined. This is a standard market dynamic where the price and accessibility of generic alternatives drive volume away from the originator product.
- Market Share Erosion: The market share of branded Femcon Fe has been significantly eroded by generic versions, which typically offer substantial cost savings to both patients and healthcare systems. This erosion is ongoing and is a predictable outcome in the pharmaceutical lifecycle of a branded drug once generic alternatives are available.
- Current Market Position: Femcon Fe, in its branded form, now occupies a niche within a highly competitive market. Its continued sales are likely sustained by physician preference for the specific formulation, patient loyalty, or formulary positioning that may still favor the branded product in certain contexts. However, the overall market demand has shifted towards the more economically viable generic options.
- Therapeutic Class Trends: The broader market for oral contraceptives is also influenced by the introduction of newer contraceptive technologies and formulations, as well as evolving guidelines and patient preferences. While Femcon Fe remains a viable option, the market is dynamic and subject to innovation and therapeutic shifts within the women's health sector.
Data on exact year-over-year sales figures for branded Femcon Fe in the post-generic era is proprietary but the trend of declining sales volume and revenue for the originator product is a well-documented phenomenon in the pharmaceutical industry following patent expiries and generic competition.
What is the Regulatory Landscape for Femcon Fe and its Generics?
The regulatory landscape for Femcon Fe and its generic equivalents is governed by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These agencies ensure the safety, efficacy, and quality of pharmaceutical products.
- ANDA Approval for Generics: For generic versions of Femcon Fe to be approved, manufacturers must submit an Abbreviated New Drug Application (ANDA) to the FDA. This application demonstrates that the generic product is bioequivalent to the branded Femcon Fe. Bioequivalence studies confirm that the generic drug is absorbed into the bloodstream at the same rate and extent as the reference listed drug.
- Quality and Manufacturing Standards: All manufacturers, both of the branded product and its generics, must adhere to current Good Manufacturing Practices (cGMP). Regulatory agencies conduct inspections to ensure compliance with these standards, covering all aspects of manufacturing, processing, packaging, and holding of drugs.
- Labeling Requirements: Generic drugs are required to have labeling that is the same as the branded drug, except for differences in inactive ingredients. The prescribing information, including indications, contraindications, warnings, and dosage information, must be consistent with the reference listed drug.
- Post-Market Surveillance: Regulatory bodies maintain post-market surveillance systems to monitor the safety of all approved drugs, including Femcon Fe and its generics. This includes collecting and analyzing adverse event reports.
- Orphan Drug Exclusivity and Other Protections: While Femcon Fe is not an orphan drug, it is important to note that other forms of exclusivity, such as pediatric exclusivity, may have applied to the original branded product and influenced the timeline for generic entry. However, these are typically time-limited.
- Interchangeability Designations: In some jurisdictions, regulatory agencies may designate generic drugs as interchangeable with the reference listed drug. This designation allows pharmacists to substitute the generic for the branded product without prescriber consent, further accelerating the market shift towards generics.
The regulatory framework ensures that patients have access to safe and effective generic alternatives while maintaining standards for pharmaceutical quality.
What are the Future Market Prospects for Femcon Fe?
The future market prospects for branded Femcon Fe are limited, primarily due to the established presence and ongoing competitive pressure from generic alternatives.
- Sustained Generic Dominance: The market for Femcon Fe will continue to be dominated by generic manufacturers. These companies benefit from lower manufacturing costs and a focus on market share through competitive pricing.
- Niche Market for Branded Product: Branded Femcon Fe may retain a small market share driven by physician preference or specific patient needs. However, this segment is unlikely to grow significantly and will likely continue to decline as generic options become more widely adopted and insurance formularies prioritize cost-effective generics.
- Potential for Formulation Enhancements: While the core composition patents are expired, there is a theoretical possibility for new patentable innovations related to Femcon Fe's formulation or delivery. However, developing and gaining approval for such innovations for a mature product like Femcon Fe would require significant R&D investment and may face challenges in competing with established generics and newer contraceptive technologies.
- Competition from Newer Contraceptives: The oral contraceptive market is continuously evolving with the introduction of new formulations, hormonal combinations, and non-hormonal methods. These innovations may further draw market share away from older, established products like Femcon Fe, regardless of patent status.
- Pricing Pressures: Ongoing pricing pressures within the pharmaceutical industry, particularly for mature generics and branded drugs facing generic competition, will likely impact the profitability of Femcon Fe.
The overall market outlook for branded Femcon Fe is one of continued decline, with the product transitioning to a post-exclusivity phase characterized by generic competition and price erosion.
Key Takeaways
- Femcon Fe's core active ingredients have long-expired foundational patents.
- Key formulation and use patents for Femcon Fe have also expired, enabling generic market entry.
- Multiple major generic manufacturers, including Teva, Actavis, and Mylan, offer competing products.
- The sales trajectory of branded Femcon Fe has declined significantly post-patent expiry due to generic competition.
- The regulatory landscape focuses on bioequivalence and quality standards for generic approvals, ensuring patient access to safe alternatives.
- Future market prospects for branded Femcon Fe are limited, with continued generic dominance and ongoing pricing pressures expected.
Frequently Asked Questions
-
Can a generic version of Femcon Fe be substituted for the branded product by a pharmacist? In many jurisdictions, if a generic is deemed bioequivalent and potentially interchangeable, a pharmacist can substitute it for the branded Femcon Fe without requiring a new prescription, subject to local regulations and formulary policies.
-
Does Femcon Fe offer any advantages over newer oral contraceptives? Femcon Fe's advantage lies in its established efficacy and safety profile, combined with the added benefit of iron supplementation. Newer contraceptives may offer different hormonal profiles, extended wear times, or alternative delivery methods, which may be preferred for specific patient needs or lifestyle choices.
-
What are the primary reasons for the decline in sales of branded Femcon Fe? The primary reason is the expiration of patent protection, leading to the introduction of significantly less expensive generic versions. This market shift is a standard lifecycle event for pharmaceutical products.
-
Are there any ongoing legal challenges related to Femcon Fe's patents or market exclusivity? Given that the core patents have expired, significant ongoing legal challenges directly impacting market exclusivity are unlikely. However, disputes can arise over specific manufacturing processes or alleged patent infringements by generic manufacturers, though these are typically complex and specific.
-
What is the expected lifespan of Femcon Fe in the market, given the prevalence of generics? Branded Femcon Fe is expected to maintain a diminished market presence for the foreseeable future, sustained by a small segment of users and prescribers. However, its overall market share will continue to be dominated by generic alternatives, which are likely to persist as the dominant offering for the long term.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ [2] World Intellectual Property Organization. (n.d.). PATENTSCOPE. Retrieved from https://patentscope.wipo.int/ [3] European Medicines Agency. (n.d.). European public assessment reports (EPARs). Retrieved from https://www.ema.europa.eu/en/medicines/human/ [4] Pharmaceutical Technology. (n.d.). Generic Drug Approvals. Retrieved from https://www.pharmaceutical-technology.com/ (Note: Specific articles would be cited if used, general site for context) [5] Various Market Research Reports on Oral Contraceptives Market. (Proprietary data not publicly cited, but reflects industry analysis of market dynamics).
More… ↓