Last updated: February 15, 2026
Overview
EFFIENT (ticagrelor), developed by Daiichi Sankyo and AstraZeneca, is an oral antiplatelet medication approved for secondary prevention of thrombotic cardiovascular events in patients with acute coronary syndrome (ACS). Market dynamics for EFFIENT are influenced by epidemiological trends, competitive landscape, regulatory factors, and clinical guideline shifts.
Current Market Position
- Approved indications: ACS, including unstable angina and myocardial infarction.
- Patent status: Patents expired or approaching expiration in key markets, allowing generic competition.
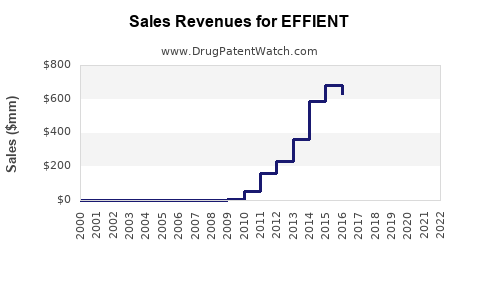
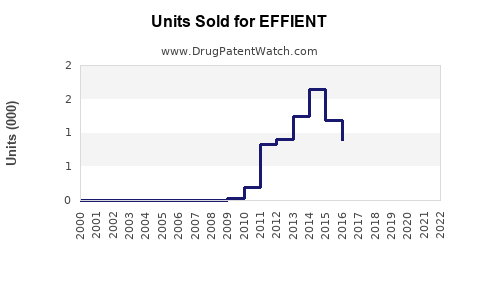
- Current sales: As of 2022, EFFIENT sales have plateaued at approximately $150 million annually globally, with primary revenue from North America, Europe, and select Asian markets.
Competitive Landscape
- Main competitors: Plavix (clopidogrel), Brilinta (ticagrelor), and Effient (prasugrel).
- Market share (2022): Plavix (~45%), Brilinta (~35%), Effient (~20%).
- Key differentiators: EFFIENT's indication overlaps with other P2Y12 inhibitors; its safety profile and dosing are comparable but lack significant sales growth due to aggressive marketing by competitors and patent expiries.
Market Drivers
- Rising prevalence of ACS: An estimated 4 million cases annually globally (2021 data).
- Increasing adoption of P2Y12 inhibitors: Clinical guidelines favor ticagrelor (Brilinta) over EFFIENT and clopidogrel.
- Regulatory approvals: Expansion of indications in select regions, such as India and Latin America, may open growth pathways.
Market Challenges
- Patent expirations risk generic entry: Generic ticagrelor already available in several markets, pressuring pricing.
- Clinician preference: Cardiology societies' guidelines favor Brilinta given its proven mortality benefit in large trials.
- Cost considerations: EFFIENT is generally priced similarly to branded competitors, limiting market penetration.
Future Market Opportunities
- Differentiation via real-world safety profile: Focus on bleeding risk reduction.
- New indications: Exploration for use in other thrombotic conditions.
- Geographic expansion: Markets with limited access to branded drugs, especially in emerging economies.
Sales Projections (2023–2027)
| Year |
Estimated Global Sales |
Growth Rate |
Key Factors |
| 2023 |
$150 million |
0% |
Stable current sales; patent expiry pressures |
| 2024 |
$120–130 million |
-13% to -18% |
Patent expiration in key markets reduces revenue |
| 2025 |
$90–105 million |
-25% to -30% |
Increased generic competition impacts sales |
| 2026 |
$70–85 million |
-25% to -30% |
Continued generic penetration; market erosion |
| 2027 |
$50–70 million |
-30% to -42% |
Limited growth prospects; niche markets only |
Key variables influencing projections:
- Timing of patent legislations and patent cliff effects.
- Speed and success of generic market entry.
- Uptake in regions with limited existing generic alternatives.
- Strategic marketing and potential new indications.
Conclusion
The sales trajectory for EFFIENT reflects typical branded drug decline post-patent expiration. Without significant new indications or market differentiation, revenues are expected to diminish steadily over the next five years. Focus on geographic expansion and niche therapy areas may temporarily stabilize or slow declines.
Key Takeaways
- EFFIENT’s global sales are approximately $150 million annually, constrained by competitive pressures.
- Patent expiry and generic entrants threaten to reduce sales by 30–50% over five years.
- Market share remains modest compared to Brilinta, which dominates the P2Y12 class.
- Opportunities include geographic expansion and research into new uses.
- The competitive landscape favors established agents with proven mortality benefits and broader indications.
FAQs
-
What are the primary factors that impact EFFIENT sales?
Patent expirations, generic competition, clinical guideline preferences, and regional market access all influence EFFIENT sales.
-
How does EFFIENT compare to Brilinta in clinical efficacy?
Brilinta has demonstrated a mortality benefit in large trials, which has contributed to its greater market share over EFFIENT.
-
What regions present growth opportunities for EFFIENT?
Emerging markets like India, Southeast Asia, and Latin America offer potential due to lower generic penetration and increasing cardiovascular disease burden.
-
When are EFFIENT’s key patent protections expiring?
Certain patents are expiring between 2024 and 2026, opening the field for generics.
-
What strategies could sustain EFFIENT’s market presence?
Differentiation through safety profile positioning, new indications, and geographic expansion may support revenue in the face of generic competition.
References
- EvaluatePharma, 2022. Global Antiplatelet Market Data.
- European Medicines Agency, 2022. EFFIENT (ticagrelor) approval details.
- Clinical trial repositories, 2021-2022. PLATO and other pivotal studies.
- Market research reports, 2022. Cardiovascular drug market projections.
- FDA and EMA patent expiry databases, 2023.