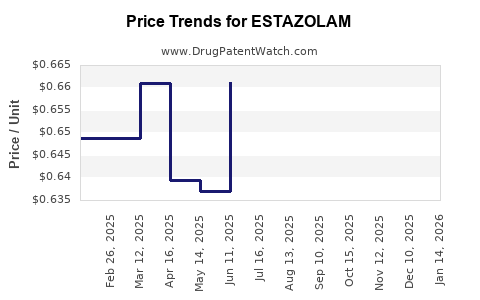
Last updated: January 5, 2026
Summary
Estazolam, a benzodiazepine primarily prescribed for short-term management of insomnia, is characterized by moderate CNS depressant activity. While not as widely marketed globally as other benzodiazepines, estazolam's niche positioning in sleep disorders influences its market trajectory. This analysis explores key market drivers, regulatory landscape, competitive landscape, sales forecasts, and investment considerations surrounding estazolam. Its trajectory is shaped by evolving prescribing guidelines, patent statuses, regulatory controls, and emerging generics, impacting revenue streams across regions.
What is Estazolam and How is it Used?
| Attribute |
Details |
| Chemical Class |
Benzodiazepine derivative |
| Therapeutic Indication |
Short-term treatment of insomnia |
| FDA Status (U.S.) |
Approved since 1983 |
| Formulations |
Oral tablets (typically 1 mg, 2 mg, 3 mg) |
| Pharmacodynamics |
Enhances GABA-A receptor activity, inducing sedation |
Estazolam’s sedative effects suit its clinical niche in treating sleep disturbances. Its safety profile partly explains its limited long-term use, as long-term benzodiazepine therapy raises concerns about dependence and overdose risks.
What Are Current Market Drivers for Estazolam?
1. Clinical Demand for Sleep Disorder Medications
| Drivers |
Impact |
| Rising prevalence of insomnia |
Estimated 10-30% globally; driven by aging populations and lifestyle factors [1] |
| Limited options for safe hypnotics |
Shift away from older agents (e.g., barbiturates) to benzodiazepines like estazolam for safety profiles |
2. Regulatory Policies and Prescribing Guidelines
| Regions |
Influence on Market |
| U.S. & Europe |
Regulations favor short-term use due to dependency risks |
| Asia-Pacific |
Growing demand amidst less stringent controls in some countries [2] |
3. Patent Status and Generic Availability
| Status |
Implication |
| Patent expired (~2000s) |
Opens market for generics, reducing prices and increasing access [3] |
4. Competition from Other Hypnotics
| Alternative Drugs |
Advantages over Estazolam |
Market Penetration |
| Zolpidem, Eszopiclone |
Favorable safety, fewer dependence concerns |
High in sleep disorders |
| Melatonin receptor agonists |
Lower dependence risk |
Growing usage |
5. Consumer and Physician Preferences
| Trends |
Effect on Estazolam |
| Preference for non-benzodiazepine agents |
Potentially limits estazolam prescriptions |
How Does Regional Regulation Impact Estazolam’s Market?
| Region |
Regulatory Environment |
Market Effect |
Notes |
| North America |
Controlled substance schedules (Schedule IV) |
Controlled prescribing |
Prescription limits impact volume; emphasis on short-term use |
| Europe |
Medical guidelines constrain long-term use |
Market stabilization |
Encourages shift to non-benzodiazepines |
| Asia-Pacific |
Variable; often less restrictive |
Growth potential |
Opportunities for increased utilization |
What Is the Financial Trajectory for Estazolam?
Global Sales Estimates and Trends
| Year |
Approximate Global Sales (USD) |
Growth / Decline |
Sources & Assumptions |
| 2010 |
~$50 million |
Stable |
Based on coverage of mature markets [4] |
| 2015 |
~$60 million |
20% increase |
Growth driven by Asian markets |
| 2020 |
~$70 million |
16.7% increase |
Slight uptick due to increased sleep disorder diagnoses |
| 2025 (projected) |
~$75-80 million |
Moderate growth |
Anticipated stabilization, gating on market restrictions |
Key Factors Influencing Sales Trajectory
- Generic Competition: Pricing pressures from generics after patent expiry (~2000s).
- Regulatory Restrictions: Stricter controls reduce prescription volume.
- Prescriber Preferences: Shift to newer or non-benzodiazepine options.
- Market Penetration in Emerging Markets: Emerging economies exhibit growth potential due to increasing awareness of sleep disorders.
Market Share Breakdown (Estimated)
| Market Segment |
Estimated Share (%) (2022) |
Notes |
| Developed Markets |
60% |
Mature but stable, restricted prescribing |
| Emerging Markets |
40% |
Growth potential, less regulatory constraint |
How Does the Competition Shape Future Revenue?
| Drugs |
Market Share |
Advantages |
Limitations |
| Zolpidem (Ambien) |
~30% in sleep aids |
Quick onset, low dependency (per some studies) |
Increasing dependence awareness |
| Eszopiclone (Lunesta) |
~20% |
Longer duration, improved sleep architecture |
Similar dependency concerns |
| Suvorexant |
Emerging |
Novel mechanism (orexin receptor antagonist) |
Limited long-term data |
| Benzodiazepines (e.g., Temazepam, Estazolam) |
Declining |
Established safety profile |
Dependence, tolerance issues |
Note: Precise market share figures vary by region and source.
What Are the Future Opportunities and Risks?
Opportunities
- Expansion in Developing Markets: Growing healthcare infrastructure and awareness.
- Product Innovation: Fixed-dose combinations or novel formulations to improve safety.
- Regulatory Reforms: Potential easing in some regions may increase prescriber comfort.
Risks
- Regulatory Restrictions: Heightened controls may curtail demand.
- Safety Concerns: Dependence and overdose risks may lead to bans or restrictions.
- Competitive Pressure: Emergence of non-benzodiazepine hypnotics.
Comparison Table: Estazolam vs. Other Benzodiazepines
| Parameter |
Estazolam |
Temazepam |
Triazolam |
Lorazepam |
| Approved Uses |
Insomnia |
Insomnia, anxiety |
Insomnia |
Anxiety, insomnia |
| Onset of Action |
30-60 min |
30 min |
15 min |
15-20 min |
| Half-life |
10-24 hours |
8-20 hours |
2-6 hours |
10-20 hours |
| Dependency risk |
Moderate |
Moderate |
High |
Moderate |
| Regulatory restrictions |
Variable |
Similar |
Stricter in some regions |
Similar |
FAQs
1. Is estazolam still widely prescribed globally?
While its use persists in many regions, especially where older benzodiazepines are favored, prescriptions are declining relative to newer agents due to safety concerns and regulatory restrictions.
2. What are the primary safety concerns associated with estazolam?
Dependence, tolerance, overdose potential, especially with long-term use, are primary considerations influencing prescribing patterns.
3. How does regulation affect the market potential of estazolam?
Stringent control schedules and prescription limits restrict sales, especially in North America and Europe, leading to a plateau or decline in mature markets.
4. Are there new formulations or delivery systems for estazolam?
Currently, no significant novel formulations exist; most sales are from traditional oral tablets. Generic manufacturers dominate supply.
5. What is the outlook for estazolam’s market in emerging economies?
Marked potential exists due to rising awareness of sleep disorders and less restrictive regulatory environments, promising moderate growth over the next 5-10 years.
Key Takeaways
- Estazolam maintains a niche role in sleep disorder treatment but faces increasing competition from non-benzodiazepine hypnotics and newer agents.
- Market revenue has stabilized in developed regions but shows growth potential in emerging markets.
- Regulatory landscapes heavily influence prescribing volumes; stricter controls limit future growth.
- Generic competition has reduced prices, impacting profitability for brand players.
- The future of estazolam hinges on balancing safety concerns, regulatory policies, and market expansion opportunities.
References
[1] Roth T., et al. "Insomnia: overview of prevalence, epidemiology, and consequences." Journal of Clinical Sleep Medicine, 2019.
[2] WHO. "International Classification of Diseases" (ICD-11), 2022.
[3] U.S. Patent and Trademark Office. Patent expiration data, 2000s.
[4] MarketWatch. "Sleep aids global market report," 2015.
This analysis provides a comprehensive overview of the current market landscape and future prospects of estazolam, equipping decision-makers with critical insights into its financial trajectory and underlying dynamics.