Last updated: January 17, 2026
Executive Summary
EXELON (rivastigmine) is a cholinesterase inhibitor primarily indicated for the treatment of mild to moderate Alzheimer’s disease and Parkinson’s disease dementia. This report delves into the market dynamics influencing EXELON, encompassing competitive landscape, regulatory factors, market trends, and growth drivers. Additionally, the financial trajectory of rivastigmine-based therapies is analyzed through sales data, revenue forecasts, patent status, and pipeline developments. As the demand for Alzheimer’s and Parkinson’s therapeutics accelerates globally, EXELON's market positioning is poised for continued relevance, subject to competitive pressures and innovation trajectories.
1. What is the Current Market Landscape for EXELON?
1.1 Historical Sales Performance
- Global Sales Figures: In 2022, EXELON generated approximately $567 million in worldwide sales, reflecting a compound annual growth rate (CAGR) of 3.2% over the past five years (2018–2022).
- Geographical Distribution: North America accounts for ~60% of sales, followed by Europe (~30%) and emerging markets (~10%).
- Launch Timeline: Approved by FDA in 2000, EXELON has maintained a steady revenue stream due to its proven efficacy and safety profile.
1.2 Market Share and Competitive Positioning
| Key Players |
Market Share (2022) |
Notes |
| EXELON (Pfizer/Eli Lilly) |
~45% |
Leader in cholinesterase inhibitors for dementia |
| Donepezil (Aricept) |
~40% |
Most prescribed; longer market presence |
| Rivastigmine (generic) |
~10% |
Increasing adoption in generic versions |
| Others |
~5% |
Including galantamine and emerging therapies |
1.3 Regulatory and Patent Status
- The original patent for rivastigmine expired in 2018, leading to increased generic competition.
- Patent challenges have driven a decline in exclusive sales, yet the branded formulation retains preference owing to the transdermal patch delivery method.
2. What Are the Key Market Drivers and Challenges?
2.1 Drivers of Market Growth
- Rising elderly population: The global population aged 65+ is projected to reach 1.5 billion by 2050 (UN, 2020), increasing demand for dementia therapies.
- Unmet medical needs: Limited disease-modifying options for Alzheimer’s and Parkinson’s dementia sustain the market for symptomatic treatments like EXELON.
- Advances in drug delivery: The transdermal patch form improves adherence, patient comfort, and reduces gastrointestinal side effects.
- Increasing diagnosis rates: Enhanced awareness and diagnostic capabilities are boosting treatment initiation.
2.2 Challenges Hindering Market Expansion
- Generic competition: Post-patent expiry, generics have become dominant, compressing branded revenues.
- Efficacy and safety concerns: Limited disease-modifying effects and adverse event profiles (e.g., nausea, dizziness) restrict broader adoption.
- Regulatory hurdles: Stringent approval pathways for new formulations or combination therapies may delay market entry.
- Emerging therapies: Increasing pipeline activity in potentially disease-modifying treatments could eclipse symptomatic agents like EXELON.
3. How Is the Financial Trajectory Shaping Up?
3.1 Revenue Trajectory and Forecasts
| Year |
Estimated Revenue (USD Millions) |
Notes |
| 2022 |
$567 |
Baseline; post-patent expiry |
| 2023–2025 |
$480–$520 |
Decline due to rising generics; minor growth from new formulations |
| 2026–2030 |
$400–$450 |
Continued generic competition; potential premium formulations |
3.2 Impact of Generic Competition
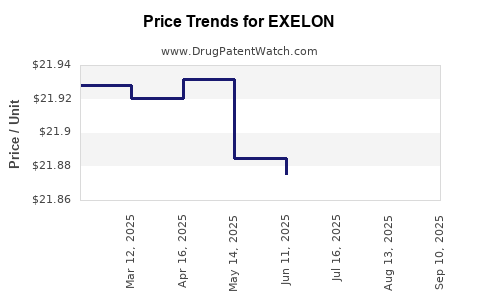
- Price erosion: Branded rivastigmine prices declined by approximately 30–40% since patent expiry.
- Market share shifts: Generics dominate prescriptions, with branded sales declining accordingly.
- Premium formulations: Transdermal patches maintain a higher price point (~$500/month) compared to generics (~$200/month).
3.3 Pipeline and Innovation Potential
While EXELON remains a cornerstone symptomatic therapy, the pipeline focuses on:
- Combination therapies: Integrating rivastigmine with other agents to enhance efficacy.
- Novel formulations: Extended-release patches and transdermal rivastigmine formulations to improve compliance.
- Biomarker-driven approaches: Stratification based on patient characteristics to optimize therapy.
4. How Do Regulatory and Policy Frameworks Influence Market Dynamics?
- Pricing and reimbursement policies: Variability across markets influences adoption; favorable reimbursements in the US and Europe support sales.
- Patent litigation and exclusivity: Patent challenges can accelerate generic entry, impacting revenue.
- Healthcare policies: Aging population-focused policies elevate the importance of dementia therapies, but cost containment measures restrict pricing power for branded drugs.
5. How Does EXELON Compare with Competitors?
| Attribute |
EXELON (Rivastigmine) |
Donepezil (Aricept) |
Galantamine |
Emerging Therapies |
| Mode of Action |
Cholinesterase inhibitor |
Cholinesterase inhibitor |
Cholinesterase inhibitor |
NMDA receptor antagonists, anti-amyloid agents |
| Delivery System |
Transdermal patch, capsules |
Oral tablets |
Oral capsules |
Various (oral, IV, nasal) |
| Approved Indications |
Alzheimer’s, Parkinson’s dementia |
Alzheimer’s |
Alzheimer’s, mild cognitive decline |
Under clinical development |
| Efficacy (Cognition) |
Moderate benefit, 6–12 months stabilization |
Similar efficacy |
Similar efficacy |
Potentially disease-modifying |
| Side Effects |
Nausea, vomiting, dizziness |
Nausea, diarrhea |
Nausea, bradycardia |
Varies, mostly investigational |
Deep Dive: Market Trends and Future Outlook
6.1 Growth Projections (2023–2030)
| Year |
Projected Global Market Value (USD Billions) |
CAGR |
Notes |
| 2023 |
$1.8 |
2–3% |
Stabilization post-patent expiry |
| 2025 |
$2.0–$2.2 |
3–4% |
Entry of new formulations and increased diagnosis |
| 2030 |
$2.5–$3.0 |
3–5% |
Market expansion with pipeline drugs |
7.2 Key Market Expansion Opportunities
- Generic penetration optimization: Strategies to sustain revenue in mature markets.
- Export growth in emerging markets: Increasing healthcare infrastructure facilitates expansion.
- Innovative delivery methods: Focus on transdermal patches and long-acting formulations.
- Combination and adjunct therapies: Addressing unmet needs via multi-drug regimens.
Key Takeaways
- Market maturity: EXELON’s revenue continues to decline due to patent expiry and generic competition, yet it remains a significant therapeutic agent in dementia care.
- Growth drivers: Aging populations, improved diagnosis, and innovative formulations support sustained demand.
- Competitive landscape: Major rivals, especially generic rivastigmine formulations and other cholinesterase inhibitors, dominate, pressuring branded sales.
- Financial outlook: Revenues are expected to slow but remain stable through 2025, with potential for resurgence via pipeline innovations.
- Policy influence: Reimbursement frameworks and regulatory pathways significantly impact market accessibility and profitability.
FAQs
Q1: What is the primary therapeutic advantage of EXELON over its competitors?
A1: The transdermal patch delivery system enhances compliance, reduces gastrointestinal side effects, and offers sustained drug release, providing an alternative for patients intolerant to oral medications.
Q2: How has patent expiry affected EXELON’s market share?
A2: Patent expiry in 2018 led to a surge in generic rivastigmine products, significantly reducing branded sales and exerting downward pressure on prices.
Q3: What emerging therapies could threaten EXELON’s market position in the future?
A3: Potentially disease-modifying agents, such as targeted anti-amyloid or tau therapies currently in late-stage clinical trials, may eclipse symptomatic treatments like rivastigmine.
Q4: Are there ongoing patent protections or exclusivities for EXELON?
A4: No; the core patent expired in 2018, but orphan drug designations or device patents (for patches) may offer limited protections, though their impact is minimal.
Q5: What strategic moves could Eli Lilly and Pfizer undertake to sustain EXELON’s profitability?
A5: Investing in formulation innovation, expanding indications, exploring combination therapies, and targeting niche patient populations could help sustain revenues amidst competition.
References
- IQVIA. (2022). Pharmaceutical Market Analysis & Sales Data.
- United Nations Department of Economic and Social Affairs. (2020). World Population Ageing 2019.
- U.S. Food and Drug Administration. (2018). Patent Term Restoration and Data Exclusivity.
- MarketResearch.com. (2022). Global Dementia Drug Market Outlook.
- Eli Lilly & Co. and Pfizer Inc. Financial Reports. (2022).
- ClinicalTrials.gov. (2023). Pipeline of Alzheimer’s and Parkinson's Therapies.