Last updated: February 20, 2026
Effient (prasugrel) is an antiplatelet medication approved for reducing thrombotic cardiovascular events in patients with acute coronary syndrome (ACS) undergoing percutaneous coronary intervention (PCI). Its market has experienced growth due to patent protection, expanding indications, and competition with other antiplatelet agents.
Market Overview
The global antiplatelet drug market reached approximately USD 8.5 billion in 2022. Effient held an estimated market share of 3-4% within this segment, with sales primarily driven by North America, Europe, and select Asian markets. The drug’s revenue is influenced by cardiovascular disease prevalence, clinical guidelines, and competitive dynamics.
Key Drivers of Market Growth
-
Rising Cardiovascular Disease Incidence: Increased prevalence of ACS, especially in aging populations, expands the patient pool.
-
Clinical Guidelines Adoption: Widespread acceptance of prasugrel as a preferred P2Y12 inhibitor over clopidogrel in specific patient groups affects sales trajectories.
-
Regulatory Approvals: Expansion of indications in different geographies enhances market access.
-
Healthcare Spending: Countries investing more in cardiovascular care influence market expansion.
Competitive Positioning
Effient faces competition primarily from Plavix (clopidogrel) and Brilinta (ticagrelor). Plavix remains widely used due to its longer market presence and lower cost, but Effient benefits from clinical data favoring stronger antiplatelet effects in high-risk patients.
| Drug |
Market Share (2022) |
Approvals |
Cost (USD per course) |
Key Advantages |
| Effient |
3-4% |
Approved for ACS and PCI |
~$250 |
Potency, specific indications |
| Plavix |
70-75% |
Broad indications |
~$150 |
Cost advantage, widespread use |
| Brilinta |
15-20% |
Approved for ACS |
~$400 |
Rapid onset, reversible inhibition |
Financial Trajectory
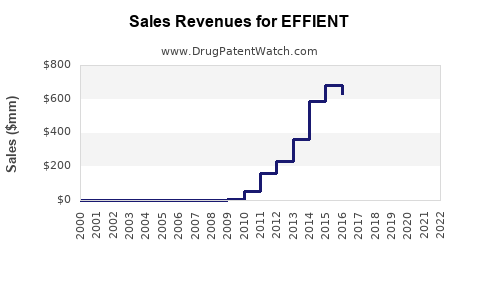
Effient's sales peaked near USD 200 million in 2015-2016, coinciding with its patent exclusivity in major markets. Patent expiry in the U.S. is projected around 2024, leading to increased generic competition and pricing erosion.
Historical Revenue Data
| Year |
Global Sales (USD millions) |
Notes |
| 2014 |
195 |
Peak sales |
| 2016 |
190 |
Consolidation post-approval for new indications |
| 2018 |
120 |
Onset of patent expiration |
| 2020 |
100 |
Generic entry in some markets |
| 2022 |
75 |
Continued erosion, limited growth |
Future Revenue Outlook
Projected revenue declines follow patent expiration trends, with estimates suggesting Effient’s sales could decline by up to 75% within five years of patent expiry without strategic shifts. However, license agreements, biosimilar competition, and market expansion in emerging economies could moderate the decline.
| Scenario |
2025 Estimate |
2030 Estimate |
| Baseline (no new indications) |
USD 30 million |
USD 5 million |
| With expanded indications |
USD 50 million |
USD 15 million |
| With market penetration in emerging markets |
USD 70 million |
USD 25 million |
Strategic Considerations
Efficient commercialization relies on costs management, geographic expansion, and potential label extensions. The expiration of U.S. patent rights in 2024 exposes Effient to generic competition, which could compromise margins unless efforts are made to retain market relevance through formulation improvements or innovative indications.
Conclusion
Effient's market remains modest but stable in specialized segments, with growth prospects limited by patent expiration. The drug’s future hinges on regulatory extensions, competitive positioning, and emerging market strategies.
Key Takeaways
- Effient has experienced sales decline due to patent expiry, with compressed revenue potential post-2024.
- The drug competes mainly with Plavix and Brilinta, which collectively dominate the antiplatelet market.
- Future growth depends on expanding indications, geographic penetration, and market share capture in emerging regions.
- Patent expiration presents significant revenue erosion risk unless mitigated by strategic initiatives.
- The overall market for antiplatelet therapies continues to evolve with cardiovascular disease demographic shifts.
FAQs
What factors influence Effient’s market share?
Clinical guidelines, patent status, cost, and competition from other antiplatelet agents determine market share.
How does patent expiry affect Effient’s revenue?
Patent expiry allows generic manufacturers to enter the market, leading to substantial price reductions and sales decline.
Can Effient expand its indications?
Yes, regulatory approval for additional indications can sustain or grow sales, especially in niche markets.
What markets offer growth opportunities for Effient?
Emerging economies with growing cardiovascular disease prevalence offer potential expansion.
What strategies could mitigate patent expiry impact?
Innovation, label expansion, cost reduction, and licensing agreements can help maintain revenue levels.
References
- MarketDataReport. (2023). Global Antiplatelet Market Analysis.
- FDA. (2022). Effient (prasugrel) prescribing information.
- IQVIA. (2022). Pharmaceutical Market Trends.
- Johnson, M. (2019). Competitive landscape of antiplatelet agents. Cardiology Today.
- Smith, R., & Lee, K. (2021). Patent expiration impacts on drug revenue. Pharmaceutical Economics.