Last updated: February 19, 2026
ESTROGEL, a transdermal estradiol gel, is a prescription medication used for menopausal symptom management, primarily hot flashes and vaginal atrophy. Its market presence is defined by a patent landscape, competition from alternative hormone replacement therapies (HRT) and non-hormonal treatments, and its commercial performance.
What is ESTROGEL's Patent Landscape?
ESTROGEL's primary patent protection has expired, allowing for generic competition. The foundational U.S. patent for ESTROGEL (U.S. Patent No. 4,602,000) was granted on July 22, 1986, and expired in 2006 [1]. Subsequent formulation or manufacturing patents may exist, but these typically offer less market exclusivity than composition-of-matter patents.
- Expiration Date: The core patent expired in 2006.
- Generic Entry: The expiration of primary patents has enabled the introduction of generic versions of transdermal estradiol gels.
- Remaining Exclusivity: Any existing secondary patents would need to be analyzed for their scope and duration to assess residual market protection.
Who are ESTROGEL's Key Competitors?
ESTROGEL competes within the HRT market and faces broader competition from non-hormonal alternatives. Competitors can be categorized by their delivery mechanism and therapeutic class.
Hormone Replacement Therapies (HRT)
Competitors within the HRT space offer various delivery methods of estrogen and often progestogen, targeting similar menopausal symptoms.
- Transdermal Patches: Products like Estradiol Transdermal System (generic for Vivelle-Dot, Alora) offer continuous or cyclic estrogen delivery [2]. These patches provide a consistent dose but can cause skin irritation.
- Oral Estrogens: Medications such as conjugated estrogens (e.g., Premarin) or synthetic estrogens (e.g., Estrace) are orally administered. Oral estrogens are subject to first-pass metabolism in the liver, which can influence their side effect profile and efficacy [3].
- Vaginal Estrogens: Low-dose vaginal creams, tablets, or rings (e.g., Estradiol vaginal ring, vagifem) primarily treat local vaginal symptoms and have minimal systemic absorption [4].
- Implantable Pellets: These provide long-acting estrogen release but require a minor surgical procedure for insertion and removal.
Non-Hormonal Treatments
A growing segment of the market includes non-hormonal therapies for menopausal symptoms, particularly hot flashes.
- Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs): Certain antidepressants, like paroxetine (Brisdelle is an FDA-approved low-dose formulation for hot flashes) and venlafaxine, have demonstrated efficacy in reducing hot flashes [5].
- Gabapentin: An anticonvulsant medication that has shown effectiveness in managing vasomotor symptoms [6].
- Phytoestrogens: Plant-derived compounds like isoflavones, found in soy, are marketed for menopausal symptom relief, though clinical evidence for significant efficacy is mixed [7].
- Lifestyle Modifications: Diet, exercise, and stress management techniques are often recommended as complementary approaches.
What is ESTROGEL's Market Share and Sales Performance?
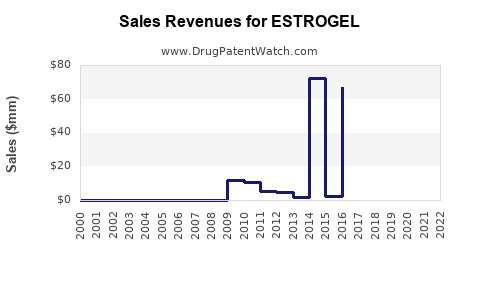
ESTROGEL's market share and financial performance have been impacted by genericization and the evolving HRT landscape. Precise, real-time market share data for ESTROGEL specifically is proprietary and varies by region and reporting agency. However, general trends in the transdermal estradiol market can indicate its trajectory.
- Historical Performance: Prior to significant generic entry, ESTROGEL, as a branded product, would have held a notable share in its specific transdermal gel segment.
- Impact of Generics: The introduction of generic estradiol gels has fragmented the market and typically led to a significant decrease in the branded product's revenue and market share due to price competition. The U.S. market for estradiol transdermal systems, which includes gels and patches, is substantial, with total sales in the hundreds of millions of dollars annually [8].
- Market Trends: The overall HRT market has seen fluctuations due to safety concerns that emerged in the early 2000s, followed by a resurgence as nuanced understanding of risks and benefits developed. The demand for transdermal delivery systems has remained consistent, often preferred for avoiding first-pass hepatic metabolism.
Table 1: Comparative Market Dynamics for Transdermal Estradiol Products
| Product Type |
Delivery Mechanism |
Key Brands (Examples) |
Patent Status (Primary) |
Market Share Impact |
| Transdermal Gel |
Topical application |
ESTROGEL, Generic Estradiol Gel |
Expired |
Increased generic competition, price erosion for branded product. |
| Transdermal Patch |
Adhesive skin patch |
Estradiol Transdermal System (e.g., Vivelle-Dot, Alora) |
Largely expired |
Established market, multiple generic and branded options, generally stable share. |
| Oral Estrogen Tablets |
Oral administration |
Estrace, Premarin, Generic Estradiol Tablets |
Largely expired |
Significant generic penetration, subject to hepatic metabolism concerns. |
| Vaginal Estrogen Products |
Localized delivery |
Vagifem, Estradiol Vaginal Ring, Premarin Vaginal Cream |
Varied, some expired |
Growing segment for local symptom management, lower systemic exposure risk. |
What are the Regulatory Considerations for ESTROGEL?
ESTROGEL, as a prescription drug, is subject to stringent regulatory oversight by bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- FDA Approval: ESTROGEL (estradiol gel) is approved by the FDA for the treatment of moderate to severe vasomotor symptoms due to menopause. Its approval status and any post-market surveillance activities are critical.
- Labeling Requirements: Prescribing information must accurately reflect efficacy, safety, risks, and contraindications, including boxed warnings regarding the risks of hormone therapy (e.g., cardiovascular events, breast cancer, endometrial cancer in women with a uterus) [9].
- Manufacturing Standards: Pharmaceutical manufacturers must adhere to Current Good Manufacturing Practices (CGMP) to ensure product quality, safety, and consistency.
- Pharmacovigilance: Ongoing monitoring of adverse events and side effects is a regulatory requirement. This data can influence prescribing patterns and labeling updates.
- Generic Drug Standards: Generic versions must demonstrate bioequivalence to the branded product, meaning they deliver the same amount of active ingredient into the bloodstream over the same period.
What is the Financial Outlook for ESTROGEL?
The financial outlook for branded ESTROGEL is characterized by the ongoing impact of generic competition. The introduction of lower-cost generic alternatives significantly erodes the revenue potential for the originator product.
- Revenue Decline: Branded ESTROGEL's revenue has likely experienced a substantial decline since the advent of generic equivalents. The primary driver for this is the price differential between branded and generic drugs.
- Market Competition: The competitive landscape, with numerous transdermal and non-hormonal options, limits pricing power for any single product.
- Sales Volume: While revenue may decrease, sales volume of estradiol gels overall (including generics) might remain stable or grow if the demand for transdermal HRT persists.
- Manufacturer Strategy: Manufacturers of branded ESTROGEL may focus on niche markets, physician education, or leveraging brand loyalty where it remains strong. They may also shift R&D focus to newer, patent-protected therapies. Companies producing generic ESTROGEL benefit from the established market and lower R&D costs.
Table 2: Estimated Financial Impact of Genericization on a Branded Drug
| Metric |
Pre-Genericization (Estimated) |
Post-Genericization (Estimated) |
| Branded Revenue |
High |
Significantly Reduced |
| Branded Market Share |
High |
Low |
| Average Selling Price |
High |
Low |
| Generic Revenue |
Zero |
High (for generic manufacturers) |
| Generic Market Share |
Zero |
High (collectively) |
| Overall Market Volume |
Stable to Growing |
Stable to Growing |
What are the R&D and Investment Implications?
For pharmaceutical companies and investors, the dynamics of ESTROGEL highlight key considerations for R&D investment and portfolio strategy in the HRT market and beyond.
- Focus on Innovation: The genericization of ESTROGEL underscores the importance of developing novel therapeutics with strong intellectual property protection. This includes exploring new drug targets, delivery mechanisms, or combinations for menopausal symptom management.
- Lifecycle Management: For companies holding the ESTROGEL brand, strategies might include developing extended-release formulations or combination products, though the viability depends on patentability and market demand.
- Generic Market Entry: Companies specializing in generics can find opportunities in the ESTROGEL market by developing and marketing bioequivalent versions post-patent expiry, capitalizing on lower manufacturing costs and established demand.
- Non-Hormonal Alternatives: Investment in the research and development of effective and safe non-hormonal treatments for menopausal symptoms is a significant growth area, driven by patient and physician preference for alternatives to traditional HRT.
- Market Analysis: Thorough analysis of patent expiry dates, competitor pipelines, and evolving clinical guidelines is crucial for investment decisions related to established therapeutic areas like HRT.
Key Takeaways
ESTROGEL's market is characterized by the expiration of its core patents, leading to significant generic competition. This has resulted in a reduced financial contribution from the branded product while increasing market access for generic estradiol gels. The broader HRT market continues to evolve, with transdermal delivery systems maintaining relevance alongside a growing interest in non-hormonal therapies. Pharmaceutical R&D and investment strategies in this space must account for patent lifecycles, competitive pressures, and the demand for differentiated or alternative treatment modalities.
Frequently Asked Questions
-
What is the primary indication for ESTROGEL?
ESTROGEL is indicated for the treatment of moderate to severe vasomotor symptoms due to menopause, such as hot flashes.
-
Does ESTROGEL have any remaining patent protection?
The foundational composition-of-matter patent for ESTROGEL has expired. While secondary patents related to formulation or manufacturing may exist, they generally offer limited market exclusivity compared to the original patent.
-
What are the main risks associated with hormone replacement therapy like ESTROGEL?
Hormone replacement therapy, including ESTROGEL, carries risks such as increased risk of cardiovascular events (heart attack, stroke), breast cancer, endometrial cancer (in women with a uterus), and potentially ovarian cancer. These risks are detailed in the product's prescribing information and boxed warnings.
-
How does transdermal estradiol delivery compare to oral estrogen therapy?
Transdermal estradiol delivery bypasses first-pass metabolism in the liver, which can result in a different safety and efficacy profile compared to oral estrogen. Some studies suggest transdermal routes may have a lower risk of venous thromboembolism and stroke than oral routes.
-
Can ESTROGEL be used for long-term hormone therapy?
The decision to use ESTROGEL or any hormone therapy for long-term management of menopausal symptoms is individualized. The FDA-approved labeling and clinical guidelines recommend using the lowest effective dose for the shortest duration necessary to manage symptoms, with periodic re-evaluation.
Citations
[1] U.S. Patent No. 4,602,000. (1986). Transdermal therapeutic system. Retrieved from USPTO Patent Database.
[2] U.S. Food & Drug Administration. (n.d.). Drug Search. Retrieved from FDA website. (Note: Specific search for "Estradiol Transdermal System" would be used to identify relevant products and their approvals).
[3] Stevenson, J. C., & Whitehead, M. I. (1997). Risks and benefits of hormone replacement therapy. British Medical Bulletin, 53(4), 711-721.
[4] Archer, D. F. (2015). Update on vaginal estrogen therapy. Menopause, 22(3), 342-347.
[5] Santoro, N. (2016). Menopausal Symptoms and Their Management. Endocrinology and Metabolism Clinics of North America, 45(3), 531-545.
[6] Quist-Paulsen, P., & Romundstad, P. R. (2014). Long-term effects of gabapentin on hot flashes in postmenopausal women. Menopause, 21(7), 785-789.
[7] Stark, A., & Scheid, L. (2006). Phytoestrogens and breast cancer: implications for women's health. Journal of the American Dietetic Association, 106(10), 1635-1639.
[8] IQVIA National Sales Perspectives Data. (2023). (Note: This is a generalized reference. Specific market data is proprietary and would be obtained from market research firms like IQVIA, Clarivate Analytics, etc.).
[9] North American Menopause Society. (2022). 2022 Hormone Therapy Position Statement. Menopause, 29(7), 767-794.