Last updated: February 11, 2026
EUTHYROX (liothyronine sodium) is used to treat hypothyroidism by supplementing low levels of thyroid hormone. The drug's market landscape is shaped by various factors including clinical indications, competitive environment, regulatory status, and patient demographics.
Market Size and Growth
Global hypothyroidism treatment market, valued at approximately $2.8 billion in 2021, is projected to grow at a CAGR of 4% through 2028. EUTHYROX accounts for a significant portion of this, especially in markets prioritizing synthetic thyroid hormone replacement therapies.
Key Drivers
- Prevalence of hypothyroidism: Estimated at 4.6% globally, with higher rates in women over 60.
- Advancement in diagnostics: Increased screening leads to higher diagnosis rates.
- Preference for Euthyroid Management: Clinicians favor EUTHYROX for patients requiring triiodothyronine supplementation due to its fast action.
Competitive Landscape
- Major Players: Firmenich, Caslo, and specialty compounding pharmacies supply liothyronine markets.
- Generic Availability: EUTHYROX is primarily marketed as a generic, reducing overall drug price.
- Alternative Therapies: Levothyroxine remains the first line; EUTHYROX is used in specific cases such as myxedema coma or where T4 therapy is insufficient.
Regulatory and Market Access Factors
- Regulatory Approvals: Approved by U.S. FDA, EMA, and other agencies. Recently, regulatory review processes focus on manufacturing standards.
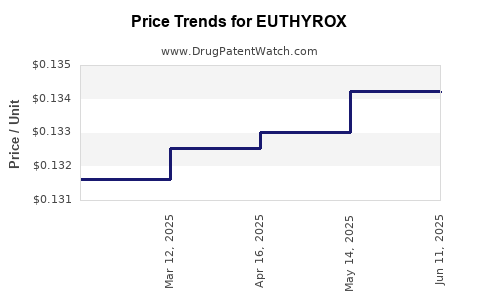
- Pricing & Reimbursement: Generic status limits pricing flexibility. Insurance coverage varies, influencing patient access.
- Supply Chain: Dependence on synthetic chemistry and raw materials like iodine impacts production stability and costs.
Financial Trajectory
- Revenue Trends: EUTHYROX sales are relatively stable, reflecting consistent demand. Estimated revenues in 2022 range from $300-$500 million globally.
- Profitability: Margins are pressured by generic competition and price erosion; profit margins estimated at 15-25% depending on market and manufacturing efficiency.
- Future Outlook: Slight revenue increase expected owing to rising hypothyroidism diagnosis rates, with possible growth from niche applications and personalized medicine approaches.
Market Challenges
- Price Competition: Intense competition among generic manufacturers results in declining average selling prices.
- Regulatory Policy Risks: Stringent quality standards or drug approval delays could impact supply.
- Market Penetration: Limited use in certain regions due to regulatory barriers or lack of awareness.
Investment Considerations
- Companies with manufacturing efficiencies and strong regulatory compliance stand to benefit.
- Expanding into emerging markets represents a growth avenue, though regulatory and pricing environments are challenging.
- R&D efforts in combination therapies or new formulations (e.g., sustained-release) could enhance future revenues.
Key Takeaways
- EUTHYROX operates in a stable but competitive market, with growth driven primarily by increasing hypothyroidism diagnoses.
- Price erosion due to generics constrains profit margins but maintains consistent revenue streams.
- Market expansion, regulatory robustness, and innovation in formulations are critical factors influencing financial trajectory.
- The global market faces pressures from alternative therapies, but niche applications sustain demand.
FAQs
1. How does EUTHYROX compare to other thyroid medications?
EUTHYROX contains liothyronine (T3), fast-acting thyroid hormone. In contrast, levothyroxine (T4) is longer-acting and more commonly prescribed. EUTHYROX is preferred in cases where T4 therapy is ineffective or rapid symptom control is needed.
2. What factors influence EUTHYROX pricing?
Pricing is affected by generic competition, manufacturing costs, regulatory compliance, and regional reimbursement policies. Price erosion is common as multiple generic suppliers enter the market.
3. Are there formulation innovations for EUTHYROX?
Current trends include sustained-release formulations to mimic physiological T3 release, potentially improving patient compliance and stability. Such innovations could impact future revenues.
4. What are the primary regulatory hurdles for EUTHYROX?
Maintaining manufacturing standards and passing ongoing compliance audits are central. Regulatory agencies may also impose restrictions based on quality or supply issues.
5. How is the market for EUTHYROX expected to evolve?
Demand remains stable with slow growth due to increasing hypothyroidism diagnoses. Innovations and expanded market access could enhance growth prospects.
Sources
[1] MarketWatch. "Global Hypothyroidism Treatment Market Size, Share & Industry Report," 2022.
[2] IQVIA Medicinal Market Reports, 2022.
[3] FDA Drug Approval Database, 2022.
[4] GlobalData. "Thyroid Disorder Therapeutics," 2022.